Summary
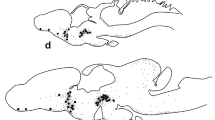
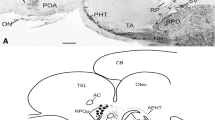
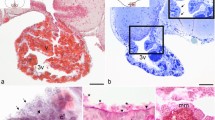
In cichlid, poecilid and centrarchid fishes luteinizing hormone releasing hormone (LHRH)-immunoreactive neurons are found in a cell group (nucleus olfactoretinalis) located at the transition between the ventral telencephalon and olfactory bulb. Processes of these neurons project to the contralateral retina, traveling along the border between the internal plexiform and internal nuclear layer, and probably terminating on amacrine or bipolar cells. Horseradish peroxidase (HRP) injected into the eye or optic nerve is transported retrogradely in the optic nerve to the contralateral nucleus olfactoretinalis where neuronal perikarya are labeled. Labeled processes leave this nucleus in a rostral direction and terminate in the olfactory bulb. The nucleus olfactoretinalis is present only in fishes, such as cichlids, poecilids and centrarchids, in which the olfactory bulbs border directly the telencephalic hemispheres. In cyprinid, silurid and notopterid fishes, in which the olfactory bulbs lie beneath the olfactory epithelium and are connected to the telencephalon via olfactory stalks, the nucleus olfactoretinalis or a comparable arrangement of LHRH-immunoreactive neurons is lacking. After retrograde transport of HRP in the optic nerve of these fishes no labeling of neurons in the telencephalon occurred. It is proposed that the nucleus olfactoretinalis anatomically and functionally interconnects and integrates parts of the olfactory and optic systems.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ariëns-Kappers CU, Huber GC, Crosby EC (1967) The comparative anatomy of the nervous system of vertebrates including man. Vol III, Hafner, New York
Baker BL, Dermody WC, Reel JR (1975) Distribution of gonadotropin-releasing hormone in the rat brain as observed with immunocytochemistry. Endocrinology 97:125–135
Barry J (1979) Extra-hypophyseal immunoreactive LH-RH-pathways in primates. Proc Int Symp Neuroendocrinol Regulatory Mech, pp 13–25
Barry J, Dubois MP, Poulain P (1973) LRF producing cells of the mammalian hypothalamus. Z Zellforsch 146:351–366
Bonin W (1941) Le nerf terminal et son ganglion. Naturaliste Canadien 68:33–50
Brookover C, Jackson TS (1911) The olfactory nerve and the nervus terminalis of Ameiurus. J Comp Neurol 21:237
Cowan WM, Clarke PGH (1976) The development of the isthmo optic nucleus. Brain Behav Evol 13:345–275
Crim LW, Evans DM (1980) LH-RH stimulated gonadotropin release from the rainbow trout pituitary gland: an in vitro assay for detection of teleost gonadotropin releasing factor(s). Gen Comp Endocrinol 40:283–290
Crim JW, Dickhoff WW, Gorbman A (1978) Comparative endocrinology of piscine hypothalamic hypophysiotropic peptides: distribution and activity. Am Zool 18:411–424
Dubois MP, Billard R, Breton B, Peter RE (1979) Comparative distribution of somatostatin, LH-RH, neurophysin, and alpha-endorphin in the rainbow trout: an immunocytological study. Gen Comp Endocrinol 37:220–232
Ebbesson SOE, Meyer DL (1981) Efferents to the retina have multiple sources in teleost fishes. Science, in press
Ferguson JL, Mulvanny JP, Brauth SE (1978) Distribution of neurons projecting to the retina of Caiman crocodilus Brain Behav Evol 15:294–306
Fritzsch B, Himsteadt W (1981) Pretectal neurons project to the salamander retina. Neurosci Lett 24:13–17
Goos HJTH, Murathanoglu O (1977) Localisation of gonadotropin releasing hormone (GRH) in the forebrain and neurohypophysis of the trout (Salmo gairdneri). Cell Tissue Res 81:163–168
Halpern M, Wang RT, Colman DR (1976) Centrifugal fibers to the eye in a nonavian vertebrate: Source revealed by horseradish peroxidase studies. Science 194:1185–1188
Holmgren N (1920) Zur Anatomie und Histologie des Vorderund Zwischenhirns der Knochenfische. Acta Zool 1:137–315
Hoogland PV, Welker E (1981) Telencephalic projections to the eye in Python reticularis. Brain Res 213:173–176
Itaya SK (1980) Retinal efferents from the pretectal area in the rat. Brain Res 201:436–441
Jennes L, Stumpf WE (1980a) LHRH-systems in the brain of the golden hamster. Cell Tissue Res 209:239–256
Jennes L, Stumpf WE (1980b) LHRH-neuronal projections to the inner and outer surface of the brain. Neuroendocrinol Lett 2:241–245
Kudo K (1923) Contribution to the knowledge of the brain of bony fishes. Kon Akad v Wetensch Amsterdam, Proc sect sc 26:65
La Vail J, La Vail MM (1972) Retrograde axonal transport in the central nervous system. Science 176:1416–1472
Malmgren L, Olsson Y (1978) A sensitive method for histochemical demonstration of horseradish peroxidase in neurons following retrograde axonal transport. Brain Res 148:279–294
Margolis-Kazan H, Peute J, Schreibman MP, Halpern LR (1981) Ultrastructural localization of gonadotropin and luteinizing hormone releasing hormone in the pituitary gland of a teleost fish (the platyfish). J Exp Zool 215:99–102
Maturana HR, Frenk S (1965) Synaptic connections of the centrifugal fibers in the pigeon retina. Science 150:359–361
Meyer DL, Ebbesson SOE (1981) Retinofugal and retinopetal connections in the upside-down catfish (Synodontis nigriventris). Cell Tissue Res 218:389–401
Meyer DL, Fibig E, Ebbesson SOE (1981) A note on the reciprocal connections between the retina and the brain in the puffer fish Tetraodon fluviatilis. Neurosci Lett 23:111–115
Miles FA (1970) Centrifugal effects in the avian retina. Science 170:992–995
Münz H, Claas B (1981) Centrifugal innervation of the retina in cichlid and poecilid fishes. A horseradish peroxidase study. Neurosci Lett 22:223–226
Münz H, Stumpf WE, Jennes L (1981) LHRH-systems in the brain of platyfish. Brain Res 221:1–13
Nieuwenhuys R (1967) Comparative anatomy of olfactory centres and tracts. Progr Brain Res 23:1–64
Northcutt GR, Braford MR Jr (1980) New observations on the organization and evolution of the telencephalion of actinopterygian fishes. In: Ebbesson SOE (ed) Comparative neurology of the telencephalon. Plenum Press, New York London, pp 41–98
Nozaki M, Kobayashi H (1979) Distribution of LHRH-like substance in the vertebrate brain as revealed by immunohistochemistry. Arch Histol Jpn 42:201–219
Peyrichoux J, Weidner C, Repérant J, Miceli D (1977) An experimental study of the visual system of cyprinid fish using HRP method. Brain Res 130:531–537
Repérant J, Vesselkin NP, Rio JP, Ermakova TV, Miceli D, Peyrichoux J, Weidner C (1981) La voie visuelle centrifuge n'existe-t-elle que chez les oiseaux? Rev Can Biol 40:29–46
Rossi A, Palombi F (1969) A neurofibrillar body in the perikaryon of nervus terminalis ganglion cells in teleosts. Z Zellforsch 93:395–403
Sandeman DC, Rosenthal NP (1974) Efferent axons in the fish optic nerve and their effect on the retinal ganglion cells. Brain Res 68:41–54
Schmidt JT (1979) The laminar organization of optic nerve fibres in the tectum of goldfish. Proc R Soc Lond (Biol) 205:287–306
Schreibman MP, Halpern R, Goos HJTh, Margolis-Kazan H (1979) Identification of luteinizing hormone-releasing hormone (LHRH) in the brain and pituitary gland of a fish by immunocytochemistry. J Exp Zool 210:153–160
Schwanzel-Fukuda M, Silverman AJ (1980) The nervus terminalis of the guinea pig: a new luteinizing hormone releasing hormone (LHRH) neuronal system. J Comp Neurol 191:213–225
Sheldon RE (1909) The nervus terminals in the carp. J Comp Neurol 19:191–201
Silverman AJ, Krey LC (1978) The luteinizing hormone releasing hormone (LHRH) neuronal networks of the guinea pig brain. I. Intra and extra-hypothalamic projections. Brain Res 157:233–246
Stell WK (1972) The morphological organization of the vertebrate retina. In: Fuortes MGF (ed) Handbook of sensory physiology Vol VII/2. Springer, Berlin, pp 129–213
Vanegas H, Amat J, Essayag-Millan E (1973) Electrophysiological evidence of tectal efferents to the fish eye. Brain Res 54:309–313
Wagner HJ (1973) Die nervösen Netzhautelemente von Nannacara anomala (Cichlidae, Teleostei). Z Zellforsch 137:63–86
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Münz, H., Claas, B., Stumpf, W.E. et al. Centrifugal innervation of the retina by luteinizing hormone releasing hormone (LHRH)-immunoreactive telencephalic neurons in teleostean fishes. Cell Tissue Res. 222, 313–323 (1982). https://doi.org/10.1007/BF00213215
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00213215