Summary
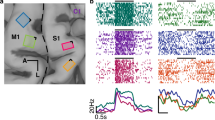
Previous studies have shown that the amplitude of somatosensory evoked potentials is diminished prior to, and during, voluntary limb movement. The present study investigated the role of the motor cortex in mediating this movement-related modulation in three chronically prepared, awake monkeys by applying low intensity intracortical microstimulation (ICMS) to different sites within the area 4 representation of the arm. Air puff stimuli were applied to the contralateral arm or adjacent trunk at various delays following the ICMS. Somatosensory evoked potentials were recorded from the primary somatosensory cortex, areas 1 and 3b, with an intracortical microelectrode. The principal finding of this study was that very weak ICMS, itself producing at most a slight, localized, muscle twitch, produced a profound decrease in the magnitude of the short latency component of the somatosensory evoked potentials in the awake money. Higher intensities of ICMS (suprathreshold for eliciting electromyographic (EMG) activity in the “target” muscle, i.e. that muscle activated by area 4 stimulation) were more likely to decrease the evoked response and produced an even greater decrease. The modulation appeared to be, in part, central in origin since (i) it preceded the onset of EMG activity in 23% of experiments, (ii) direct stimulation of the muscle activated by ICMS, which mimicked the feedback associated with the small ICMS-induced twitch, was often ineffective and (iii) the modulation was observed in the absence of EMG activity. Peripheral feedback, however, may also make a contribution. The results also indicate that the efferent signals from the motor cortex can diminish responses in the somatosensory cortex evoked by cutaneous stimuli, in a manner related to the somatotopic order. The effects are organized so that the modulation is directed towards those neurones serving skin areas overlying, or distal to, the motor output.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Armstrong DM, Drew T (1984) Topographical localization in the motor cortex of the cat for somatic afferent responses and evoked movements. J Physiol (Lond) 350: 33–54
Asanuma H, Rosén I (1972) Topographical organization of cortical efferent zones projecting to distal forelimb muscles in the monkey. Exp Brain Res 14: 243–256
Asanuma H, Arnold A, Zarzecki P (1976) Further study on the excitation of pyramidal tract cells by intracortical microstimulation. Exp Brain Res 26: 443–461
Asanuma H, Stoney SD, Abzug C (1968) Relationship between afferent input and motor outflow in cat motorsensory cortex. J Neurophysiol 31: 670–681
Bentivoglio M, Rustioni A (1986) Corticospinal neurons with branching axons to the dorsal column nuclei in the monkey. J Comp Neurol 253: 260–276
Bioulac B, Lamarre Y (1979) Activity of post-central cortical neurons of the monkey during conditioned movements of a deafferented limb. Brain Res 172: 427–437
Chapman CE, Jiang W, Lamarre Y (1988) Modulation of lemniscal input during conditioned arm movements in the monkey. Exp Brain Res 72: 316–334
Cheema S, Rustioni A, Whitsel BL (1985) Sensorimotor cortical projections to the primate cuneate nucleus. J Comp Neurol 240: 196–211
Cheney PD, Fetz EE (1985) Comparable patterns of muscle facilitation evoked by individual corticomotoneuronal (CM) cells and by single intracortical microstimuli in primates: evidence for functional groups of CM cells. J Neurophysiol 53: 786–804
Cohen LG, Starr A (1987) Localization, timing and specificity of gating of somatosensory evoked potentials during active movement in man. Brain 110: 451–467
Cole JD, Gordon G (1983) Timing of corticofugal actions on the gracile and cuneate nuclei of the cat. J Physiol (Lond) 340: 139–152
Connors BW, Malenka RC, Silva LR (1988) Two inhibitory postsynaptic potentials, and GABAA and GABAB receptormediated responses in neocortex of rat and cat. J Physiol (Lond) 406: 443–468
Coulter JD (1974) Sensory transmission through lemniscal pathway during voluntary movement in the cat. J Neurophysiol 37: 831–845
DeFelipe J, Conley M, Jones EG (1986) Long-range focal collateralization of axons arising from corticocortical cells in monkey sensory-motor cortex. J Neurosci 6: 3749–3766
Dyhre-Poulson P (1978) Perception of tactile stimuli before ballistic and during tracking movements. In: Gordon G (ed) Active touch. Pergamon Press, Oxford, pp 171–176
Evarts EV (1972) Contrasts between activity of precentral and postcentral neurons of cerebral cortex during movement in the monkey. Brain Res 40: 25–31
Evarts EV (1981) Role of motor cortex in voluntary movements in primates. In: Brooks VB (eds) Handbook of physiology, Sect 1. The nervous system, Vol II, Part 2. American Physiological Society, Bethesda MD, pp 1083–1120
Fromm C, Evarts EV (1982) Pyramidal tract neurons in somatosensory cortex: central and peripheral inputs during voluntary movement. Brain Res 238: 186–191
Gardner EP, Hamalaïnen HA, Warren S, Davis J, Yong W (1984) Somatosensory evoked potentials (SEPs) and cortical single unit responses elicited by mechanical tactile stimuli in awake monkeys. Electroenceph Clin Neurophysiol 58: 537–552
Ghez C, Lenzi GL (1971) Modulation of sensory transmission in cat lemniscal system during voluntary movement. Pflügers Arch 323: 273–278
Ghez C, Pisa M (1972) Inhibition of afferent transmission in cuneate nucleus during voluntary movement in the cat. Brain Res 40: 145–151
Giuffrida R, Sanderson P, Sapienza S (1985) Effects of microstimulation of movement-evoked cortical foci on the activity of neurons in the dorsal column nuclei. Somatosens Res 2: 237–247
Harris H, Jabbur SJ, Morse RW, Towe AL (1965) Influence of the cerebral cortex on the cuneate nucleus of the monkey. Nature 208: 1215–1216
Humphrey D, Corrie WS (1978) Properties of pyramidal tract neuron system within a functionally defined subregion of primate motor cortex. J Neurophysiol 41: 216–243
Jabbur SJ, Towe AL (1961) Cortical excitation of neurons in dorsal column nuclei of cat, including an analysis of pathways. J Neurophysiol 24: 499–509
Jiang W, Chapman CE, Lamarre Y (1988) The topographical modulation of somatosensory transmission to SI cortex produced by microstimulation of MI cortex in the monkey. Abstr Soc Neurosci 14: 509
Jiang W, Lamarre Y, Chapman CE (1986) Modulation of somatosensory input to sensory cortex produced by intracortical microstimulation of motor cortex in the monkey. Abstr Soc Neurosci 12: 259
Jones EG (1975) Some aspects of the organization of the thalamic reticular complex. J Comp Neurol 162: 285–308
Jones EG, Coulter JD, Hendry SHC (1978) Intracortical connectivity of architectonic fields in the somatic sensory, motor and parietal cortex of monkeys. J Comp Neurol 181: 291–348
Jones EG, Wise SP (1977) Size, laminar and columnar distribution of efferent cells in the sensory-motor cortex of monkeys. J Comp Neurol 175: 391–438
Jones SF, Halonen J-P, Shawkat F (1989) Centrifugal and centripetal mechanisms involved in the ‘gating’ of cortical SEPs during movement. Electroenceph Clin Neurophysiol 74: 36–45
Kuypers HGJM (1960) Central cortical projections to motor and somato-sensory cell groups: an experimental study in the rhesus monkey. Brain 83: 161–184
Lamarre Y, Joffroy AF, Filion M, Bouchoux R (1970) A stereotaxic method for repeated sessions of central unit recording in the paralyzed or moving animal. Rev Can Biol 29: 371–376
Lemon RN, Muir RB, Mantel GWH (1987) The effects upon the activity of hand and forearm muscles of intracortical stimulation in the vicinity of corticomotor neurones in the conscious monkey. Exp Brain Res 66: 621–637
Murphy JT, Kwan HC, MacKay WA, Wong YC (1978) Spatial organization of precentral cortex in awake primates. III. Input-output coupling. J Neurophysiol 41: 1132–1139
Powell TPS, Mountcastle VB (1959) Some aspects of the functional organization of the cortex of the postcentral gyrus of the monkey: a correlation of findings obtained in a single unit analysis with cytoarchitecture. Bull Johns Hopkins Hosp 105: 133–162
Ranck Jr JB (1975) which elements are excited in electrical stimulation of mammalian central nervous system: a review. Brain Res 98: 417–440
Rosén I, Asanuma H (1972) Peripheral afferent inputs to the forelimb area of the monkey motor cortex: input-output relations. Exp Brain Res 14: 257–273
Rushton DN, Rothwell JC, Craggs MD (1981) Gating of somatosensory evoked potentials during different kinds of movement in man. Brain 104: 465–491
Soso MF, Fetz EE (1980) Responses of identified cells in postcentral cortex of awake monkeys during comparable active and passive joint movements. J Neurophysiol 43: 1090–1110
Stoney SD, Thompson WD, Asanuma H (1968) Excitation of pyramidal tract cells by intracortical microstimulation: effective extent of stimulating current. J Neurophysiol 31: 659–669
Towe AL, Jabbur SJ (1961) Cortical inhibition of neurons in dorsal column nuclei of cat. J Neurophysiol 24: 488–498
Tsumoto T, Nakamura S, Iwama K (1975) Pyramidal tract control over cutaneous and kinesthetic sensory transmission in the cat thalamus. Exp Brain Res 22: 281–294
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jiang, W., Chapman, C.E. & Lamarre, Y. Modulation of somatosensory evoked responses in the primary somatosensory cortex produced by intracortical microstimulation of the motor cortex in the monkey. Exp Brain Res 80, 333–344 (1990). https://doi.org/10.1007/BF00228160
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00228160