Abstract
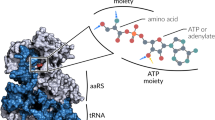
An operational RNA code relates specific amino acids to sequences/structures in RNA hairpin helices which reconstruct the seven-base-pair acceptor stems of transfer RNAs. These RNA oligonucleotides are aminoacylated by aminoacyl tRNA synthetases. The specificity and efficiency of aminoacylation are generally determined by three or four nucleotides which are near the site of amino acid attachment. These specificity-determining nucleotides include the so-called “discriminator base” and one or two base pairs within the first four base pairs of the helix. With three examples considered here, nucleotide sequence variations between the eubacterial E. coli tRNA acceptor stems and their human cytoplasmic and mitochondrial counterparts are shown to include changes of some of the nucleotides known to be essential for aminoacylation by the cognate E. coli enzymes. If the general locations of the specificity-determining nucleotides are the same in E. coli and human RNAs, these RNA sequence variations imply a similar covariation in sequences/structures of the E. coli and human tRNA synthetases. These covariations would reflect the integral relationship between the operational RNA code and the design and evolution of tRNA synthetases.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Biou V, Yaremchuk A, Tukalo M, Cusack S (1994) The 2.9 A crystal structure of T. thermophilus seryl-tRNA synthetase complexed with tRNASer. Science 263:1404–1410
Brunie S, Zelwer C, Risler J (1990) Crystallographic study at 2.5 Å resolution of the interaction of methionyl-tRNA synthetase from Escherichia coli with ATP. J Mol Biol 216:411–424
Buechter DD,Schimmel P (1993) Aminoacylation of RNA Minihelices: implications for tRNA synthetase structural design and evolution. CRC Crit Rev Biochem 28:309–322
Burbaum JJ, Schimmel P (1991) Structural relationships and the classification of aminoacyl-tRNA synthetases. J Biol Chem 266: 16965–16968
Cavarelli J, Rees B, Ruff M, Thierry J-C, Moras D (1993) Yeast tRNAAsp recognition by its cognate class II aminoacyl-tRNA synthetase. Nature 362:181–184
Cooley L, Appel B, Söll D (1982) Post-transcriptional nucleotide addition is responsible for the formation of the 5′-terminus of histidine tRNA. Proc Natl Acad Sci USA 79:6475–6479
Cusack S, Berthet-Colominas C, Hartlein M, Nassar N, Leberman R (1990) A second class of synthetase structure revealed by x-ray analysis of Escherichia coli seryl-tRNA synthetase at 2.5 A. Nature 347:249–255
Cusack S, Hartlein M, Leberman R (1991) Sequence, structure and evolutionary relationships between class 2 aminoacyl-tRNA synthetases. Nucleic Acids Res 19:3489–3498
de Duve C (1988) The second genetic code. Nature 333:117–118
Eriani G, Delarue M, Poch O, Gangloff J, Moras D (1990) Partition of tRNA synthetases into two classes based on mutually exclusive sets of sequence motifs. Nature 347:203–206
Francklyn C, Musier-Forsyth K, Schimmel P (1992) Small RNA helices as substrates for aminoacylation and their relationship to the charging of transfer RNAs. Eur J Biochem 206:315–321
Francklyn C, Schimmel P (1989) Aminoacylation of RNA minihelices with alanine. Nature 337:478–481
Francklyn C, Schimmel P (1990) Enzymatic aminoacylation of an eight-base-pair microhelix with histidine. Proc Natl Acad Sci USA 87:8655–8659
Francklyn C, Shi J-P, Schimmel P (1992) Overlapping nucleotide determinants for specific aminoacylation of RNA microhelicies. Science 255:1121–1125
Frugier M, Florentz C, Giege R (1994) Efficient aminoacylation of resected RNA helices by class II aspartyl-tRNA synthetase dependent on a single nucleotide. EMBO J 13:2218–2226
Frugier M, Florentz C, Giege R (1992) Anticodon-independent aminoacylation of an RNA minihelix with valine. Proc Natl Acad Sci USA 89:3990–3994
Hou Y-M, Schimmel P (1989a) Evidence that a major determinant for the identity of a transfer RNA is conserved in evolution. Biochemistry 28:6800–6804
Hou Y-M, Schimmel P (1989b) Modeling with in vitro kinetic parameters for the elaboration of transfer RNA identity in vivo. Biochemistry 28:4942–4947
Hou Y-M, Schimmel P (1988) A simple structural feature is a major determinant of the identity of a transfer RNA. Nature 333:140–145
Hountondji C, Dessen P, Blanquet S (1986) Sequence similarities among the family of aminoacyl-tRNA synthetases. Biochimie 68: 1071
Jukes TH, Osawa S, Watanabe K, Muto A (1992) Recent evidence for evolution of the genetic code. Microbiol Rev 56:229–264
Ludmerer SW, Schimmel P (1987) Gene for yeast glutamine tRNA synthetase encodes a large amino-terminal extension and provides a strong confirmation of the signature sequence for a group of the aminoacyl-tRNA synthetase. J Biol Chem 262:10801–10806
Martinis SA, Schimmel P (1992) Enzymatic aminoacylation of sequence-specific RNA minihelices and hybrid duplexes with methionine. Proc Natl Acad Sci USA 89:65–69
Martinis SA, Schimmel P (1993) Microhelix aminoacylation by a class I tRNA synthetase. J Biol Chem 268:6069–6072
McClain WH, Foss K (1988) Changing the identity of a tRNA by introducing a G-U wobble pair near the 3′ acceptor end. Science 240: 793–796
Moller W, Janssen GMC (1992) Statistical evidence for remnants of the primordial code in the acceptor stem of prokaryotic transfer RNA. J Mol Evol 34:471–477
Moras D (1992) Structural and functional relationships between aminoacyl-tRNA synthetases. Trends Biochem Sci 17:159–164
Musier-Forsyth K, Schimmel P (1992) Functional contacts of a transfer RNA synthetase with 2′-hydroxyl groups in the RNA minor groove. Nature 357:513–515
Musier-Forsyth K, Usman N, Scaringe S, Doudna J, Green R, Schimmel P (1991) Specificity for aminoacylation of an RNA helix: an unpaired, exocylic amino group in the minor groove. Science 253: 784–786
Nureki O, Niimi T, Muto Y, Kanno H, Kohno T, Muramatsu T, Kawai G, Miyazawa T, Giege R, Florentz C, Yokoyama S (1993) Conformational change of tRNA upon interaction of the identity-determinant set with aminoacyl-tRNA synthetase. In: Nierhaus KH, Franceschi F, Subramanian AR, Erdmann VA, Wittmann-Liebold B (eds) The translation apparatus. Plenum Press, New York, pp 59–66
Park SJ, Schimmel P (1988) Evidence for interaction of an aminoacyl transfer RNA synthetase with a region important for the identity of its cognate transfer RNA. J Biol Chem 263:16527–16530
Perona JJ, Rould MA, Steitz TA, Risler J-L, Zelwer C, Brunie S (1991) Structural similarities in glutaminyl- and methionyl-tRNA synthetases suggest a common overall orientation of tRNA binding. Proc Natl Acad Sci USA 88:2903–2907
Rould MA, Perona JJ, Söll D, Steitz TA (1991) Structural basis of anticodon loop recognition by glutaminyl-tRNA synthetase. Nature 352:213–218
Rould MA, Perona JJ, Söll D, Steitz TA (1989) Structure of E. coli Glutaminyl-tRNA synthetase complexed with tRNAGln and ATP at 2.8 Å resolution. Science 246:1135–1142
Ruff M, Krishnaswamy S, Boeglin M, Poterszman A, Mitschler A, Podjarny A, Rees B, Thierry JC, Moras D (1991) Class II aminoacyl transfer RNA synthetases: crystal structure of yeast aspartyltRNA synthetase complexed with tRNAAsp. Science 252:1682–1689
Sampson JR, Saks ME (1993) Contributions of discrete tRNASer domains to aminoacylation by E. coli seryl-tRNA synthetase: a kinetic analysis using model RNA substrates. Nucleic Acids Res 21:4467–4475
Schimmel P, Henderson B (1994) Possible role of aminoacyl RNA complexes in non-coded peptide synthesis and origin of coded synthesis. Proc Natl Acad Sci USA 91:11283–11286
Schimmel P, Giegé R, Moras D, Yokoyama S (1993)An operational RNA code for amino acids and potential relationship to genetic code. Proc Natl Acad Sci USA 90:8763–8768
Shi J-P, Francklyn C, Hill K, Schimmel P (1990) A nucleotide that enhances the charging of RNA minihelix sequence variants with alanine. Biochemistry 29:3621–3626
Shi J-P, Martinis SA, Schimmel P (1992) RNA tetraloops as minimalist substrates for aminoacylation. Biochemistry 31:4931–4936
Sprinzl M, Hartmann T, Weber J, Blank J, Zeidles R (1989) Compilation of tRNA sequences and sequences of tRNA genes. Nucleic Acids Res 17:r1-r172
Starzyk RM, Webster TA, Schimmel P (1987) Evidence for dispensable sequences inserted into a nucleotide fold. Science 237:1614–1618
Steinberg S, Misch A, Sprinzl M (1993) Compilation of tRNA sequences and sequences for tRNA genes. Nucleic Acids Res 21: 3011–3015
Trézéguet V, Edwards H, Schimmel P (1991) A single base pair dominates over the novel identity of an Escherichia coli tyrosine tRNA in Saccharomyces cerevisiae. Mol Cell Biol 11:2744–2751
Webster TA, Tsai H, Kula M, Mackie GA, Schimmel P (1984) Specific sequence homology and three-dimensional structure of an aminoacyl transfer RNA synthetase. Science 226:1315–1317
Author information
Authors and Affiliations
Additional information
Based on part of a presentation made at a workshop- “Aminoacyl-tRNA Synthetases and the Evolution of the Genetic Code”-held at Berkeley, CA, July 17–20, 1994
Rights and permissions
About this article
Cite this article
Schimmel, P. An operational RNA code for amino acids and variations in critical nucleotide sequences in evolution. J Mol Evol 40, 531–536 (1995). https://doi.org/10.1007/BF00166621
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00166621