Summary
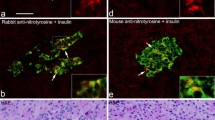
Islet amyloid peptide (or diabetes-associated peptide), the major component of pancreatic islet amyloid found in type-2 diabetes, has been identified by electronmicroscopic immunocytochemistry in pancreatic B-cells from five non-diabetic human subjects, and in islets from five type-2 diabetic patients. The greatest density of immunoreactivity for islet amyloid peptide was found in electrondense regions of some lysosomal or lipofuscin bodies. The peptide was also localised by quantification of immunogold in the secretory granules of B-cells, and was present in cytoplasmic lamellar bodies. Acid phosphatase activity was also demonstrated in these organelles. Immunoreactivity for insulin was found in some lysosomes. These results suggest that islet amyloid peptide is a constituent of normal pancreatic B-cells, and accumulates in lipofuscin bodies where it is presumably partially degraded. In islets from type-2 diabetic subjects, amyloid fibrils and lipofuscin bodies in B-cells showed immunoreactivity for the amyloid peptide. Abnormal processing of the peptide within B-cells could lead to the formation of islet amyloid in type-2 diabetes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Barren AJ, Davies ME, Grubb A (1984) The place of human gamma trace (cystatin C) amongst the cysteine proteinase inhibitors. Biochem Biophys Res Commun 120:631–636
Bell ET (1959) Hyalinization of the islets of Langerhans in nondiabetic individuals. Am J Pathol 35:801–805
Borg LAH, Schnell AH (1986) Lysosomes and pancreatic islet function: intracellular insulin degradation and lysosomal transformations. Diabetes Res 33:277–285
Carrel RW (1988) Alzheimer's disease: enter a protease inhibitor. Nature 331:478–479
Chio KS, Tappel AL (1969) Synthesis and characterization of fluorescent products derived from malonaldehyde and amino acids. Biochemistry 8:2821–2827
Clark A, Holman RR, Matthews DR, Hockaday TDR, Turner RC (1984) Non-uniform distribution of islet amyloid in the pancreas of ‘maturity-onset’ diabetic patients. Diabetologia 27:527–528
Clark A, Cooper GJS, Lewis CE, Morris JF, Willis AC, Reid KBM, Turner RC (1987) Islet amyloid formed from diabetes associated peptide may be pathogenic in type 2 diabetes. Lancet ii: 231–234
Clark A, Morris JF, Ostle LR, Edwards CA, Lewis CE, Sutton R, Britton BJ, Cooper GJS, Turner RC, Rothbard JB (1988a) Ultrastructural identification of diabetes associated peptide/ islet amyloid polypeptide in secretory granules and lysosomes of B-cells in human and monkey pancreatic islets. Diabetic Med 5:A20
Clark A, Wells CA, Buley ID, Cruickshank JK, Vanhegan RI, Matthews DR, Cooper GJS, Holman RR, Turner RC (1988b) Islet amyloid, increased A-cells, reduced B-cells and exocrine fibrosis: quantitative changes in the pancreas in type 2 diabetes. Diabetes Res 9:151–160
Cooper GJS, Willis AC, Clark A, Turner RC, Sim RB, Reid KBM (1987) Purification and characterisation of a peptide from amyloid-rich pancreases of type 2 diabetic patients. Proc Natl Acad Sci USA 84:8628–8632
Davidson HW, Hutton JC (1987) The insulin-secretory-granule carboxypeptidase H: purification and demonstration of involvement in proinsulin processing. Biochem J 245:575–585
Ehrlich JC, Ratner IM (1961) Amyloidosis of the islets of Langerhans. Am J Pathol 38:49–59
Gomori G (1941) Distribution of acid phosphatase in the tissues under normal and under pathologic conditions. Arch Pathol 32:189–199
Halban PA, Wollheim CB, Blondel B, Niesor E, Renold AE (1979) Perturbation of hormone storage and release induced by cyproheptadine in rat pancreatic islets in vitro. Endocrinology 104:1096–1106
Halban PA, Wollheim CB (1980) Intracellular degradation of insulin stores by rat pancreatic islets in vitro: an alternative pathway for homeostasis of pancreatic insulin content. J Biol Chem 13:6003–6006
Howard CF (1986) Longitudinal studies on the development of diabetes in individual Macaca nigra. Diabetologia 29:301–306
Jayne EP (1950) Cytochemical studies of age pigments in the human heart. J Gerontol 5:319–325
Johnson EH, O'Brien TD, Hayden DW, Jordan K, Ghobrial HKG, Mahoney WC, Westermark P (1988) Immunolocalization of islet amyloid polypetide (IAPP) in pancreatic beta cells by means of peroxidase-antiperoxidase (PAP) and protein A-gold techniques. Am J Pathol 130:1–8
Mann DMA, Yates PO (1974) Lipoprotein pigments: their relationship to ageing in the human nervous system 1. The lipofuscin content of nerve cells. Brain 97:481–488
Meda P (1978) Lysosomes in normal pancreatic beta-cells. Diabetologia 14:305–310
Mosselman S, Hoppener JWM, Zandberg J, van Mansfeld ADM, Geurts van Kessel AHM, Lips CJM, Janz HS (1988) Islet amyloid polypeptide: identification and chromosomal localization of the human gene FEBS Lett 239:227–232
Opie E (1901) The relation of diabetes mellitus to lesions of the pancreas. Hyaline degeneration of the islets of Langerhans. J Exp Med 5:527–540
Orci L, Ravazzola M, Amherdt M, Yanaihara C, Yanaihara N, Halban P, Renold AE, Perrelet A (1984) Insulin, not C-peptide (proinsulin) is present in crinophagic bodies of the pancreatic B-cell. J Cell Biol 98:222–228
Orci L, Ravazzola M, Amherdt M, Madsen O, Perrelet A (1986) Conversion of proinsulin to insulin occurs coordinately with acidification of maturing secretory vesicles. J Cell Biol 103:2273–2281
Palsdottir A, Abrahamson M, Thorsteinsson L, Arnason A, Olafsson I, Grubb A, Jensson O (1988) Mutation in cystatin C gene causes hereditary brain haemorrhage. Lancet ii: 603–604
Ponte P, Gonzalez-de-Whitt P, Schilling J, Miller J, Hus D, Greenberg B, Davis K, Wallace W, Lieberburg I, Fuller F, Cordell B (1988) A new A4 amyloid mRNA contains a domain homologous to serine proteinase inhibitors. Nature 331:525–527
Pras M, Prelli F, Franklin EC, Frangione B (1983) Primary structure of an amyloid prealbumin variant in familial polyneuropathy of Jewish origin. Proc Natl Acad Sci USA 30:539–542
Rosenfeld MG, Mermod JJ, Amara SG, Swanson LW, Sawchenko PE, Rivier J, Vale WW, Evans RN (1983) Production of a novel neuropeptide encoded by the calcitonin gene via tissuespecific RNA processing. Nature 304:129–135
Sanke T, Bell GI, Sample C, Rubenstein AH, Steiner DF (1988) An islet amyloid peptide is derived from an 89-amino acid precursor by proteolytic processing. J Biol Chem 263:17243–17246
Schneider HM, Storkel S, Will W (1980) Das Amyloid der Langerhansschen Inseln und seine Beziehung zum Diabetes mellitus. Dtsch Med Wochenschr 105:1143–1147
Skoglund G, Ahren B, Lundquist I (1987) Biochemical determination of islet lysosomal enzyme activities following crinophagystimulating treatment with diazoxide in mice. Diabetes Res 6:81–84
Westermark P (1973) Fine structure of the islets of Langerhans in insular amyloidosis. Virchows Arch [A] 359:1–8
Westermark P, Wernstedt C, Wilander E, Hayden DW, O'Brien TD, Johnson KH (1987a) Amyloid fibrils in human insulinoma and islets of Langerhans of the diabetic cat are derived from a neuropeptide-like protein also present in normal islet cells. Proc Natl Acad Sci USA 84:3881–3885
Westermark P, Wernstedt C, O'Brien TD, Hayden DW, Johnson KH (1987b) Islet amyloid in type 2 human diabetes mellitus and adult diabetic cats contains a novel putative polypeptide hormone. Am J Pathol 127:414–417
Westermark P, Wilander E, Westermark GT, Johnson KH (1987c) Islet amyloid polypeptide-like immunoreactivity in the islet B-cells of type 2 (non-insulin dependent) diabetic and non-diabetic individuals. Diabetologia 30:887–892
Zwahlen R, Richardson BP, Hauser RE (1979) The production and elimination of myeloid bodies by cultured pancreatic islet cells. J Ultrastruct Res 67:340–356
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Clark, A., Edwards, C.A., Ostle, L.R. et al. Localisation of islet amyloid peptide in lipofuscin bodies and secretory granules of human B-cells and in islets of type-2 diabetic subjects. Cell Tissue Res. 257, 179–185 (1989). https://doi.org/10.1007/BF00221649
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00221649