Abstract
There exists an intricate relationship between attention and working memory. Recent work has further established that attention and working memory fluctuate synchronously, by tightly interleaving sustained attention and working memory tasks. This work has raised many open questions about physiological signatures underlying these behavioral fluctuations. Across two experiments, we explore pupil dynamics using real-time triggering in conjunction with an interleaved sustained attention and working memory task. In Experiment 1, we use behavioral real-time triggering and replicate recent findings from our lab (deBettencourt et al., 2019) that sustained attention fluctuates concurrently with the number of items maintained in working memory. Furthermore, highly attentive moments, detected via behavior, also exhibited larger pupil sizes. In Experiment 2, we develop a novel real-time pupil-triggering technique to track pupil size fluctuations in real time and trigger working memory probes. We show that this pupil triggering procedure reveals differences in sustained attention, as indexed by response time. These experiments reflect methodological advances in real-time triggering and further disentangle the relationship among general arousal, sustained attention, and working memory.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Attention and working memory are closely linked. In particular, recent work has interleaved sustained attention and working memory tasks to demonstrate that attention and working memory fluctuate synchronously (deBettencourt et al., 2019). That is, lapses of sustained attention covary with lapses of working memory (Adam & deBettencourt, 2019). Various biomarkers of attention and working memory have been discovered, including pupil size, which may provide access and insight into the interrelationship between these mechanisms (Robison & Unsworth, 2018; Unsworth & Robison, 2016). In particular, these biomarkers may provide important insight into the synchronous cognitive dynamics of attention and working memory.
Extensive research has implicated pupil size as a key measure of attention, arousal and general task engagement (Clewett et al., 2020; Eldar et al., 2013; Gilzenrat et al., 2010; Joshi et al., 2016; Kahneman & Beatty, 1966). In particular, differences in pupil size have been implicated in sustained attention (Decker et al., 2020; Unsworth & Robison, 2016; van den Brink et al., 2016) and working memory tasks (Robison & Brewer, 2020; Robison & Unsworth, 2018; Unsworth & Robison, 2015; Zokaei et al., 2019). Taken together with our previous research, this body of work has implicated pupil size as a relevant biomarker and motivated our exploration into whether pupil size fluctuations covaried with sustained attention and working memory fluctuations. To investigate this, we adapted our procedure to continuously record pupil size while participants performed our interleaved sustained attention and working memory task.
In particular, we leveraged real-time triggering to examine the relationship between sustained attention and working memory, as well as whether pupil size can provide a continuous index of these cognitive processes. Real-time triggering can be a powerful approach for exploring the consequences of endogenous cognitive and physiological fluctuations. Real-time triggering procedures can automatically and adaptively design experiments, contingent to fluctuations of cognitive state. That is, by tracking cognitive dynamics in real time, we can detect aberrant or optimal moments, whenever they occur. We have previously employed this experimental approach by examining behavioral data in real time and monitoring for lapse-prone attentional states (deBettencourt et al., 2019; deBettencourt et al., 2018). We successfully detected moments when attention was extremely high or low, which provided a powerful investigation of these infrequent but important moments. We also leveraged the additional power from the real-time triggering procedure to establish and examine the relationship between cognitive processes (e.g., attention and memory). Other related work has developed similar triggering approaches derived from biomarkers instead of behavior (Chew et al., 2019; Hinds et al., 2013; Yoo et al., 2012). This work inspired of extensions of our work, to examine biomarkers during our behavioral triggering procedure and to design triggering platforms contingent to biomarkers and examine the behavioral consequences.
The goal of this experiment is to explore how pupil size relates to sustained attention and working memory. In Experiment 1, we used real-time triggering derived from behavioral fluctuations of sustained attention. Behavioral real-time triggering tracked trial-to-trial fluctuations of response time and triggered working memory probes whenever prepotent responses were especially fast (i.e., lapsing attentional states) or slow (i.e., attentive states). In Experiment 2, we developed a novel real-time triggering procedure derived from fluctuations of pupil size. Pupil real-time triggering tracked trial-to-trial fluctuations of pupil size and triggered working memory probes whenever pupil sizes were especially large or small. These distinct and complementary real-time triggering procedures allow us to directly target extremely attentive and inattentive states, as measured by behavior and pupillometry.
Experiment 1
The goal of Experiment 1 was to examine physiological signatures that underlie sustained attention and working memory, using an interleaved task.
Methods
Participants
Thirty-five people participated in Experiment 1 for $25 payment (20 female, mean age = 25.5 years). Two participants left the study early without completing it. One participant accidentally participated twice; their second session was excluded from analysis. Two participants were excluded for working memory performance that was worse than chance-level guessing. This resulted in a final sample size of n = 30 participants. This sample size exceeds our target of 24, based on prior work (deBettencourt et al., 2019). All participants reported normal or corrected-to-normal color vision and provided informed consent to a protocol approved by the University of Chicago Social & Behavioral Sciences Institutional Review Board.
Apparatus
Participants were seated 75 cm away from an LCD monitor (120-Hz refresh rate). Stimuli were presented using Python and PsychoPy.
Stimuli
Stimuli were shapes, either circles (diameter = 1.5°) or squares (1.5° × 1.5°). Each display consisted of six shapes at 4° eccentricity. The shape positions were consistent for all trials to minimize intertrial visual transients. The color of each shape was one of nine distinct colors (red, blue, green, yellow, magenta, cyan, white, black, orange), and each display contained six shapes of random, unique colors. A central black fixation dot (0.1°) appeared at the center and turned white after a key press. For whole report working memory probes, a multicolored square (1.5° × 1.5°) composed of all nine colors appeared at each of the six locations, and the mouse cursor appeared at the central fixation position.
Procedure
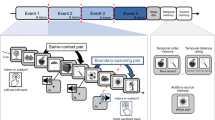
Participants completed an interleaved sustained attention and working memory task (Fig. 1a), adapted from a recent publication (deBettencourt et al., 2019). Critically, the sustained attention and working memory tasks relied on orthogonal dimensions of the stimuli: Shape was the relevant dimension for the sustained attention task, and color was the relevant dimension for the working memory task. The key difference from the previously published work was that the retention interval duration was increased (from 1 s to 2 s) to better capture slow pupillary responses. In total, participants completed five blocks of 800 trials. Due to time constraints, three participants completed only four blocks.
a Task design. Each trial was an array of six items (either circles or squares) of different colors. For the sustained attention task, participants responded to the shape, and for the working memory task participants reported the color. To encourage habitual responding, one of the shapes was much more frequent (10% lure trials, squares). For the whole report working memory task, after a 2-s delay, participants selected the color of each item. b Sustained attention accuracy. Accuracy was higher for lure versus nonlure trials (p < .001), a key signature of sustained attention tasks. The height of the bars depicts the average accuracy, and error bars are the standard error of the mean. Data from each participant are overlaid as small gray dots connected with lines. c Pupil size evoked by the sustained attention task. Lure trials (teal) evoked large pupil sizes than nonlure trials (black). Pupil size (unsmoothed, all trials) is plotted in arbitrary units (au) relative to a 100 ms baseline prior to the stimulus onset. Each line is the population average time course, and the shaded area is the standard error of the mean. d Working memory performance. In the whole report color working memory task, performance on each trial ranged from 0 (no items correct) to 6 (all items correct). The black line depicts the average proportion of trials, and error bars are within-subject standard error of the mean. Data from each participant are overlaid as small gray dots connected with gray lines. e Pupil size during the working memory retention. Pupil size increased during the blank retention interval. Pupil size (unsmoothed, all probe trials) is plotted in arbitrary units (au) relative to a 100 ms baseline prior to the stimulus onset. The black line is the population average time course, and the shaded area is the standard error of the mean
In the sustained attention task, participants viewed displays consisting of six circles or six squares. Their task was to respond according to the shape of the stimuli: If the shapes were squares, they pressed the “s” key, and if the shapes were circles, they pressed the “d” key. To effectively manipulate sustained attention, one shape was especially prevalent (approximately 90% circles, 10% squares). To reduce stimulus transients, the stimuli were at fixed positions and remained on the screen for 800 ms with no interstimulus interval in all experiments.
In the working memory task, participants were asked to remember the colors of each of the six shapes that most recently appeared. Whole report color working memory probes appeared infrequently; approximately 4%–5% of trials were probed. Memory probes only appeared after nonlure trials. During the first 80 trials, probes were inserted randomly. For the rest of the block, memory probe trials were inserted contingent to response behavior afterwards (see real-time triggering procedure below). During a working memory probe, all shapes disappeared and the screen went blank and gray with only the central black fixation dot for a brief delay interval (2 s). Then, multicolored squares, which included all nine possible colors, appeared at each location. The participants selected one of the nine colors at each location using the mouse cursor before the screen would advance. After making a response at a particular location, a large black square appeared around the outside of that entire multicolored square. After the last response, the screen went blank again (1 s) before resuming the sustained attention task.
Real-time triggering procedure
The goal was to use occasional working memory probes to link fluctuations in sustained attentional state with working memory performance. Rather than randomly inserting working memory probes, probes were “triggered” based on behavioral fluctuations of sustained attention. We tracked trial-by-trial fluctuations of sustained attentional state by continuously monitoring RT. For each trial i, we calculated a measure of attentional state using the average response time of the three most recent trials (i-3, i-2, i-1). We triggered memory probes whenever this measure of sustained attentional state, pretrial RTs, exceeded fast or slow thresholds, based on the cumulative mean and standard deviation (Trials 1, 2, …, i-1). These adaptive thresholds were individually tailored for each participant, which accounts for substantial individual variance in average RTs. We initiated the real-time triggering procedure after the first 80 trials, to obtain preliminary estimates of the mean and standard deviation for that block. This enabled the thresholds to be completely reset at the start of each block, thus accounting for any RT trends across blocks over the course of the experiment and continued to be adaptively updated across the trials within each block. In the sustained attention task, faster responses (faster than the mean RT minus the standard deviation) indicated a lapsing sustained attentional state, while slower responses (slower than the mean RT plus the standard deviation) indicated an attentive state. This is consistent with prior work using a short response deadline (deBettencourt et al., 2019; deBettencourt et al., 2018; cf. Cheyne et al., 2009). Whenever pretrial RTs exceeded one standard deviation away from the cumulative mean RT (either fast or slow), we triggered a memory probe. When a memory probe was triggered, a blank gray screen appeared with the fixation dot for the duration of the retention interval (2 s) and then the response screen appeared. Working memory probes belonged to the frequent category (i.e., circles).
While the RT-based thresholds were the primary criteria for triggering, we had additional criteria to optimize the targeting of attentional state, based on behavioral responses and eye-tracking measures. To ensure a reliable pretrial RT measure of attentional state, we required that participants had made responses to each of the three most recent trials. We also ensured that no probe trials had appeared in the three most recent trials. Finally, any eye blinks during the retention interval aborted the probe trial. Participants were not informed about the real-time triggering procedure.
Eye tracking
We monitored pupil size and gaze position using a desk-mounted infrared eye tracking system (EyeLink 1000 Plus, SR Research, Ontario, Canada). Eye tracking data were binocularly sampled at 1000 Hz, and head position was stabilized with a chin rest and calibrated with a 5-point calibration procedure. We report eye-tracking results for the left eye, but both eyes were highly reliably correlated (r = .98). Pupil size data are expressed in arbitrary units, corresponding to the size of the pupil as measured by the infrared camera. Using gaze position, distance, and pixel size, we calculated the degree of visual angle from central fixation. We detected missing data, blinks and saccades (>0.5°) using an automatic artifact pipeline, developed in our laboratory for analyzing eye-tracking data collected during EEG experiments. One possible concern is that pupil size differences are driven by stimulus-specific differences in luminance of the categorical colors. However, these stimuli were presented peripherally at 4° eccentricity, and 6 of 9 colors appeared on every trial.
Analysis
Behavioral performance was analyzed for the interleaved sustained attention and working memory task. Sustained attention performance to each trial was characterized using accuracy and RT. Accuracy to infrequent and frequent trials were combined into a single nonparametric measure of sensitivity of A′ and compared versus chance (0.5). A′ was calculated as A′ = 0.5 + (h − fa) ∗ (1 + h − fa)/(4 ∗ h ∗ (1 − fa)), where h corresponds to hit rate, and fa to false alarm rate (Donaldson, 1992). Whole report working memory performance was characterized as the number of items per trial for which the participants selected the correct color. To examine the influence of our real-time triggering design, we calculated the trailing window RT (what served to trigger memory probes). We also calculated the working memory performance for each memory probe.
Pupil size was also analyzed for the sustained attention and working memory task. We calculated the average pupil size evoked by each stimulus, subtracting a 100 ms prestimulus baseline from each trial. We compared the average pupil size during lure and nonlure trials after artifact rejection. We also calculated the average pupil size evoked for all timepoints during the working memory retention interval (n = 2,000) after artifact rejection, subtracting a 100 ms prestimulus baseline. To examine the pupil size revealed by real-time triggering, we calculated the pupil size of the stimulus that served as a memory probe. We examined the pupil size for all trials and also after artifact rejection. We depict unsmoothed pupillometry data, as many hundreds of trials were included in the analyses.
Statistics
Because some of the data violated the assumption of normality, all statistics were computed using a nonparametric random-effects approach in which participants were resampled with replacement 100,000 times. Null hypothesis testing was performed by calculating the proportion of the iterations in which the bootstrapped mean was in the opposite direction. Exact p values are reported; p values that were smaller than 1 in 1,000 are approximated as p < .001. Hypotheses were directional and thus one-sided unless otherwise noted. The mean and standard error of the mean are reported as descriptive statistics. Correlations were computed using the nonparametric Spearman rank-order correlation function. Effect sizes were computed as Cohen’s d. All data and code will be made available upon publication.
Results
We first examined whether the interleaved sustained attention and working memory task with eye tracking (Fig. 1a) revealed established behavioral and pupillary signatures. Sustained attention accuracy was lower for lure trials compared with nonlure trials (acclure = 56.86±3.04%; accnonlure = 95.59±0.85%; d = 2.50, p < .001; Fig. 1b), a well-characterized signature of sustained attention task designs. Lure trials evoked a larger pupillary response (snonlure = −0.03±3.42, slure = 29.36±4.60; d = 1.56, p < .001; Fig. 1c), calculated by averaging the pupil size over the time duration of the current trial and two subsequent trials (t = 0–2,400 ms). Even after excluding trials with incorrect responses, lure trials evoked a larger pupillary response (snonlure = −0.51±3.43, slure = 26.20±4.97; d = 1.22, p < .001). Overall, sustained attention performance was well above chance (A′ = 0.86±0.01, chance = 0.5; d = 4.46, p < .001). Working memory performance was calculated as the number of items correctly reported in working memory probes (m = 2.24±0.11). The blank retention interval (t = 0–2,000 ms) evoked a large pupil response (sret = 49.55±8.12; Fig. 1e). Working memory performance (m) was positively correlated with pupil size during the retention interval (sret) across individuals, such that higher accuracy was observed when the pupil was larger (r = .62; p < .001). These findings show that participants successfully performed the interleaved sustained attention and working memory task, and the task revealed established pupillary signatures.
Prior work has established that RT in similar sustained attention tasks tracks fluctuations of sustained attention. Specifically, faster responses in this task reflect worse attentional states, as when participants are responding more quickly, they are more likely to lapse (i.e., incorrectly respond to lure trials). We calculated the average preceding RTs over a trailing window of the three most recent trials. Indeed, we replicated prior work that faster responses precede lapses (RTlapse = 324±40 ms, RTnonlapse = 444±48 ms, p < .001). That is, in this task, faster responses to nonlure trials index worse attentional states.
We used real-time triggering to deploy working memory probes at specific moments, contingent to fluctuations of attentional state. As participants performed the sustained attention task, we tracked attentional state in real time by continually monitoring fluctuations of RT. Then, we triggered working memory probes (Fig. 2a) during moments when the participant was especially fast (operationalized as inattentive moments) or slow (operationalized as attentive moments). We adapted this real-time triggering procedure for concurrent eye tracking by extending the retention interval (2 s from 1 s) to accommodate the slower pupillary response and discontinuing any working memory probes if we detected a blink during the retention interval.
a Behavioral triggering. Behavioral fluctuations of RT (black line) were monitored in real time. We computed average RTs over a trailing window of the three most recent trials. Working memory probes (blue or green dots) were triggered whenever RT was faster than the fast threshold (blue, one standard deviation below average RT) or slower than the slow threshold (green, one standard deviation above average RT). b The real-time triggering procedure successfully detected faster RTs (blue) and slower RTs (green). The dashed gray line indicates the average RT before memory probes. c Working memory lapses concurrently with sustained attention. Participants remembered fewer items when working memory probes were triggered due to faster RTs (inattentive moments) versus slower RTs (attentive moments). The dashed gray line indicates the average working memory performance. d Pupil size differences covary with sustained attention. Average pupil size was computed over same interval as the trailing window for RTs. Pupil sizes were smaller when responses were faster (inattentive moments) versus slower (attentive moments). Bar heights depict the population average, and error bars are the standard error of the mean. Data from each participant are overlaid as small gray dots
First, we investigated whether this real-time triggering manipulation was successful. Overall, 146±6 probes were triggered in real time over the course of the experiment. Indeed, we successfully triggered trials when participants were responding fast or slow (RTfast = 240±8 ms, RTslow = 519±13 ms; Fig. 2b). That is, we continuously monitored behavior so as to detect attentive or inattentive moments. There was no reliable difference between the numbers of fast and slow trials per participant (nfast = 74.2±3.8, nslow = 72.1±3.6; d = 0.13, p = .61).
Next, we examined whether attention fluctuated concurrently with working memory. If so, then participants would remember fewer items during inattentive moments. Indeed, we found that working memory performance was worse for fast- versus slow-triggered trials (mfast = 2.20±0.10, mslow = 2.29±0.11; d = 0.35; p = .03; Fig. 2c). This behavioral pattern replicates the findings from our previous study—that sustained attention and working memory lapse concurrently.
Finally, we examined whether real-time triggering also captured differences in pupil size. Prior to fast-triggered working memory probes, pupil sizes were reliably smaller (sfast = −9.12±5.22, sslow = 9.05±5.45; d = 0.31, p = 0.04; Fig. 2d). This result was remained reliable even after excluding any trials with eye artifacts (sfast = −13.96±5.67; sslow = 14.67±6.41; d = 0.44, p = .008). Thus, real-time triggering based on behavior revealed smaller pupil size during inattentive trials. Note that we are not claiming that RT and pupil size are redundant or directly equivalent measures of attentional state. Rather, that these moments detected by our real-time triggering method also detected moments with reliable differences in pupil size.
Discussion
This experiment replicated the behavioral finding that sustained attention and working memory lapse concurrently, initially demonstrated in a prior publication from our laboratory (deBettencourt et al., 2019). We also extended these results to demonstrate that moments of low attention, operationalized as response time and detected by our automatic real-time triggering procedure, also reflected moments when the pupil size is smaller. These results also raise an important question about whether pupil size could directly serve as an independent and continuous index to track fluctuations of sustained attention and/or working memory. This required two key goals (1) to update our procedure to adaptively design our experiment contingent to the dynamics of a biomarker, namely tonic fluctuations of pupil size and (2) examine whether this real-time triggering procedure revealed moment-to-moment covariation between (a) pupil size and sustained attention and (b) pupil size and working memory.
Experiment 2
The goals of Experiment 2 were to develop a real-time triggering procedure based on moment-to-moment fluctuations of pupil size to explore the consequences of this triggering procedure for behavior.
Methods
Participants
Twenty-seven people participated in Experiment 2 for $25 payment (14 female, mean age = 23.9 years). Two participants left the study early without completing it. One subject was excluded due to multiple issues with data collection during the first two blocks of the task. Three individuals who had participated in Experiment 1 also participated in Experiment 2. This sample size matches our target of 24, based on prior work (deBettencourt et al., 2019). All participants reported normal or corrected-to-normal color vision and provided informed consent to a protocol approved by the University of Chicago Social & Behavioral Sciences Institutional Review Board.
Apparatus and stimuli
Apparatus and stimuli are the same as in Experiment 1.
Procedure
Procedure was the same as in Experiment 1. The difference was that the working memory probes appeared contingent to fluctuations of pupil diameter, not RT. In total, participants completed 5 blocks of 800 trials. For one participant, the response keys for the sustained attention task were swapped. For another participant, only data from the last four blocks was included in the analysis due to multiple errors in the first block.
Real-time triggering procedure
Rather than randomly distributing working memory probes, memory probes were “triggered” based on real-time pupil size fluctuations. For each trial i, we calculated the average pupil size of the left pupil using a trailing window over the three most recent trials (i-3, i-2, i-1). We triggered memory probes whenever pupil sizes were especially small or large. In real time, we developed individually tailored and adaptively updated thresholds inspired by our procedure from RT triggering, using the cumulative mean and standard deviation of pupil size (from trials 1, 2, …, i-1). When the trailing window of pupil size exceeded one standard deviation away from the cumulative mean pupil size, a working memory probe was triggered. We initiated the real-time triggering procedure after the first 80 trials. We also ensured that no probe trials had appeared in the three most recent trials. In addition, any eye blinks during the retention interval aborted the probe trial. Participants were not informed that their pupils controlled when memory probes would appear.
Eye tracking
For the first two subjects, eye tracking data were binocularly sampled at 1000 Hz. To accelerate real-time computation, we switched to monocular tracking of the left eye for all subsequent subjects. All other details were the same as Experiment 1.
Analysis
In addition to the analyses from Experiment 1, we examined the continuous relationship between sustained attention behavior and working memory behavior. To do this, we selected all working memory probes that were triggered for each participant, regardless of whether they were triggered due to large or small pupil sizes. For each participant, we correlated our index of sustained attention (i.e., pretrial RTs) with our index of working memory (the number correct on the working memory probe). Then, we examined whether across participants there was a reliably positive correlation. To conduct statistics across participants, we z-transformed the r values.
Statistics
Statistics were the same as in Experiment 1.
Results
Sustained attention accuracy was lower for lure trials compared with nonlure trials (acclure = 51.32±3.72%; accnonlure = 94.60±0.84%; d = 2.50, p < .001; Fig. 3a). Lure trials evoked a larger pupillary response (snonlure = −2.29±3.48, slure = 27.68±6.48; d = 1.00, p < .001; Fig. 3b). Even after excluding trials with incorrect responses, lure trials evoked a larger pupillary response (snonlure = −2.99±3.52, slure = 27.68±6.48; d = 1.12, p < .001). Overall, sustained attention performance was well above chance (A ′ = 0.84±0.02, chance = 0.5; d = 4.41, p < .001). Working memory performance was calculated as the number of items correctly reported in working memory probes (m = 2.07±0.13; Fig. 3c). The blank retention interval (t=0–2000 m) evoked a large pupil response (sret = 58.47±9.81; Fig. 3d). Working memory performance (m) was positively correlated with pupil size evoked during the retention interval (sret) across individuals (r = .50; p = .01). These findings replicated those from Experiment 1 and demonstrated that the new pupil triggering task revealed established pupillary signatures.
a Sustained attention accuracy. Accuracy was higher for lure versus nonlure trials (p < .001), a key signature of sustained attention tasks. The height of the bars depicts the average accuracy, and error bars are the standard error of the mean. Data from each participant are overlaid as small gray dots connected with lines. b Pupil size evoked by the sustained attention task. Lure trials (teal) evoked large pupil sizes than nonlure trials (black). Pupil size (unsmoothed, all probe trials) is plotted in arbitrary units (au) relative to a 100 ms baseline prior to the stimulus onset. Each line is the population average time course, and the shaded area is the standard error of the mean. c Working memory performance. In the whole report color working memory task, performance on each trial ranged from 0 (no items correct) to 6 (all items correct). The black line depicts the average proportion of trials, and error bars are within-subject standard error of the mean. Data from each participant are overlaid as small gray dots connected with gray lines. d Pupil size during the working memory retention. Pupil size increased during the blank retention interval. Pupil size (unsmoothed, all probe trials) is plotted in arbitrary units (au) relative to a 100-ms baseline prior to the stimulus onset. The black line is the population average time course, and the shaded area is the standard error of the mean
We also replicated the finding that RT is a signature of sustained attentional state. We calculated a measure of attentional state by averaging RT over a trailing window of the 3 most recent trials. Indeed, faster responses preceded lapses (RTlapse = 311±38 ms, RTnonlapse = 372±42 ms, d = 2.09, p < .001). Therefore, in this task, we again verified that faster responses reflected moments of worse sustained attention.
The critical difference is that Experiment 2 used pupil real-time triggering to deploy working memory probes at specific moments, contingent to fluctuations of pupil size (rather than RT). As participants performed the sustained attention task, we continually monitored fluctuations of pupil size via real-time eye tracking. Then, we triggered working memory probes (Fig. 4a) during moments when the pupil sizes were especially small or large (# probes = 190±10).
a Pupil real-time triggering example. Pupil size fluctuations (black line) were monitored in real time. We computed averaged pupil size over a trailing window of the three most recent trials. Working memory probes (blue or green dots) were triggered whenever pupil size was smaller than the small threshold (blue, one standard deviation below average pupil size) or larger than the large threshold (green, one standard deviation above average pupil size). b Real-time triggering successfully detected extreme differences in pretrial pupil sizes. The average pupil size was computed over the preceding three trials before memory probes. As expected by the real-time triggering design, the procedure successfully detected moments of small (blue) and large (green) pupils. The dashed line indicates the average pupil size before memory probes. c Sustained attention covaries with pupil size differences. RT was measured over the three trials preceding each memory probe. Small (blue) versus large (green) pupils covaried with sustained attention behavior (RTs, averaged over the three preceding trials). Smaller pupils covaried with worse sustained attention (i.e., faster RTs). The dashed line indicates the mean pretrial RTs. d Pupil size differences do not covary with working memory fluctuations. Working memory was operationalized as the number correct on the whole report working memory probe. Small (blue) and large (green) pupils did not influence working memory performance. The dashed line indicates the mean working memory accuracy for real-time triggered memory probes. The height of the bar depicts the population average, and error bars are within-subject standard error of the mean. Data from each participant are overlaid as small gray dots
First, we investigated whether this real-time triggering manipulation was successful. Indeed, we successfully triggered trials when participants’ pupils were large or small (ssmall = −194±21, slarge = 243±24; Fig. 4b), relative to mean pupil size. That is, we successfully continuously monitored for deviations in pupil size as participants performed the sustained attention task.
We next examined whether our measure of sustained attention, RT, fluctuated concurrently with pupil size. If so, then participants would be responding more quickly when pupil size was smaller. Indeed, we found that RTs were faster for small- versus large-triggered trials (RTsmall = 336±10 ms; RTlarge = 353±11 ms; d = 0.70; p < .001; Fig. 4c). This demonstrates the complement of the relationship from Experiment 1, that real-time triggering based on pupil size can reveal reliable differences in RT.
We then examined whether pupil triggering would also predict differences in working memory performance. We did not observe a reliable effect of pupil triggering on working memory behavior (msmall = 2.08±0.13; mlarge = 2.08±0.14; d = 0.001, p = .50; Fig. 4d).
Finally, we examined whether these results were consistent with Experiment 1, by considering the relationship between attention and working memory behavior. For each participant, we selected all triggered working memory probes, regardless of whether they were triggered due to small or large pupils. Then, we correlated the behavioral measure of sustained attention (i.e., pretrial RTs) with the behavioral measure of working memory (number correct for that memory probe). Therefore, for each participant, we obtain a correlation coefficient, and we examine the reliability of the correlation coefficients across participants. Across participants, there was a modest but reliably positive relationship between pretrial RTs and working memory performance (mean r = .03±0.02; d = 0.36, p = .04). As such, these results confirmed our prior demonstrations of synchronous fluctuations between sustained attention, as indexed by RT, and working memory behavior.
Discussion
In Experiment 2, we developed real-time pupil triggering procedure to track fluctuations of a physiological measure as participants performed an interleaved sustained attention and working memory task. We observed a reliable relationship between average evoked pupil size and aggregate working memory performance. Furthermore, pupil real-time triggering was sufficient to reveal reliable changes in sustained attention behavior. However, our real-time triggering procedure did not give rise to differences in working memory accuracy, suggesting that tonic differences in pupil size did not account for the observed covariation between attention and working memory. As such, we explore both intra- and inter-individual relationships between tonic vs. evoked pupil sizes and working memory. Evoked pupil sizes were correlated with working memory across individuals, but tonic pupil sizes were not predictive of working memory performance fluctuations within individuals. These findings explore a new frontier for how real-time triggering can be used to covertly and continuously track cognitive states via biomarkers in conjunction with sophisticated behavioral paradigms. Future work could further improve and refine these pupil triggering procedures. In addition, this work suggests ways to disentangle the relationship between attention and working memory. That is, sustained attention might reflect multiple components, including task-specific stimulus processing and general arousal, and the former might account for the relationship with working memory while the latter might account for the relationship with pupil sizes.
General discussion
Across two experiments we measured pupil size as participants performed an interleaved sustained attention and working memory task. We triggered working memory probes in real time at precise moments, either based on RTs to the sustained attention task (Experiment 1) or pupil size (Experiment 2). These experiments shed further light on the relationship between sustained attention and working memory, and how pupil dynamics is implicated in these cognitive processes. First, across both experiments, we observed that the sustained attention task and the retention interval elicited canonical pupil responses. Second, in Experiment 1, we replicated the behavioral covariation between sustained attention and working memory, showing that WM performance was lower when subjects were performing worse on the sustained attention task. Furthermore, we identified that these moments of low sustained attention also reflect smaller pupil size. In the second experiment, we explore pupil dynamics more directly, by designing a real-time triggering procedure contingent to fluctuations of tonic pupil size. This platform revealed that pupil dynamics track differences in sustained attention, but not working memory. In sum, via complementary automated real-time triggering systems, both experiments explore the theoretical conceptualization of attention, arousal, and working memory, and implicate pupil size in this intricate relationship.
These findings reveal new insight into our theoretical understanding of sustained attention, and its interaction with other prominent cognitive processes, including arousal and working memory. This lends additional support into the conceptualization of sustained attention as a complex cognitive process, encompassing multiple, distinct subcomponent processes (Adam et al., 2015; Hakim et al., 2020). In this study and in our prior work (deBettencourt et al., 2019), we index sustained attention dynamics behaviorally via RT and show that these behavioral dynamics covary with the number of items held in working memory. One hypothesis is that this observed covariation between sustained attention and working memory reflects a shared reliance on general arousal. However, in this study, we examine a putative biomarker of arousal, tonic pupil size. We discover that pupil size dynamics selectively covary with attention, but not working memory. This suggests that differences of arousal, as indexed by pupil size, cannot explain the observed covariation between attention and working memory. The observed covariation may reflect a shared cognitive resource, potentially related to higher-level cognition or stimulus processing. On the other hand, this suggests that real-time triggering contingent to pupil size would be a powerful approach to manipulate sustained attention. In sum. this suggests new insights into the intricate and complex relationship between sustained attention, arousal, and working memory.
Real-time triggering
This study demonstrates the potential for real-time triggering techniques from multiple behavioral and/or physiological indices within the context of the same task. As we have also demonstrated in prior work (deBettencourt et al., 2019), this technique can directly target moments that may be infrequent but important. By monitoring behavioral or pupil size fluctuations in real time, we develop powerful means to adaptively design the experiment. In this study, we employed two distinct real-time triggering procedures. First, we focused on RT fluctuations in Experiment 1, and next we focused on pupil size fluctuations in Experiment 2. These procedures allow prospective and a priori experimental designs, that specifically control for other potential explanatory variables (for example, stimulus type, accuracy). Through behavioral real-time triggering (Fig. 5a), we can investigate the consequences of extreme differences in attentional state (operationalized by speed of responding), which would be unlikely to be sampled randomly, and reveal reliable differences in pupil size and working memory. Alternatively, pupil real-time triggering can be used to obtain a sensitive assay of how variation in pupil size track variations in cognitive performance by emphasizing the trials with the biggest differences in pupil size (Fig. 5b) and reveal modest yet reliable differences in responding. This work opens the potential for designing experiments where real-time triggering is derived jointly from behavior and physiological measures, either during moments of convergence (e.g., slow responses and large pupils) or divergence (e.g., slow responses and small pupils). By comparing the behavioral consequences of multiple indices, we can begin to disentangle complex cognitive dynamics.
a Behavioral triggering tracks extreme differences in RT. A histogram of RTs from all trials of a representative participant in Experiment 1 is depicted in gray. Trials that are eligible for triggering are overlaid, either fast trials (blue) or slow trials (green). The black line depicts RTs in milliseconds (ms) that would have been obtained by randomly sampling. b Pupil triggering tracks extreme differences in pupil size. A histogram of pupil size from all trials of a representative participant in Experiment 2 is depicted in gray. Trials that are eligible for triggering are overlaid, either small trials (blue) or large trials (green). The black line depicts pupil sizes in arbitrary units (au) that would have been obtained by randomly sampling
Pupil triggering
A key advance in Experiment 2 is the development of real-time pupil triggering for this interleaved sustained attention and working memory task. Currently, most attempts to track sustained attentional states in real time have required overt and repetitive behavioral responses. Developing techniques to track attention using pupil size could examine cognitive dynamics covertly and continuously, even without any behavioral response demands (Mathôt et al., 2016). The development of abilities to covertly track sustained attentional state could be especially beneficial in educational scenarios to improve learning outcomes or occupational scenarios where even subtle fluctuations of attention could be catastrophic. For pupil real-time triggering, we designed our task based on the pupil signatures observed in Experiment 1. However, other work has implicated that intermediate pupil sizes might be more optimal than high or low (Murphy et al., 2011). Future studies could further improve on this initial demonstration of pupil real-time triggering and can attempt to build upon these findings to develop procedures to prospectively target working memory states. While our studies here did not reveal any nonmonotonic relationship between pupil size and cognitive performance, that was also not the focus of our investigation. Future work using real time triggering methods may choose to more specifically and selectively target certain extreme pupil sizes (very large vs. slightly large, very small vs. slightly small) and/or target intermediate pupil size (close to the median pupil size). Alternatively, future work could investigate the relationship between pupil size during the retention interval and performance on working memory tasks by adaptively shortening the retention interval based on fluctuations in pupil size. As such, real-time triggering presents a robust platform that can be precisely tailored to investigate a specific brain-biomarker relationship.
In this study, we present positive steps forward in exploring the cognitive states and biomarkers underlying lapsing attention. Our findings reveal that lapsing sustained attention states may co-occur with differences in pupil size. This provides additional, complementary insight into the complex physiological signatures that underly lapses of sustained attention (deBettencourt et al., 2015; Esterman & Rothlein, 2019; Rosenberg et al., 2016). In sum, examining pupil size sheds light on moment-by-moment and trial-by-trial fluctuations of sustained attention.
References
Adam, K. C. S., & deBettencourt, M. T. (2019). Fluctuations of attention and working memory. Journal of Cognition, 2(1), 33. https://doi.org/10.5334/joc.70
Adam, K. C. S., Mance, I., Fukuda, K., & Vogel, E. K. (2015). The contribution of attentional lapses to individual differences in visual working memory capacity. Journal of Cognitive Neuroscience, 27(8), 1601–1616. https://doi.org/10.1162/jocn_a_00811
Chew, B., Hauser, T. U., Papoutsi, M., Magerkurth, J., Dolan, R. J., & Rutledge, R. B. (2019). Endogenous fluctuations in the dopaminergic midbrain drive behavioral choice variability. Proceedings of the National Academy of Sciences, 116(37), 18732–18737. https://doi.org/10.1073/pnas.1900872116
Cheyne, J. A., Solman, G. J., Carriere, J. S., & Smilek, D. (2009). Anatomy of an error: A bidirectional state model of task engagement/disengagement and attention-related errors. Cognition, 111(1), 98–113. https://doi.org/10.1016/j.cognition.2008.12.009
Clewett, D., Gasser, C., & Davachi, L. (2020). Pupil-linked arousal signals track the temporal organization of events in memory. Nature Communications, 11(1), 4007. https://doi.org/10.1038/s41467-020-17851-9
deBettencourt, M. T., Cohen, J. D., Lee, R. F., Norman, K. A., & Turk-Browne, N. B. (2015). Closed-loop training of attention with real-time brain imaging. Nature Neuroscience, 18(3), 470–475. https://doi.org/10.1038/nn.3940
deBettencourt, M. T., Keene, P. A., Awh, E., & Vogel, E. K. (2019). Real-time triggering reveals concurrent lapses of attention and working memory. Nature Human Behaviour, 3(8), 808–816. https://doi.org/10.1038/s41562-019-0606-6
deBettencourt, M. T., Norman, K. A., & Turk-Browne, N. B. (2018). Forgetting from lapses of sustained attention. Psychonomic Bulletin & Review, 25(2), 605–611. https://doi.org/10.3758/s13423-017-1309-5
Decker, A., Finn, A., & Duncan, K. (2020). Errors lead to transient impairments in memory formation. Cognition, 204, Article 104338. https://doi.org/10.1016/j.cognition.2020.104338
Donaldson, W. (1992). Measuring recognition memory. Journal of Experimental Psychology. General, 121(3), 275–277. https://doi.org/10.1037//0096-3445.121.3.275
Eldar, E., Cohen, J. D., & Niv, Y. (2013). The effects of neural gain on attention and learning. Nature Neuroscience, 16(8), 1146–1153. https://doi.org/10.1038/nn.3428
Esterman, M., & Rothlein, D. (2019). Models of sustained attention. Current Opinion in Psychology, 29, 174–180. https://doi.org/10.1016/j.copsyc.2019.03.005
Gilzenrat, M. S., Nieuwenhuis, S., Jepma, M., & Cohen, J. D. (2010). Pupil diameter tracks changes in control state predicted by the adaptive gain theory of locus coeruleus function. Cognitive, Affective, & Behavioral Neuroscience, 10(2), 252–269. https://doi.org/10.3758/CABN.10.2.252
Hakim, N., deBettencourt, M. T., Awh, E., & Vogel, E. K. (2020). Attention fluctuations impact ongoing maintenance of information in working memory. Psychonomic Bulletin & Review. https://doi.org/10.3758/s13423-020-01790-z
Hinds, O., Thompson, T. W., Ghosh, S., Yoo, J. J., Whitfield-Gabrieli, S., Triantafyllou, C., & Gabrieli, J. D. E. (2013). Roles of default-mode network and supplementary motor area in human vigilance performance: Evidence from real-time fMRI. Journal of Neurophysiology, 109(5), 1250–1258. https://doi.org/10.1152/jn.00533.2011
Joshi, S., Li, Y., Kalwani, R. M., & Gold, J. I. (2016). Relationships between pupil diameter and neuronal activity in the locus coeruleus, colliculi, and cingulate cortex. Neuron, 89(1), 221–234. https://doi.org/10.1016/j.neuron.2015.11.028
Kahneman, D., & Beatty, J. (1966). Pupil diameter and load on memory. Science, 154(3756), 1583–1585. https://doi.org/10.1126/science.154.3756.1583
Mathôt, S., Melmi, J.-B., van der Linden, L., & Van der Stigchel, S. (2016). The mind-writing pupil: A human-computer interface based on decoding of covert attention through pupillometry. PLOS ONE, 11(2), Article e0148805. https://doi.org/10.1371/journal.pone.0148805
Murphy, P. R., Robertson, I. H., Balsters, J. H., & O’connell, R. G. (2011). Pupillometry and P3 index the locus coeruleus-noradrenergic arousal function in humans. Psychophysiology, 48(11), 1532–1543. https://doi.org/10.1111/j.1469-8986.2011.01226.x
Robison, M. K., & Brewer, G. A. (2020). Individual differences in working memory capacity and the regulation of arousal. Attention, Perception, & Psychophysics. https://doi.org/10.3758/s13414-020-02077-0
Robison, M. K., & Unsworth, N. (2018). Pupillometry tracks fluctuations in working memory performance. Attention, Perception, & Psychophysics. https://doi.org/10.3758/s13414-018-1618-4
Rosenberg, M. D., Finn, E. S., Scheinost, D., Papademetris, X., Shen, X., Constable, R. T., & Chun, M. M. (2016). A neuromarker of sustained attention from whole-brain functional connectivity. Nature Neuroscience, 19(1), 165–171. https://doi.org/10.1038/nn.4179
Unsworth, N., & Robison, M. K. (2015). Individual differences in the allocation of attention to items in working memory: Evidence from pupillometry. Psychonomic Bulletin & Review, 22(3), 757–765. https://doi.org/10.3758/s13423-014-0747-6
Unsworth, N., & Robison, M. K. (2016). Pupillary correlates of lapses of sustained attention. Cognitive, Affective, & Behavioral Neuroscience, 16(4), 601–615. https://doi.org/10.3758/s13415-016-0417-4
van den Brink, R. L., Murphy, P. R., & Nieuwenhuis, S. (2016). Pupil diameter tracks lapses of attention. PLoS ONE, 11(10), e0165274. https://doi.org/10.1371/journal.pone.0165274
Yoo, J. J., Hinds, O., Ofen, N., Thompson, T. W., Whitfield-Gabrieli, S., Triantafyllou, C., & Gabrieli, J. D. E. (2012). When the brain is prepared to learn: Enhancing human learning using real-time fMRI. NeuroImage, 59(1), 846–852. https://doi.org/10.1016/j.neuroimage.2011.07.063
Zokaei, N., Board, A. G., Manohar, S. G., & Nobre, A. C. (2019). Modulation of the pupillary response by the content of visual working memory. Proceedings of the National Academy of Sciences, 116(45), 22802–22810. https://doi.org/10.1073/pnas.1909959116
Acknowledgements
This research was supported by National Institute of Health grant R01MH087214, the Office of Naval Research grant N00014-12-1-0972, and F32MH115597 (M.T.dB.).
Author information
Authors and Affiliations
Contributions
All authors conceived of the study and contributed to the study design. P.A. Keene and M.T. deBettencourt collected and analyzed the data. P.A. Keene and M.T. deBettencourt wrote an initial draft of the manuscript, which all authors read and edited.
Corresponding author
Ethics declarations
Competing interests
No competing interests.
Additional information
Open practices statement
The data and materials for all experiments are available online (https://github.com/debetten/KeenedeBettencourt_PupilTrigger), and none of the experiments was preregistered.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Keene, P.A., deBettencourt, M.T., Awh, E. et al. Pupillometry signatures of sustained attention and working memory. Atten Percept Psychophys 84, 2472–2482 (2022). https://doi.org/10.3758/s13414-022-02557-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3758/s13414-022-02557-5