Abstract
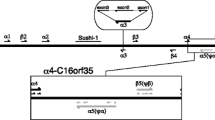
We report the cloning of a cDNA and two corresponding β-globin genes of the Atlantic salmon (Salmo salar L.) as well as two genes for α-globins. Nucleotide sequence analysis of the cDNA shows that the predicted β-globin peptide comprises 148 amino acids with a calculated molecular mass of 16,127 Da and an overall amino acid similarity of 40–50% to higher vertebrates and 60–90% to fish sequences. The study of the genomic organization of α- and β-globin genes shows that, as is the case in Xenopus, the salmon genes are adjacent. Two sets of linked α- and β-globin genes were isolated and restriction-enzyme polymorphisms indicate that they belong to two distinct loci, possibly as a result of the salmon tetraploidy. In each locus the α- and β-globin genes are oriented 3′ to 3′ relative to each other with the RNA coding sequences located on opposite DNA strands. This is the first evidence for this type of arrangement found for globin genes. Moreover, while the linkage found in salmon and Xenopus supports the hypothesis of an initial tandem duplication of a globin ancestor gene, our results raise the question of the actual original orientation of the duplicated genes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Barra D, Petruzzelli R, Bossa F, Brunari M (1983) Primary structure of hemoglobin from trout (Salmo irideus): amino acid sequence of the b chain of trout HbI. Biochem Biophys Acta 742:72–77
Brent R, Ausubel FM, Kingston RE, Moore DD, Smith JA, Seidman JG, Struhl K (1987) Current Protocols in Molecular Biology. In: Greene Publishing Associates and Wiley-Interscience (ed) Massachusetts General Hospital and Harvard Medical School
Brunari M (1975) Molecular adaptation to physiological requirements: The hemoglobin system of trout. Curr Topics Cell Regul 9:1–39
Choi OR, Engel JD (1986) A 3′ enhancer is required for temporal and tissue-specific transcriptional activation of the chicken adult β-globin gene. Nature 323:731–734
Colbert EH (1969) Evolution of the vertebrates. In: Wiley A (ed) Academic Press, New York, pp 1–24
Collins FS, Weissman SM (1984) The molecular genetics of human hemoglobin. Prog Nucleic Acid Res Mol Biol 31:317–421
Dodgson JB, Strommer J, Engel JD (1979) Isolation of the chicken β-globin gene and a linked embryonic β-like globin gene from a chicken DNA recombinant library. Cell 17:879–887
Dolan M, Dodgson JB, Engel JD (1983) Analysis of the chicken beta-globin gene: Nucleotide sequence of the locus, microheterogeneity at the 5′-end of the beta-globin mRNA, an aberrant nuclear species. J Mol Chem 283:3983–3990
Emerson BM, Nickol J, Jackson PD, Felsenfeld G (1987) Analysis of the tissue-specific enhancer at the 3′ end of the chicken adult β-globin gene. Proc Natl Acad Sci USA 84:4786–4790
Fisher WK, Nash AR, Thompson EOP (1977) Haemoglobins of the shark, Heterodontus portusjacksoni III. Amino Acid Sequence of the β-chain. Aust J Biol Sci 30:487–506
Fyhn UE, Withler R (1991) Ontogeny of hemoglobins in chinook salmon, Oncorhynchus tshawytscha. Comp Biochem Physiol 9813:201–208
Giles MA, Randall DJ (1980) Oxygenation characteristics of the polymorphic hemoglobins of coho salmon (Oncorhynchus kisutch) at different developmental stages. Comp Biochem Physiol 65A:265–271
Giles MA, Rystephanuk DM (1989) Ontogenetic variation in the multiple hemoglobins of Arctic Charr, Salvelinus alpinus. Can J Aquat Sci 46:804–809
Goodman M, Moore GW (1975) Darwinian evolution in the genealogy of haemoglobin. Nature 253:603–608
Grujic-Injac B, Braunitzer G, Stanyl A (1980) Hemoglobin beta-chain: carp. Hoppe-Seyler's Z Physiol Chem 361:1629–1639
Higgins DG, Sharp P (1988) CLUSTAL: A package for performing multiple sequence alignment on a microcomputer. Gene 73:237–244
Higgs DR, Vickers MA, Wilkie AOM, Pretorius IM, Jarman AP, Weatherall DJ (1989) A review of the molecular genetics of the human α-globin gene cluster. Blood 73:1081–1104
Hosbach HA, Wyler T, Weber R (1983) The Xenopus laevis globin gene family: chromosomal arrangement and gene structure: Cell 32:45–53
Iuchi I (1973) Chemical and physiological properties of the larval and the adult hemoglobins in the rainbow trout, Salmo Gairdnerii Irideus. Comp Biochem Physiol 44B:1087–1101
Jeffreys AJ, Wilson V, Wood D, Simons JP (1980) Linkage of adult α- and β-globin gene duplication by tetraploidization. Cell 21:555–564
Jones RW, Old JM, Trent RJ, Clegg JB, Weatherall DJ (1981) Major rearrangement in the human β-globin gene cluster. Nature 291:39
Lifton RP, Goldberg ML, Karp RW, Hogness DS (1977) The organization of the histone genes in Drosophila melanogaster: Functions and evolutionary implication. Cold Spring Harbor Symp Quant Biol 42:47–55
Karlsson S, Nienhuis AW (1985) Developmental regulation of human globin genes. An Rev Biochem 54:1071–1108
Knochel W, Korge E, Basner A, Meyerhof W (1986) Globin evolution in the genus Xenopus: Comparative analysis of cDNAs coding for adult globin polypeptides of Xenopus borealis and Xenopus tropicalis. J Mol Evol 23:211–223
Kollias G, Hurst J, deBoer E, Grosveld F (1987) The human β-globin gene contains a developmental specific enhancer. Nucleic Acids Res 15:5739–5747
Marotta CA, Wilson JT, Forget BG, Weissman SM (1977) Human beta-globin RNA: III. Nucleotide sequence derived from complementary DNA. J Biol Chem 252:5040–5053
Maxson R, Mohun T, Kedes L (1983) Distinct organizations and patterns of expression of early and late histone gene sets in the sea urchin. Nature 301:120–125
Miller SA, Dykes DD, Polesky HF (1988) A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res 16:1215
Nickol JM, Felsenfeld G (1988) Bidirectional control of the chicken β- and ε-globin genes by a shared enhancer. Proc Natl Acad Sci USA 85:2548–2552
Orkin SH (1990) Globin gene regulation and switching: circa 1990. Cell 63:665–672
Orkin SH, Jr Kazanian HH (1984) The mutation and polymorphisms of the human β-globin gene and its surrounding DNA. Ann Rev Genet 18:131–171
Patient RK, Elkington JA, Kay RM, Williams JG (1980) Internal organization of the major adult α- and β-globin genes of X. laevis. Cell 21:565–573
Patient RK, Banville D, Brewer AC, Elkington JA, Greaves DR, Lloyd MM, Williams JG (1982) The organization of the tadpole and adult β-globin genes of Xenopus laevis. Nucleic Acids Res 10:7935–7945
Perutz MF (1970) The bohr effect and combination with organic phosphates. Nature 228:734–739
Petruzzelli R, Barra D, Goffredo BM, Bossa F, Coletta M, Brunori M (1984) Amino-acid sequence of β-chain of hemoglobin IV from trout (Salmo irideus). Biochim Biophys Acta 789:69–73
Powers DA, Edmunson AB (1972) Multiple hemoglobins of catostomid fish. J Bio Chem 247:6686–6693
Purucker M, Bodine D, Lin H, McDonagh K, Nienhuis AW (1990) Structure and function of the enhancer 3′ to the human Aγ globin gene. Nucleic Acids Res 18:7407–7415
Rodosavljevic D, Crkvenjakov R (1989) Genomic sequence for rat beta-globin major gene. Nucleic Acids Res 17:4368
Rodewald K, Braunitzer G (1984) Hemoglobin beta-chain: Goldfish. Hoppe-Seyler's Z Physiol Chem 365:95–104
Rodewald K, Oberthuer W, Braunitzer G (1987) Homeothermic fish and hemoglobin primary structure from bluefin tuna (Thunnus thynnus). Hoppe-Seyler's Z Physiol Chem 368:795–805
Sheets MD, Ogg SC, Wickens MP (1990) Point mutations in AAUAAA and the poly(A) addition site: effects on the accuracy and efficiency of cleavage and polyadenylation in-vitro. Nucleic Acids Res 18:5799–5805
Stalder J, Wirthmuller U, Beck J, Gruber A, Meyerhof W, Knochel W, Weber R (1988) Primary structure and evolutionary relationship between the adult α-globin genes and their flanking regions of Xenopus laevis and Xenopus tropicalis. J Mol Evol 28:64–71
Westman K (1970) Hemoglobin polymorphism and its ontogeny in sea-running and landlocked Atlantic salmon (Salmo salar L.). Ann Acad Sci Fenn [A] IV Biologica 170:1–24
Wickens M (1990) How the messenger got its tail: addition of poly(A) in the nucleus. TIBS 15:277–281
Wilkins NP, Iles TD (1966) Haemoglobin polymorphism and it ontogeny in herring (Clupea harengus) and sprat (Sprattus sprattus). Comp Biochem Physiol 17:1141–1158
Wilkins NP (1985) Ontogeny and evolution of salmonid hemoglobins. Int Rev Cytol 94:269–298
Williams JG, Key RM, Patient RK (1980) The nucleotide sequence of the major beta-globin mRNA from Xenopus laevis. Nucleic Acids Res 8:4247–4258
Wolff J, Gannon F (1988) cDNA and deduced amino acid sequence of the Salmo salar (Atlantic salmon) adult hemoglobin alpha chain. Nucleic Acids Res 16:1215
Author information
Authors and Affiliations
Additional information
Correspondence to: F. Gannon
Rights and permissions
About this article
Cite this article
Wagner, A., Deryckere, F., McMorrow, T. et al. Tail-to-tail orientation of the Atlantic salmon alpha- and beta-globin genes. J Mol Evol 38, 28–35 (1994). https://doi.org/10.1007/BF00175492
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00175492