Abstract
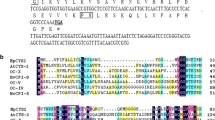
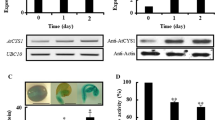
The pea (Pisum sativum) gene Cyp15a encodes a protein with sequence similarity to cysteine proteases. Expression of Cyp15a was investigated during pea seedling development and in response to environmental stress. Cyp15a shows increased transcription and elevated mRNA levels in plant tissues that are partially dehydrated or treated with 0.6 M mannitol. Cyp15a mRNA levels also increase in seedlings treated with 0.2–0.25 M NaCl or KCl. During development, Cyp15a mRNA levels increase within 6 to 12 h in cotyledons and axes during germination and continue to increase for at least 96 h. Illumination of dark-grown seedlings increased Cyp15a mRNA abundance in elongating and non-elongating stem tissues. GA and ABA, which modulate the abundance of many seed-localized cysteine proteases, did not significantly modulate Cyp15a mRNA levels in stems. The protein encoded by Cyp15a contains a typical amino-terminal secretory targeting domain. This domain is followed by a pro-sequence containing ca. 110 amino acids that is found in other cysteine proteases. Polyclonal antibodies, directed against CYP15a, recognized both the larger pro-form and the cleaved mature form of CYP15a on western blots. Immunolocalization assays indicated that both forms of the protein are located in cell walls of stem cortical cells.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Baker EN: Structure of actinidin, after refinement at 1.7 Å resolution. J Mol Biol 141: 441–484 (1980).
Bray EA: Molecular responses to water deficit. Plant Physiol 103: 1035–1040 (1993).
Chamberlain JP: Fluorographic detection of radioactivity in polyacrylamide gels with the water-soluble fluor, sodium salicylate. Anal Biochem 98: 132–135 (1979).
Chrispeels MJ: Sorting of proteins in the secretory system. Annu Rev Plant Physiol Plant Mol Biol 42: 21–53 (1991).
Creelman RA, Mason HS, Bensen RJ, Boyer JS, Mullet JE: Water deficit and abscisic acid cause differential inhibition of shoot versus root growth in soybean seedlings. Plant Physiol 92: 205–214 (1990).
Creelman RA, Zeevaart JAD: Abscisic acid accumulation in spinach leaf slices in the presence of penetrating and non penetrating solutes. Plant Physiol 77: 25–28 (1985).
Deitrich RA, Maslyar DJ, Heupel RC, Harada JJ: Spatial patterns of gene expression in Brassica napus seedlings: identification of a cortex-specific gene and localization of mRNAs encoding isocitrate lyase and a polypeptide homologous to proteinases. Plant Cell 1: 73–80 (1989).
Goretzki L, Schmitt M, Mann K, Calvete J, Churcholowski N, Kramer M, Günzel WA, Jänicke G, Graeff H: Effective activation of the proenzyme form of the urokinasetype plasminogen activator (pro-uPA) by the cysteine protease cathepsin. FEBS Lett 297: 112–118 (1992).
Granell A, Harris N, Pisabarro AG, Carbonell J: Temporal and spatial expression of a thiol protease gene during pea ovary senescence, and its regulation by gibberellin. PLant J 2: 907–915 (1992).
Guerrero FD, Jones JT, Mullet JE: Turgor-responsive gene transcription and RNA levels increase rapidly when pea shoots are wilted. Sequence and expression of three inducible genes. Plant Mol Biol 15: 11–26 (1990).
Hayat MA: Principles and Techniques of Electron Microscopy, 3rd ed. CRC Press, Boca Raton, FL (1989).
Holwerda BC, Galvin NJ, Baranski TJ, Rogers JC: In-vitro processing of aleurain, a barley vacuolar thiol protease. Plant Cell 2: 1091–1106 (1990).
Holwerda BC, Padgett HS, Rogers JC: Proaleurain vacuolar targeting is mediated by short contiguous peptide interactions. Plant Cell 4: 307–318 (1992).
Holwerda BC, Rogers JC: Purification and characterization of aleurain. A plant thiol protease functionally homologous to mammalian cathepsin H. Plant Physiol 99: 848–855 (1992).
Kalinski A, Melroy DL, Dwivedi RS, Herman EM: A soybean vacuolar protein (P34) related to thiol proteases is synthesized as a glycoprotein precursor during seed maturation. J Biol Chem 267: 12068–12076 (1992).
Kamphuis IG, Drenth J, Baker EN: Thiol proteases. Comparative studies based on the high-resolution structures of papain and actinidin, and on amino acid sequence information for cathepsins B and H, and stem bromelain. J Mol Biol 182: 317–329 (1985).
Koehler SM, Ho T-HD: Hormonal regulation, processing, and secretion of cysteine proteinases in barley aleurone layers. Plant Cell 2: 769–783 (1990).
Koizumi M, Yamaguchi-Shinozaki K, Tsuji H, Shinozaki K: Structure and expression of two genes that encode distinct drought-inducible cysteine proteinases in Arabidopsis thaliana. Gene 129: 175–182 (1993).
Kornfeld R, Kornfeld S: Assembly of asparagine-linked oligosaccharides. Annu Rev Biochem 54: 631–664 (1985).
Linthorst HJ.M, van derDoes C, Brederode FT, Bol JF: Circadian expression and induction by wounding of tobacco genes for cysteine proteinase. Plant Mol Biol 21: 685–694 (1993).
Mason RW, Gal S, Gottesman MM: The identification of the major excreted protein (MEP) from a transformed mouse fibroblast cell line as a catalytically active precursor form of cathepsin L. Biochem J 248: 449–454 (1987).
Meyer RF, Boyer JS: Osmoregulation, solute distribution, and growth in soybean seedlings having low water potentials. Planta 151: 482–489 (1981).
Pierce M, Raschke K: Synthesis and metabolism of abscisic acid in detached leaves of Phaseolus vulgaris L. after loss and recovery of turgor. Planta 153: 156–165 (1981).
Raschke K: Stomatal action. Annu Rev Plant Physiol 26: 309–340 (1975).
Reddy GK, Dhar SC: Purification and characterization of collagenolytic property of renal cathepsin L for arthritic rat. Int J Biochem 24: 1465–1473 (1992).
Rogers JC, Dean D, Heck GR: Aleurain: A barley thiol protease closely related to mammalian cathepsin H. Proc Natl Acad Sci USA 82: 6512–6516 (1985).
Sambrook J, Fritsch EF, Maniatis T: Molecular Cloning: A Laboratory Manual. Cold Spring Harbor Press, Cold Spring Harbor, NY (1989).
Schaffer MA, Fischer RL: Analysis of mRNAs that accumulate in response to low temperature identifies a thiol protease gene in tomato. Plant Physiol 87: 431–436 (1988).
Skriver K, Mundy J: Gene expression in response to abscisic acid and osmotic stress. Plant Cell 2: 503–512 (1990).
Smith SM, Gottesman MM: Activity and deletion analysis of recombinant human cathepsin L expressed in Escherichia coli. J Biol Chem 264: 20487–20495 (1989).
VanVolkenburgh E, Boyer JS: Inhibitory effects of water deficit on maize leaf elongation. Plant Physiol 77: 190–194 (1985).
Watanabe H, Abe K, Emori Y, Hosoyama H, Arai S: Molecular cloning and gibberellin-induced expression of multiple cysteine proteinases of rice seeds (Oryzains). J Biol Chem 266: 16897–16902 (1991).
Wiederanders B, Brömme D, Kirschke H, Figura Kv, Schmidt B, Peters C: Phylogenetic conservation of cysteine proteinases. Cloning and expression of a cDNA coding for human cathepsin S. J Biol Chem 267: 13708–13713 (1992).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jones, J.T., Mullet, J.E. A salt- and dehydration-inducible pea gene, Cyp15a, encodes a cell-wall protein with sequence similarity to cysteine proteases. Plant Mol Biol 28, 1055–1065 (1995). https://doi.org/10.1007/BF00032666
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00032666