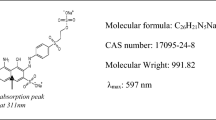
Summary
Colour removal from phenplic industrial effluents by phenol oxidase enzymes and white-rot fungi was compared. Soluble laccase and horseradish peroxidase (HRP) removed colour from pulp mill (E), cotton mill hydroxide (OH) and cotton mill sulphide (S) effluents, but rapid and irreversible enzyme inactivation took place. Entrapment of laccase in alginate beads improved decolorization by factors of 3.5 (OH) and 2 (E); entrapment of HRP improved decolorization by 36 (OH), 20 (E) and 9 (S). Beads were unsuitable for continuous use because the enzymes were rapidly released into solution. Co-polymerization of laccase or HRP with L-tyrosine gave insoluble polymers with enzyme activity. Entrapment of the co-polymers in gel beads further increased the efficiency of decolorization of E by 28 (laccase) and by 132 (HRP) compared with soluble enzymes. Maximum decolorization of all three effluents by batch cultures of Coriolus versicolor (70%–80% in 8 days) was greater than the maximum enzymic decolorization (48% of OH in 3 days by entrapped laccase). Soluble laccase (222 units ml−1) precipitated 1.2 g l−1 phenol from artificial coal conversion effluent at pH 6.0 and the rate of precipitation and enzyme inactivation was faster at pH 6.0 than at pH 8.5.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Alberti BN, Klibanov AM (1981) Enzymatic removal of dissolved aromatics from industrial aqueous effluents. Biotechnol Bioeng Symp 2:373–379
Arora DS, Sandhu DK (1985) Laccase production and wood degradation by a white-rot fungus Daedalea flavida. Enzyme Microb Technol 7:405–408
Atlow SC, Bonadonna-Aparo L, Klibanov AM (1984) Dephenolization of industrial wastewaters catalyzed by polyphenol oxidase. Biotechnol Bioeng 26:599–603
Bashan Y (1986) Alginate beads as synthetic inoculant carriers for slow release of bacteria that affect plant growth. Appl Environ Microbiol 51:1089–1098
Boman B, Ek M, Eriksson K-E, Frostell B (1988) Some aspects on biological treatment of bleached pulp effluents. Nordic Pulp Paper Res J 3:13–18
Eaton DC, Chang H-M, Joyce TW, Jeffries TW, Kirk TK (1982) Method obtains fungal reduction of the colour of extraction-stage kraft bleach effluents. TAPPI 65:89–92
Emerson E (1943) The condensation of aminoantipyrene. II A new colour test for phenolic compounds. J Org Chem 8:417–428
Eriksson K-E, Kirk TK (1986) Biopulping, biobleaching and treatment of kraft bleaching effluents with white-rot fungi. In: Robinson CW (ed) Comprehensive biotechnology, vol 3. Pergamon Press, New York, pp 271–294
Fahreus G, Reinhammar B (1967) Large scale production and purification of laccase from cultures of the fungus Polyporus versicolor and some properties of the laccase A. Acta Chem Scand 21:2367–2378
Forss K, Jokinen K, Savolainen M, Williamson H (1987) Utilization of enzymes for effluent treatment in the pulp and paper industry. In: Proc. 4th International Symposium Wood and Pulping Chemistry, Vol I, Paris, France, 1987, pp 179–183
Klibanov AM, Morris ED (1981) Horseradish peroxidase for the removal of carcinogenic aromatic amines from water. Enzyme Microb Technol 3:119–122
Klibanov AM, Tu T-M, Scott KP (1983) Peroxidase-catalyzed removal of phenols from coal conversion wastewaters. Science 221:259–261
Kringstad KP, Lindstrom K (1984) Spent liquors from pulp bleaching. Environ Sci Technol 18:236A–248A
Mosbach R (1963) Purification and some properties of laccase from Polyporus versicolor. Biochim Biophys Acta 73:204–212
Paice MG, Jurasek L (1984) Peroxidase catalyzed colour removal from bleach plant effluent. Biotechnol Bioeng 26:477–480
Peyton TO (1984) Biological disposal of hazardous waste. Enzyme Microb Technol 6:146–154
Royer G, Livernoche D, Desrochers M, Jurasek L, Rouleau D, Mayer RC (1983) Decolorization of kraft mill effluent: kinetics of a continuous process using immobilized Coriolus versicolor. Biotechnol Lett 5:121–326
Royer G, Desrochers M, Jurasek L, Rouleau D, Mayer RC (1985) Batch and continuous decolorization of bleached kraft effluents by a white-rot fungus. J Chem Technol Biotechnol 35B:14–22
Sarkar JM, Burns RG (1984) Synthesis and properties of β-D-glucosidase-phenolic copolymers as analogues of soil humic-enzyme complexes. Soil Biol Biochem 16:619–625
Shuttleworth KL, Bollag J-M (1986) Soluble and immobilized laccase as catalysts for the transformation of substituted phenols. Enzyme Microb Technol 8:171–177
Singer PC, Pfaender FK, Chinchilli J, Lamb JC (1978) Composition and biodegradability of organics in coal conversion wastewaters. In: Ayer F, Massoglia MF (eds) Environmental aspects of fuel conversion technology III, (Publication EPA-600/7–78–063), Environmental Protection Agency, Washington, DC, pp 461–486
Sjoblad RD, Bollag J-M (1977) Oxidation coupling of aromatic pesticide intermediates by a fungal phenoloxidase. Appl Environ Microbiol 33:906–910
Sjoblad RD, Bollag J-M (1981) Oxidative coupling of aromatic compounds by enzymes from soil microorganisms. In: Paul EA, Ladd JN (eds) Soil biochemistry, vol 5. Dekker, New York, pp 113–152
Sundman G, Kirk TK, Chang H-M (1981) Fungal decolorization of kraft bleach plant effluent. Fate of the chromophoric material. TAPPI 64:145–148
Author information
Authors and Affiliations
Additional information
Offprint requests to: R. G. Burns
Rights and permissions
About this article
Cite this article
Davis, S., Burns, R.G. Decolorization of phenolic effluents by soluble and immobilized phenol oxidases. Appl Microbiol Biotechnol 32, 721–726 (1990). https://doi.org/10.1007/BF00164748
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00164748