Summary
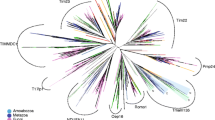
The thiolase family is a widespread group of proteins present in prokaryotes and three cellular compartments of eukaryotes. This fact makes this family interesting in order to study the evolutionary process of eukaryotes. Using the sequence of peroxisomal thiolase from Saccharomyces cerevisiae recently obtained by us and the other known thiolase sequences, a phylogenetic analysis has been carried out. It shows that all these proteins derived from a primitive enzyme, present in the common ancestor of eubacteria and eukaryotes, which evolved into different specialized thiolases confined to various cell compartments. The evolutionary tree obtained is compatible with the endosymbiotic theory for the origin of peroxisomes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Arakawa H, Takiguchi M, Amaya Y, Nagata S, Hayashi H, Mori M (1987) cDNA-derived amino acid sequence of rat mitochondrial 3-oxoacyl-CoA thiolase with no transient presequence: structural relationship with peroxisomal isozyme. EMBO J 6:1361–1366
Bodnar AG, Rachubinski RA (1990) Cloning and sequence determination of cDNA encoding a second rat liver peroxisomal 3-ketoacyl-CoA thiolase. Gene 91:193–199
Borst P (1989) Peroxisome biogenesis revisited, Biochim Biophys Acta 1008:1–13
Bout A, Teunissen Y, Hashimoto T, Benne R, Tager JM (1988) Nucleotide sequence of human peroxisomal 3-oxoacyl-CoA thiolase. Nucleic Acids Res 16:10369
Brinkmann H, Martinez P, Quigley F, Martin W, Cerff R (1978) Endosymbiotic origin and codon bias of the nuclear gene for chloroplast glyceraldehyde-3-phosphate dehydrogenase from maize. J Mol Evol 26:320–328
Cavalier-Smith T (1987) The simultaneous symbiotic origin of mitochondria, chloroplasts and microbodies. Ann NY Acad Sci 503:55–71
Clinkenbear KA, Sugiyama T, Moss J, Reed WA, Lave MD (1973) Molecular and catalytic properties of cytosolic acetoacetyl coenzyme A thiolase from avian liver. J Biol Chem 248:2275–2284
Cohen G, Rapatz W, Ruis H (1988) Sequence of the Saccharomyces cerevisiae CTA 1 gene and amino acid sequence of catalase A derived from it. Eur J Biochem 176:159–163
de Duve C (1983) Microbodies in the living cell. Sci Am 248: 74–84
Dequin S, Gloeckler R, Herbert CJ, Boutelet F (1988) Cloning, sequencing, and analysis of the yeast S. uvarum ERG10 gene encoding acetoacetyl CoA thiolase. Curr Genet 13:471–478
Duncombe GR, Frerman FE (1976) Molecular and catalytic properties of the acetoacetyl-CoA thiolase of Escherichia coli. Arch Biochem Biophys 176:159–170
Efron B (1982) The jackknife, the bootstrap and other resampling plans. Society for Industrial and Applied Mathematics, Philadelphia
Fairbairn LJ, Tanner MIA (1989) Complete cDNA sequence of human foetal liver peroxisomal 3-oxoacyl-CoA thiolase. Nucleic Acids Res 17:3588
Felsenstein J (1984) Distance methods for inferring phylogenies: a justification. Evolution 39:16–24
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791
Felsenstein J (1990) PHYLIP manual version 3.3. University Herbarium, University of California, Berkeley CA
Fitch MW, Margoliash E (1967) The construction of phylogenetic trees. Science 155:279–284
Frevert J, Kindl H (1980) Purification of glyoxysomal acetyl-acyltransferase. Hoppe-Seyler's Z Physiol Chem 361: 537–542
Fukao T, Kamijo K, Osumi T, Fujiki Y, Yamaguchi S, Orii T, Hashimoto T (1989) Molecular cloning and nucleotide sequence of cDNA encoding the entire precursor of rat mitochondrial acetoacetyl-CoA thiolase. J Biochem 106:197–204
Fukao T, Yamaguchi S, Kano M, Orii T, Fujiki Y, Osumi T, Hashimoto T (1990) Molecular cloning and sequence of the complementary DNA encoding human mitochondrial acetoacetyl-CoA thiolase and study of the variant enzymes in cultured fibroblasts from patients with 3-ketothiolase deficiency. J Clin Invest 86:2086–2092
Gehring U, Harris JI (1970) The active site cysteines of thiolase. Eur J Biochem 16:492–498
Higgins DG, Sharp PM (1989) Fast and sensitive multiple sequence alignment on a microcomputer. Comput Appl Biosci 5:151–153
Hijikata M, Ishii N, Kagamiyama H, Osumi, Hashimoto T (1987) Structural analysis of cDNA for rat peroxisomal 3-ketoacyl-CoA thiolase. J Biol Chem 262:8151–8158
Hijikata M, Wen JK, Osumi T, Hashimoto T (1990) Rat peroxisomal 3-ketoacyl-CoA thiolase gene. Occurrence of two closely related but differentially regulated genes. J Biol Chem 265:4600–4606
Igual JC, Matallana E, Gonzalez-Bosch C, Franco L, Perez-Ortin JE (1991) A new glucose-repressible gene identified from the analysis of chromatin structure in deletion mutants of yeast SUC2 locus. Yeast 7:379–389
Imamura S, Ueda S, Mizugaki M, Kawaguchi A (1990) Purification of the multienzyme complex for fatty acid oxidation from Pseudomonas fragi and reconstitution of fatty acid oxidation system. J Biochem 107:184–189
Kimura M (1983) The neutral theory of molecular evolution. Cambridge University Press, Cambridge
Kornblatt JA, Rudney H (1971) Two forms of acetoacetyl co-enzyme A thiolase in yeast. Intracellular location and relationship to growth. J Biol Chem 246:4424–4430
Kunau WH, Bühne S, de la Garza M, Kionka C, Mateblowski M, Schutz-Borchard U, Thieringer R (1988) Comparative enzymology of β-oxidation. Biochem Soc Trans 16:418–420
Kurihara T, Ueda M, Tanaka A (1988) Occurrence and possible roles of acetoacetyl-CoA thiolase and 3-ketoacyl-CoA thiolase in peroxisomes of an n-alkane-grown yeast, Candida tropicalis. FEBS Lett 229:215–218
Kurihara T, Ueda M, Tanaka A (1989) Peroxisomal acetoacetyl-CoA thiolase and 3-ketoacyl-CoA thiolase from an n-alkane-utilizing yeast, Candida tropicalis: purification and characterization. J Biochem 106:474–478
Lazarow PB, Fujiki Y (1985) Biogenesis of peroxisomes. Annu Rev Cell Biol 1:489–530
Masamune S, Palmer MAJ, Gambani R, Thompson S, Davis JT, Williams SF, Peoples OP, Sinskey AJ, Walsh CT (1989) Bio-Claisen condensation catalyzed by thiolase from Zoogloea ramigera. Active site cysteine residues. J Am Chem Soc 111:1879–1881
Middleton B (1973) The oxoacyl-CoA thiolases of animal tissues. Biochem J 132:717–730
Minard KI, McAlister-Henn L (1991) Isolation, nucleotide sequence analysis, and disruption of the MDH2 gene from Saccharomyces cerevisiae: evidence for three isozymes of yeast malate dehydrogenase. Mol Cell Biol 11:370–380
Miyazawa S, Osumi T, Hashimoto T (1980) The presence of a new 3-oxoacyl-CoA thiolase in rat liver peroxisomes. Eur J Biochem 103:589–596
Miyazawa S, Furuta S, Osumi T, Hashimoto T, Ui N (1981) Properties of peroxisomal 3-ketoacyl-CoA thiolase from rat liver. J Biochem 90:511–519
Mori T, Tsukamoto T, Mori H, Tashiro Y, Fujiki Y (1991) Molecular cloning and deduced amino acid sequence of nonspecific lipid transfer protein (sterol carrier protein 2) of rat liver: a higher molecular mass (60 kDa) protein contains the primary sequence of nonspecific lipid transfer protein as its C-terminal part. Proc Natl Acad Sci USA 88:4338–4342
Nugent JM, Palmer JD (1991) RNA-mediated transfer of the gene coxII from the mitochondrion to the nucleus during flowering plant evolution. Cell 66:473–481
Osendorp C, van Heusden PH, de Beer ALJ, Bos K, Schouten GL, Wirtz KWA (1991) Identification of the cDNA clone which encodes the 58-kDa protein containing the amino acid sequence of rat liver non-specific lipid-transfer (sterol-carrier protein 2). Homology with rat peroxisomal and mitochondrial 3-oxoacyl-CoA thiolases. Eur J Biochem 201:233–239
Pawar S, Schulz H (1981) The structure of the multienzyme complex of fatty acid oxidation from Escherichia coli. J Biol Chem 256:3894–3899
Peoples OP, Masamune S, Walsh CT, Sinskey AJ (1987) Biosynthetic thiolase from Zoogloea ramigera. Isolation and characterization of the structural gene. J Biol Chem 262:97–102
Peoples OP, Sinskey AJ (1989) Poly-β-hydroxybutyrate biosynthesis in Alcaligenes eutrophus H16. Characterization of the genes encoding β-ketothiolase and acetoacetyl-CoA reductase. J Biol Chem 264:15293–15297
Pramanik A, Pawar S, Antonian E, Schulz H (1979) Five different enzymatic activities are associated with the multienzyme complex of fatty acid oxidation from Escherichia coli. J Bacteriol 137:469–473
Rosenkrantz M, Alam T, Kim KS, Clark BJ, Srere PA, Guarente LP (1986) Mitochondrial and nonmitochondrial citrate synthases in Saccharomyces cerevisiae are encoded by distinct homologous genes. Mol Cell Biol 6:4509–4515
Saitou NM, Nei M (1987) The neighbor joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Sharp PM, Cowe E (1991) Synonymous codon usage in Saccharomyces cerevisiae. Yeast 7:657–678
Sourdis J, Nei M (1988) Relative efficiencies of the maximum parsimony and distance-matrix methods in obtaining the correct phylogenetic tree. Mol Biol Evol 5:298–311
Staack H, Binstock JF, Schulz H (1978) Purification and properties of a pig heart thiolase with broad chain length specificity and comparison of thiolases from the pig heart and Escherichia coli. J Biol Chem 253:1827–1831
Thompson S, Mayer F, Peoples OP, Masamune S, Sinskey AJ, Walsh CT (1989) Mechanistics studies on β-ketoacyl thiolase from Zoogloea ramigera: identification of the active-site nucleophil as Cys-89, its mutation to Ser-89, and kinetic and thermodynamic characterization of wild-type and mutant enzymes. Biochemistry 28:5735–5742
Thompson SL, Krisans SK (1990) Rat liver peroxisomes catalyze the initial step in cholesterol synthesis. The condensation of acetyl-CoA units into acetoacetyl-CoA. J Biol Chem 265:5731–5735
Thorsness PE, Fox TD (1990) Escape of DNA from mitochondria to the nucleus in Saccharomyces cerevisiae. Nature 346:376–379
Tomita K, Saito T, Fukui T (1983) In: Lennon DLF, Stratman WF, Zalten RN (eds) Biochemistry of metabolic processes. Elsevier, New York, p 353
Yang SY, Yang XYH, Healy-Louie G, Schulz H, Elzinga M (1990)Nucleotide sequence of the fadA gene. Primary structure of 3-ketoacyl-coenzyme A thiolase from Escherichia coli and the structural organization of the fadAB operon. J Biol Chem 265:10424–10429
Author information
Authors and Affiliations
Additional information
Offprint requests to: J.E. Pérez-Ortín
Rights and permissions
About this article
Cite this article
Igual, J.C., González-Bosch, C., Dopazo, J. et al. Phylogenetic analysis of the thiolase family. Implications for the evolutionary origin of peroxisomes. J Mol Evol 35, 147–155 (1992). https://doi.org/10.1007/BF00183226
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00183226