Abstract
The relative bioavailability of nicotine from a nasal spray was assessed in 15 smokers suffering a common cold and rhinitis according to generally accepted criteria. The patients were given a single dose of 2 mg nicotine from the nasal spray with and without concurrent administration of a nasal vasoconstrictor decongestant, xylometazoline, in randomised order. Control session measurements were made in the disease-free state.
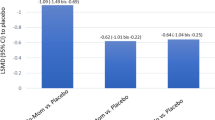
Applying strict bioequivalence criteria, we found that common cold/rhinitis slightly reduced the bioavailability of nicotine, both in its rate and extent; the geometric mean of the ratio of Cmax, AUC and tmax were 0.81, 0.93 and 1.36, respectively. The nasal vasoconstrictor, xylometazoline, normalised the extent of the bioavailability of nicotine, but further prolonged the time for absorption to almost twice that measured in the disease-free state, increasing the tmax ratio to 1.72.
The results suggest that a minor proportion of people stopping smoking with the help of a nicotine nasal spray may experience a minor reduction in the effect of the spray during common cold/rhinitis. However, the nicotine self-titration behaviour found with most smoking cessation products (except the nicotine patch) will automatically lead to an adjustment of the dosage to achieve the desired effect.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Pontiroli AE, Calderara A, Pozza G (1989) Intranasal Drug Delivery. Potential advantages and limitations from a clinical pharmacokinetic perspective. Clin Pharmacokinet 17: 299–307
Russel MAH, Jarvis MJ, Feyerabend C (1980) A new age for snuff? Lancet I: 474–475
Russel MAH, Jarvis MJ, Feyerabend C, Fernö O (1983) Nasal nicotine solution: a potential aid to give up smoking? Br Med J 286: 683–684
Jarvis MJ, Hajek P, Russel MAH, West RJ, Feyerabend C (1987) Nasal nicotine solution as an aid to cigarette withdrawal: a pilot clinical trial. Br J Addict 82: 983–988
Schneider NG (1992) Nicotine therapy in smoking cessation. Pharmacokinetic considerations. Clin Pharmacokinet 23(3): 169–172
Jackson GG, Dowling HF, Spiesman IG, Boand AV (1958) Transmission of the common cold to volunteers under controlled conditions. Arch Intern Med 101: 267–278
Olsson P, Kuylenstierna F, Johansson C-J, Gunnar PO, Bende M (1991) Pharmacokinetics of nicotine after intranasal administration. In: Adlkofer F (ed) Effects of nicotine on biological systems. Advances in pharmacologica sciences. Birkhäuser, Basel, pp 57–61
Sutherland G, Stapleton JA, Russell MAH et al (1992) Randomised controlled trial of nicotine nasal spray in smoking cessation. Lancet 340: 324–329
Hjalmarsson A, Franzon M, Westin Å, Wiklund O (1994) Effect of nicotine nasal spray in smoking cessation. A randomised, placebo-controlled, double-blind study. Arch Int Med 154: 2567–2572
Greiff L, Wollmer P, Svensson C, Andersson M, Persson CGA (1993) Effect of seasonal allergic rhinitis on airway mucosal absorption of chromium-51 labelled EDTA. Thorax 48: 648–650
Greiff L, Svensson C, Andersson M, Åkerlund A, Wollmer P, Alkner U, Persson CGA (1993) Increased exudative responsiveness in allergen and corona virus-induced airway inflammation. Allergy 48: 91
Persson CGA, Erjefält I, Alkner U, Baumgarten C, Greiff L, Gustafsson B, et al (1991) Plasma exudation as a first line respiratory mucosal defence. Clin Exp Allergy 21: 17–24
Bende M (1983) The effect of topical decongestant on blood flow in normal and infected nasal mucosa. Acta Otolaryngol (Stockh) 96: 523–527
Russell MAH (1976) Tobacco smoking and nicotine dependence. In: Gibbins, et al (eds) Researche advances in alcohol and drug problems, vol 3. Wiley, New York, pp 1–47
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lunell, E., Molander, L. & Andersson, M. Relative bioavailability of nicotine from a nasal spray in infectious rhinitis and after use of a topical decongestant. Eur J Clin Pharmacol 48, 71–75 (1995). https://doi.org/10.1007/BF00202176
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00202176