Summary
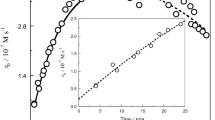
The kinetics of oxidation ofl-ascorbic acid (H2A) by the mononuclear complex [Co(NH3)5(OH2)] (ClO3)3 have been studied spectrophotometrically at 490 nm. The mechanism involves single one-electron transfers involving the pentaamminehydroxo complex and the ascorbate anions (HA− and A2−), subsequent formation of ascorbate radicals and > 90% CoII. The appropriate rate law in the 7.00 ⩽ pH ⩽ 8.40 and 0.005 ⩽ [A T] ⩽ 0.05 mol dm−3 ranges, has been established as:
The rate is slower at lower pH as the less important reactionk 1 (ROH sup3inf2 + HA− → products) becomes dominant, where R is (NH3)5CoIII. The more significant rate constantk 4 (ROH2+ + A2− → products) was calculated at 25.7 °C as (2.7 + 0.5) × 102 dm−3mol−1 s−1, with ΔH # = 105 ± 11 kJ mol−1 and ΔS # = 93 +- 45 J mol−1 K −1.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
M. Kimura, M. Yamamoto and S. Yamabe,J. Chem. Soc., Dalton Trans., 423 (1982).
K. Tsukahara and Y. Yamamoto,Bull. Chem. Soc. Jpn,54 2642 (1981).
P. Martinez, J. Zuluaga, J. Kraft and R. van Eldik,Inorg. Chim. Acta,146, 9 (1988).
K. Tsukahara, T. Izumitani and Y. Yamamoto,Bull. Chem. Soc. Jpn,55, 130 (1982).
N. H. Williams and J. K. Yandell,Aust. J. Chem.,35, 1133 (1982).
Y. Abe, S. Okada, H. Horii and S. Taniguchi,J. Chem. Soc., Perkins Trans., 715 (1987).
D. H. Macartney and N. Sutin,Inorg. Chim. Acta,74, 221 (1983).
E. Pelizetti and E. Mentasti,J. Chem. Soc., Dalton Trans., 61 (1978).
M. M. Taqui Khan and R. S. Shukla,Inorg. Chim. Acta,149, 89 (1989).
D. A. Dixon, N. P. Sadler and T. P. Dasgupta,J. Chem. Soc., Dalton Trans. (in press).
D. A. Dixon, N. P. Sadler and T. P. Dasgupta,J. Chem. Soc., Dalton Trans., 3490 (1993).
R. A. Kirby,PhD Thesis, University of the West Indies, (1990).
A. G. Sykes,Trans. Faraday Soc., 1329 (1963).
S. K. Ghosh, R. N. Bose and E. S. Gould,Inorg. Chem.,26, 3722 (1987).
F. Basolo and R. F. Murman,Inorg. Synth.,4, 171 (1953).
T. P. Dasgupta and G. M. Harris,J. Am. Chem. Soc.,90, 6360 (1967).
C. H. Langford and W. R. Muir,J. Am. Chem. Soc.,89, 3141 (1967).
J. F. Endicott, G. Hugh, M. Hoffmann and A. House,J. Chem. Educ.,46, 440 (1969).
M. Dawson, W. H. Elliott and K. M. Jones,Data for Biochemical Research, 2nd Edit., OUP, UK, p. 479.
B. H. J. Bielski in P. A. Seib and B. M. Tolbert (Eds.),Ascorbic Acid: Chemistry, Metabolism and Uses, American Chemistry Society, Washington DC, 1980, p. 82.
D. E. Cabelli and B. H. J. Bielski,J. Phys. Chem.,87, 1809 (1983).
K. Abdur-Rashid and T. P. Dasgupta, Unpublished work.
N. P. Sadler,PhD Thesis, University of the West Indies, (1986).
K. P. Stolyarov and I. A. Amantova,Talanta,14, 1237 (1967).
R. A. Sykes,J. Phys. Chem.,72, 891 (1968).
P. Martinez, J. Zuluaga, P. Noheda and R. van Eldik,Inorg. Chim. Acta,195, 249 (1992).
B. Douglas, D. H. McDaniel and J. J. Alexander,Concepts and Models of Inorganic Chemistry, 2nd Edit., Wiley, NY, 1983, p. 339.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dixon, D.A., Sadler, N.P. & Dasgupta, T.P. Mechanism of the oxidation ofL-ascorbic acid by the pentaammineaquacobalt(III) ion in aqueous solution. Transition Met Chem 20, 295–299 (1995). https://doi.org/10.1007/BF00143496
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00143496