Abstract
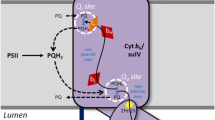
Recent work on the domain organization of the thylakoid is reviewed and a model for the thylakoid of higher plants is presented. According to this model the thylakoid membrane is divided into three main domains: the stroma lamellae, the grana margins and the grana core (partitions). These have different biochemical compositions and have specialized functions. Linear electron transport occurs in the grana while cyclic electron transport is restricted to the stroma lamellae. This model is based on the following results and considerations. (1) There is no good candidate for a long-range mobile redox carrier between PS II in the grana and PS I in the stroma lamellae. The lateral diffusion of plastoquinone and plastocyanin is severely restricted by macromolecular crowding in the membrane and the lumen respectively. (2) There is an excess of 14±18% chlorophyll associated with PS I over that of PS II. This excess is assumed to be localized in the stroma lamellae where PS I drives cyclic electron transport. (3) For several plant species, the stroma lamellae account for 20±3% of the thylakoid membrane and the grana (including the appressed regions, margins and end membranes) for the remaining 80%. The amount of stroma lamellae (20%) corresponds to the excess (14–18%) of chlorophyll associated with PS I. (4) The model predicts a quantum requirement of about 10 quanta per oxygen molecule evolved, which is in good agreement with experimentally observed values. (5) There are at least two pools of each of the following components: PS I, PS II, cytochrome bf complex, plastocyanin, ATP synthase and plastoquinone. One pool is in the grana and the other in the stroma compartments. So far, it has been demonstrated that the PS I, PS II and cytochrome bf complexes each differ in their respective pools.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- PS I and PS II:
-
Photosystem I and II
- P 700:
-
reaction center of PS I
- LHC II:
-
light-harvesting complex II
References
Albertsson P-Å (1982) Interaction between the lumenal sides of the thylakoid membrane. FEBS Lett 149(2): 186–190
Albertsson P-Å and Yu S-G (1988) Heterogeneity among Photosystem II. Isolation of thylakoid membrane vesicles with different functional antennae size of photosystem II. Biochim Biophys Acta 936: 215–221
Albertsson P-Å, Andreasson E and Svensson P (1990a) The domain organization of the plant thylakoid membrane. FEBS Lett 273(1,2): 36–40
Albertsson P-Å, Andreasson E, Persson A and Svensson P (1990b) Organization of the thylakoid membrane with respect to the four photosystems, PS Iα, PS Iβ, PS IIα and PS IIβ. In: Baltscheffsky M (ed) Current Research in Photosynthesis, Vol II, pp 923–926. Kluwer Academic Publishers, Dordrecht
Albertsson P-Å, Yu S-G and Larsson UK (1990c) Heterogeneity in Photosystem II. Evidence from fluorescence and gel electrophoresis experiments. Biochim Biophys Acta 1016: 137–140
Albertsson P-Å, Andreasson E, Svensson P and Yu S-G (1991) Localization of cytochrome f in the thylakoid membrane: Evidence for multiple domains. Biochim Biophys Acta 1098: 90–94
Albertsson P-Å (1994) Domain structure of biological membranes obtained by fragmentation and separation analysis. Meth Enzymology 228: 503–511
Albertsson P-Å, Andreasson E, Stefansson H and Wollenberger L (1994) Fractionation of thylakoid membrane. Methods Enzymology 228: 469–482
Andersson B and Anderson JM (1980) Lateral heterogeneity in the distribution of chlorophyll-protein complexes of the thylakoid membranes of spinach chloroplasts. Biochim Biophys Acta 593: 427–440
Anderson JM and Melis A (1983) Proc Natl Acad Sci USA 80: 745–749
Andreasson E, Svensson P, Weibull C and Albertsson P-Å (1988) Separation and characterization of stroma and grana membranes — evidence for heterogeneity in antenna size of both Photosystem I and Photosystem II. Biochim Biophys Acta 936: 339–350
Andreasson E and Albertsson P-Å (1993) Heterogeneity in Photosystem I — the larger antenna of Photosystem I is due to functional connection to a special pool of LHC II. Biochim Biophys Acta 1141: 175–182
Arntzen C (1983) In: Zubay G. Biochemistry. Addison Wesley Publ Co Reading, Mass, London, Amsterdam
Beattie JK, Fenson DJ, Freeman HC, Woodcock E, Hill HAO and Stokes AM (1975) An NMR investigation of electron transfer in the copper-protein, plastocyanin. Biochim Biophys Acta 405: 109–114
Björkman O and Demmig B (1987) Photon yield of O2 evolution and chlorophyll fluorescence characteristics at 77 K among vascular plants of diverse origins. Planta 170: 489–504
Blackwell M, Gibas C, Gyqax S, Roman D and Wagner B (1994) The plastoquinone diffusion coefficient in chloroplasts and its mechanistic implications. Biochim Biophys Acta 1183: 533–543
Evans JR (1986) A quantitative analysis of light distribution between the two photosystems, considering variation in both the relative amounts of the chlorophyll-protein complexes and the spectral quality of light. Photobiochem and Photobiophys 10: 135–147
Evans JR (1987) The dependence of quantum yield on wavelength and growth irradiance. Aust J Plant Physiol 14: 69–79
Flores S, Graan T and Ort DR (1983) Photobiochem Photobiophys 6: 293–304
Fork DC, and Herbert SK (1993) Electron transport and photophosphorylation by Photosystem I in vivo in plants and cyanobacteria. Photosynth Res 36: 149–168
Greenwood AD (1993) In: Lawlor DW, Photosynthesis: Molecular, Physiological and Environmental Processes, 2nd ed. Longman Group, UK
Haehnel W (1984) On the lateral electron transport between the two light reactions in spinach chloroplasts. In: Sybesma C (ed) Advances in Photosynthesis Research, Vol 1, pp 545–548. Martinus Nijhoff, Dordrecht
Hanchey P (1985) In: Salisbury FB and Ross CW. Plant Physiology, 3rd ed. Wadsworth Publ Co. Belmont, CA
Henrysson T and Sundby C (1990) Characterization of Photosystem II in stroma thylakoid membranes. Photosynth Res 25: 107–117
Joliot P, Lavergne J and Beal D (1992) Plastoquinone compartmentation in chloroplasts. I. Evidence for domains with different rates of photo-reduction. Biochim Biophys Acta 1101: 1–12
Juhler RK, Andreasson E, Yu S-G and Albertsson P-Å (1993) Composition of photosynthetic pigments in thylakoid membrane fractions from spinach. Photosynth Res 35: 171–178
Juniper BE (1978) In: Kirk JTO and Tilney-Bassett RAE. The Plastids. Elsevier/North Holland Biomedical Press, Amsterdam/New York/Oxford
Laetsch WM (1972) In: Conn EE and Stumpf PK. Outlines of Biochemistry, 3rd ed. Wiley, New York
Ledbetter MC and Porter KR (1970) Introduction to the Fine Structure of Plant Cells. Springer, Berlin/Heidelberg/New York
Melis A, Spangfort M And Andersson B (1987) Light-absorption and electron-transport balance between Photosystem II and photosystem I in spinach chloroplasts. Photochem Photobiol 45: 129–136
Menke (1980) In: Strasburger's Textbook of Botany. Longman, London/New York
Miller K (1988) In: Stryer, L. Biochemistry, 3rd ed. Freeman, New York
Murakami S and Packer L (1970) Protonation and chloroplast membrane structure. J Cell Biol 47: 332–351
Ort DR (1986) Energy transduction in oxygenic photosynthesis: An overview of structure and mechanism. In: Staehelin LA and Arnzen CJ (eds) Encyclopedia of Plant Physiology, New Series, Vol 19, pp 143–196. Springer-Verlag, Berlin/Heidelberg/New York/Tokyo
Park RB (1965) In: Bonner J and Varner JE (eds) Plant Biochemistry. Academic Press, New York
Romanowska E and Albertsson P-A (1994) Isolation and characterization of the cytochrome bf complex from whole thylakoids, grana and stroma lamellae vesicles from spinach chloroplasts. Plant Cell Physiol 35: 557–568
Sane PV, Goodchild DJ and Park RB (1970) Characterization of chloroplast Photosystems I and 2 separated by a non-detergent method. Biochim Biophys Acta 216: 162–178
Sane PV (1977) The topography of the thylakoid membrane of the chloroplast. In: Trebst A and Avron M (eds) Encyclopedia of Plant Physiology New Series, Vol 5, pp 522–542. Springer Verlag, Berlin/Heidelberg/New York
Schmid GH, Jankowicz M and Menke W (1976) Cyclic photophosphrylation and chloroplast structure in the labellum of the orchid Aceras anthropophorum. J Microsc Biol Cell 26: 25–28
Shumway LK (1973) In: Edelstein SJ, Introductory Biochemistry. Holden-Day, Inc, San Francisco
Simpson D (1979) Freeze-fracture studies on Barley plastid membranes III. Location of the light-harvesting chlorophyll-protein. Carlsberg Res Commun 44: 305–336
Staehelin LA (1986) Chloroplast structure and supramolecular organization of photosynthetic membranes. In: Staehelin LA and Arnzen CJ (eds) Enzyclopedia of Plant Physiology, New Series, Vol 19, pp 1–84. Springer-Verlag, Berlin/Heidelberg/New York/Tokyo
Stefansson H, Wollenberger L and Albertsson P-Å (1994) Fragmentation and separation of the thylakoid membrane. Effect of light-induced protein phosphorylation on domain composition. J Chromatogr 668: 191–200
Svensson P and Albertsson P-Å (1989) Preparation of highly enriched Photosystem II membrane vesicles by a non-detergent method. Photosynth Res 20: 249–259
Svensson P, Andreasson E and Albertsson P-Å (1991) Heterogeneity among Photosystem I. Biochim Biophys Acta 1060: 45–50
Vallon O, Wollman FA and Olive J (1986) Lateral distribution of the main protein complexes of the photosynthetic apparatus in Chlamydomonas reinhardtii and in spinach: an immunocytochemical study using intact thylakoid membranes and a PS II enriched membrane preparation. Photobiochem Photobiophys 12: 203–220
Weibull C and Albertsson P-Å (1988) Ultrastructure of spinach thylakoids as seen in low-temperature and conventional embeddings. J Ultrastruct Mol Struct Res 100: 55–59
Whitmarsh I (1986) Mobile electron carriers in thylakoids. In: Staehlin LA and Arntzen CI (eds) Encyclopedia of Plant Physiology, New Series. Vol 19, pp 508–527. Springer-Verlag, Berlin/Heidelberg/New York/Tokyo
Wollenberger L, Stefansson H, Yu S-G and Albertsson P-Å (1994) Isolation and characterization of vesicles originating from the chloroplast grana margins. Biochim Biophys Acta 1184: 93–102
Wollenberger L, Weibull C and Albertsson P-Å (1995) Further characterization of the chloroplast grana margins: The non-detergent preparation of granal Photosystem I cannot reduce ferredoxin in the absence of NADP+ reduction. Biochim Biophys Acta 1230: 10–22
Yu S-G, Björn G and Albertsson P-Å (1993) Characterization of a non-detergent PS II-cytochrome b/f preparation (BS) Photosynth Res 37: 227–236
Yu S-G, Stefansson H, Romanowska E and Albertsson P-Å (1994) Two dimensional electrophoresis of thylakoid membrane proteins and its application to microsequencing. Photosynth Res 41: 475–486
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Albertsson, PÅ. The structure and function of the chloroplast photosynthetic membrane — a model for the domain organization. Photosynth Res 46, 141–149 (1995). https://doi.org/10.1007/BF00020424
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00020424