Abstract
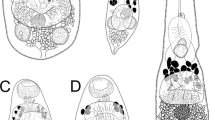
The great majority of the Procamallaninae occur in teleosts from tropical regions; however, representatives of this group are also frequent parasites of aquatic clawed toads (Xenopus spp.) in Africa. The taxonomic status of procamallanines from different Xenopus spp. and their geographical distribution is reviewed. Batrachocamallanus n. g. is created to include forms from amphibians with large numbers of mucrons on the female tail and relatively small body size. B. occidentalis n. sp. and B. siluranae n. sp. are described, while Procamallanus brevis Kung, 1948, originally recorded from an unidentified African amphibian, is considered a synonym of B. slomei (Southwell & Kirshner, 1937) n. comb. Due to the presence of spiral thickenings on its buccal capsule, B. xenopodis (Baylis, 1929) n. comb. has previously been placed in the genus Spirocamallanus Olsen, 1952. However, this species shares the apomorphic presence of numerous mucrons on the female tail, and almost identical cephalic morphology, male caudal structures and female reproductive system with other procamallanines from clawed toads. This suggests that they represent a monophyletic grouping. There is also only limited morphometric differentiation between B. xenopodis and the other proposed representatives of Batrachocamallanus (supported by a multivariate analysis of male and female specimens), which further indicates a close relationship between them. Great variability in the presence and type of buccal capsule thickenings occurs within Batrachocamallanus. Members of this genus most closely resemble the African species Procamallanus laeviconchus (Wedl, 1862), which exhibits a smooth buccal capsule similar to that of B. siluranae. Buccal capsule thickenings of the remaining Batrachocamallanus spp. probably arose independently from those described in other procamallanines. Such characters may be evolutionarily unstable and an unsuitable basis for generic classification in this subfamily. Although B. siluranae is the only Batrachocamallanus species to occur in X. tropicalis-like hosts (which represent a separate lineage from other clawed toads), its distribution, and that of its congeners, may be determined more by host-independent ecological or biogeographical factors than by an association with host phylogeny. Thus, B siluranae occurs in Xenopus spp. from tropical rain forest (including those from other host lineages) while the other forms are typically found in savanna or montane forest, and in the cases of B. slomei and B. xenopodis at least, do not show narrow host specificity to particular clawed toad taxa. Although more than one Batrachocamallanus spp. were found in X. laevis, X. muelleri and X. fraseri-like clawed toads, co-existence at the same locality never occurred, perhaps indicating a high degree of interspecific ecological segregation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ali, S.M. (1956) Studies on the nematode parasites of fishes and birds found in Hyderabad State. Indian Journal of Helminthology, 8, 1–83.
Ali, S.M. (1960) On two new species of Procamallanus Baylis, 1923 from India, with a key to the species. Indian Journal of Helminthology, 34, 129–138.
Avery, R.A. (1971) A preliminary list of parasites collected from reptiles and amphibians in northern Nigeria. British Journal of Herpetology, 4, 217–219.
Baker, M. (1987) A synopsis of the Nematoda parasitic in amphibians and reptiles. Occasional Papers in Biology, Memorial University of Newfoundland, 11, 325 pp.
Baylis, H.A. (1923) Report on a collection of parasitic nematodes, mainly from Egypt. Part III. Camallanidae, etc. Parasitology, 15, 24–38.
Baylis, H.A. (1929) Some parasitic nematodes from the Uluguru and Usambara Mountains, Tanganyika Territory. Annals and Magazine of Natural History, ser. 10, 4, 372–381.
Bookstein, F.L., Rernoff, B., Elder, R.L., Humphries, J.M., Smith, G.R. & Strauss, R.E. (1985) Morphometrics in evolutionary biology. The geometry of size and shape change, with examples from fishes. Special Publications Academy of Natural Sciences of Philadelphia, 15, 277 pp.
Chabaud, A.G. (1975) Keys to genera of the order Spirurida. Part 1. Camallanoidea, Dracunculoidea, Gnathostomatoidea, Physalopteroidea, Rictularioidea and Thelazioidea. In: Anderson, R.C.etal. (Eds) CIH Keys to the nematode parasites of vertebrates. Farnham Royal: Commonwealth Agricultural Bureaux. No. 3, 27 pp.
Frost, D.R. (1985) Amphibian species of the world. Lawrence, Kansas: Allen Press and The Association of Systematics Collections, 732 pp.
Hugot, J.-P. (1979) Description de cinq nouveaux nematodes d'un Tenrecoidea africain: Potamogale velox du Chaillu. Bulletin du Museum National d'Histoire Naturelle, Paris, 4e ser., 1, section A, no. 4, 1,057–1,073.
Ivashkin, V.M., Sobolev, A.A. & Khromova, L.A. (1971) Camallanata of animals and man and the diseases caused by them. Osnovy Nematodologii, 22, 381 pp. (In Russian: English translation, Jerusalem: IPST, 1977).
Khalil, L.F. (1969) Studies on the helminth parasites of freshwater fishes of the Sudan: Journal of Zoology, London, 158, 143–170.
Khalil, L.F. & Thurston, J.P. (1973) Studies on the helminth parasites of freshwater fishes of Uganda including the descriptions of two new species of digeneans. Revue de Zoologie et de Botanique Africaines, 87, 209–248.
Kung, C.C. (1948) On some new species of spirurids from terrestrial vertebrates, with notes on Habronema mansioni, Physaloptera paradoxa and Hartertia zuluensis. Journal of Helminthology, 22, 141–164.
Loumont, C. (1984) Current distribution of the genus Xenopus in Africa and future prospects. Revue Suisse de Zoologie, 91, 725–746.
Loumont, C. (1986) Xenopus pygmaeus, a new diploid pipid frog from rain forest of equatorial Africa. Revue Suisse de Zoologie, 93, 755–764.
Mashego, S.N. & Saayman, J.E. (1981) Observations on the prevalence of nematode parasites of the catfish, Clarias gariepinus (Burchell, 1822), in Lebowa, South Africa. South African Journal of Wildlife Research, 11, 46–48.
Moravec, F. (1974) On some nematodes of Egyptian freshwater fishes. Vestnik Ceskoslovenske Spolecnosti Zoologicke, 38, 32–51.
Moravec, F. (1975) The development of Procamallanus laeviconchus (Wedl, 1862) (Nematoda: Camallanidae). Vestnik Ceskoslovenske Spolecnosti Zoologicke, 39, 23–38.
Moravec, F. & Scholz, T. (1991) Observations on some nematodes parasitic in freshwater fishes in Laos. Folia Parasitologica, 38, 163–178.
Moravec, F. & Sey, O. (1988) Nematodes of freshwater fishes from north Vietnam. Part 1. Camallanoidea and Habronematoidea. Vestnik Ceskoslovenske Spolecnosti Zoologicke, 52, 128–148.
Olsen, L.S. (1952) Some nematodes parasitic in marine fishes. Publications of the Institute of Marine Science, University of Texas, 2, 173–215.
Petter, A.J. (1979) Essai de classification de la sous-famille des Procamallaninae (Nematoda, Camallanidae). Bulletin du Museum National d'Histoire Naturelle, Paris, 4e ser., 1, section A, no. 1, 219–239.
Petter, A.J. (1990) Nematodes de poissons du Paraguay. VI. Description de deux nouvelles especes du genre Spirocamallanus et complements a la description de Procamallanus annipetterae Kohn & Fernandes, 1988. Revue Suisse de Zoologie, 97, 327–338.
Pinto, R.M., De Fabio, S.P., Noronha, D. & Rolas, F.J.T. (1974) Procamallanus Brasileiros — parte 1 (Nematoda: Camallanoidea). Memorias do Instituto Oswaldo Cruz, 72, 205–211.
Pinto, R.M., De Fabio, S.P., Noronha, D. & Rolas, F.J.T. (1976) Novas consideracoes morfologicas e sistematicas sobre os Procamallanus Brasileiros (Nematoda, Camallanoidea). Memorias do Instituto Oswaldo Cruz, 74, 77–84.
Southwell, T & Kirshner, A. (1937) On some parasitic worms found in Xenopus laevis, the South African clawed toad. Annals of Tropical Medicine and Parasitology, 31, 245–266.
Stromberg, P.C. & Crites, J.L. (1974) Specialization, body volume, and geographical distribution of Camallanidae (Nematoda). Systematic Zoology, 23, 189–201.
Thatcher, V.E. (1991) Amazon fish parasites. Amazoniana, 11, 263–572.
Thurston, J.P. (1970) Studies on some Protozoa and helminth parasites of Xenopus, the African clawed toad. Revue de Zoologie et de Botanique Africaines, 82, 349–369.
Tinsley, R.C. (1973) Observations on Polystomatidae (Monogenoidea) from east Africa with a description of Polystoma makereri n. sp., Zeitschrift für Parasitenkunde, 42, 251–263.
Tinsley, R.C. (1981a) Interactions between Xenopus species (Anura: Pipidae). Monitore Zoologico Italiano, (N.S) Suppl., 15, 133–150.
Tinsley, R.C. (1981b) The evidence from parasite relationships for the evolutionary status of Xenopus (Anura: Pipidae). Monitore Zoologico Italiano, (N.S) Suppl., 15, 367–385.
Tinsley, R.C., Kobel, H.R. & Fischberg, M. (1979) The biology and systematics of a new species of Xenopus (Anura: Pipidae) from the highlands of central Africa. Journal of Zoology, London, 188, 69–102.
Tymowska, J. (1991) Polyploidy and cytogenetic variation in frogs of the genus Xenopus. In: Green, D.M. & Sessions, S.K. (Eds) Amphibian cytogenetics and evolution. London: Academic Press, pp. 259–297.
Watrous, L.E. & Wheeler, Q.D. (1981) The outgroup comparison method of character analysis. Systematic Zoology, 30, 1–11.
Wiley, E.O. (1981) Phylogenetics: The theory and practice of phylogenetic systematics. New York: Wiley, 439 pp.
Yeh, L.S. (1960) On a reconstruction of the genus Camallanus Railliet & Henry, 1915. Journal of Helminthology, 34, 117–124.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jackson, J.A., Tinsley, R.C. Representatives of Batrachocamallanus n. g. (Nematoda: Procamallaninae) from Xenopus spp. (Anura: Pipidae): geographical distribution, host range and evolutionary relationships. Syst Parasitol 31, 159–188 (1995). https://doi.org/10.1007/BF00009115
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00009115