Abstract
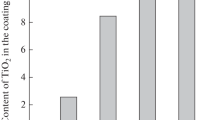
Deep eutectic solvents are nowadays considered to be very promising analogues of room temperature ionic liquids. They can make a significant contribution to the development of novel efficient, economic and environmentally friendly processes, particularly, in surface engineering and electroplating. The electrodeposition of Ni–TiO2 composite coatings from an electrolyte based on a deep eutectic solvent, ethaline, was studied in this work. Titania particles were introduced into the plating bath in the form of Degussa P 25 nanopowder. The content of titania in electrodeposited composite coatings was shown to depend on the stirring rate, current density and TiO2 concentration in the electrolyte and can reach ca. 2.35 wt %. The microstructure and the surface morphology of the obtained composite layers were characterized. The formation of nanocrystalline nickel matrix was detected. The introduction of TiO2 particles into nickel-based coatings resulted in an increase in the microhardness of deposits. The data obtained via the electrochemical impedance spectra technique revealed an improvement in the corrosion resistance of coatings due to titania particles incorporation into deposits. The Ni–TiO2 composite coatings manifested a photocatalytic activity towards the reaction of photochemical degradation of methylene blue organic dye in water solution.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Low, C.T.J., Wills, R.G.A., and Walsh, F.C., Surf. Coat. Technol., 2006, vol. 201, pp. 371–383.
Walsh, F.C. and Ponce de Leon, C. Trans. Inst. Met. Finish., 2014, vol. 92, pp. 83–98.
Ahmad, Y.H. and Mohamed, A.M.A., Int. J. Electrochem. Sci., 2014, vol. 9, pp. 1942–1963.
Li, R., Hou, Y., Liu, B., Wang, D., et al., Electrochim. Acta, 2016, vol. 222, pp. 1272–1280.
Dehgahi, S., Amini, R., and Alizadeh, M., Surf. Coat. Technol., 2016, vol. 304, pp. 502–511.
Eroglu, D. and West, A.C. J. Electrochem. Soc., 2013, vol. 160, pp. D354–D360.
Lapinski, J., Pletcher, D., and Walsh, F.C., Surf. Coat. Technol., 2011, vol. 205, pp. 5205–5209.
Zhang, Z., Wu, X., Jiang, C., and Ma, N., Surf Eng., 2014, vol. 30, pp. 21–25.
Thiemig, D. and Bund, A. Surf. Coat. Technol., 2008, vol. 202, pp. 2976–2984.
Chen, W. and Gao, W. Electrochim. Acta, 2010, vol. 55, pp. 6865–6871.
Chen, W., He, Y., and Gao, W., Surf. Coat. Technol., 2010, vol. 204, pp. 2487–2492.
Spanou, S., Kontos, A.I., Siokou, A., Kontos, A.G., et al., Electrochim. Acta, 2013, vol. 105, pp. 324–332.
Mohajeri, S., Dolati, A., and Ghorbani, M., Int. J. Electrochem. Sci., 2017, vol. 12, pp. 5121–5141.
Abdel Aal, A. and Hassan, H.B. J. Alloys Compd., 2009, vol. 477, pp. 652–656.
Mohajeri, S., Dolati, A., and Ghorbani, M., Surf. Coat. Technol., 2015, vol. 262, pp. 173–183.
Mohan Reddy, R., Praveen, B.M., and Praveen Kumar, C.M., Surf. Eng. Appl. Electrochem., 2017, vol. 53, pp. 179–185.
Mohan Reddy, R., Praveen, B.M., Praveen Kumar, C.M., and Venkatesha, T.V., Surf. Eng. Appl. Electrochem., 2017, vol. 53, pp. 258–264.
Chen, X. and Mao, S.S. Chem. Rev., 2007, vol. 107, pp. 2891–2959.
Danilov, F.I., Tsurkan, A.V., Vasil’eva, E.A., and Protsenko, V.S., Int. J. Hydrogen Energy, 2016, vol. 41, pp. 7363–7372.
Vasil’eva, E.A., Tsurkan, A.V., Protsenko, V.S., and Danilov, F.I., Prot. Met. Phys. Chem. Surf., 2016, vol. 52, pp. 532–537.
Protsenko, V.S., Vasil’eva, E.A., Smenova, I.V., Baskevich, A.S., et al., Surf. Eng. Appl. Electrochem., 2015, vol. 51, pp. 65–75.
Abbott, A.P., Ryder, K.S., and Konig, U., Trans. Inst. Met. Finish., 2008, vol. 86, pp. 196–204.
Smith, E.L., Abbott, A.P., and Ryder, K.S., Chem. Rev., 2014, vol. 114, pp. 11060–11082.
Abbott, A.P., Capper, G., Davies, D.L., Rasheed, R.K., et al., Chem. Commun., 2003, vol. 1, pp. 70–71.
Abbott, A.P., Boothby, D., Capper, G., Davies, D.L., et al., J. Am. Chem. Soc, 2004, vol. 126, pp. 9142–9147.
Abbott, A.P., El Ttaib, K., Frisch, G., Ryder, K.S., et al., Phys. Chem. Chem. Phys., 2012, vol. 14, pp. 2443–2449.
Li, R., Hou, Y., and Liang, J., Appl. Surf. Sci., 2016, vol. 367, pp. 449–458.
Li, R., Chu, Q., and Liang, J., RSC Adv., 2015, vol. 5, pp. 44933–44942.
Abbott, A.P., El Ttaib, K., Frisch, G., McKenzie, K.J., et al., Phys. Chem. Chem. Phys., 2009, vol. 11, pp. 4269–4277.
Pereira, N.M., Brincoveanu, O., Pantazi, A.G., Pereira, C.M., et al., Surf. Coat. Technol., 2017, vol. 324, pp. 451–462.
Abbott, A.P., Ballantyne, A., Harris, R.C., Juma, J.A., et al., Electrochim. Acta, 2015, vol. 176, pp. 718–726.
Kityk, A.A., Shaiderov, D.A., Vasil’eva, E.A., Protsenko, V.S., and Danilov, F.I., Electrochim. Acta, 2017, vol. 245, pp. 133–145.
Protsenko, V.S., Vasil’eva, E.A., Tsurkan, A.V., Kityk, A.A., et al., J. Environ. Chem. Eng., 2017, vol. 5, pp. 136–146.
Fransaer, J., Celis, J.P., and Roos, J.R., J. Electrochem. Soc., 1992, vol. 139, pp. 413–425.
Maurin, G. and Lavanant, A. J. Appl. Electrochem., 1995, vol. 25, pp. 1113–1121.
Guglielmi, N. J. Electrochem. Soc., 1972, vol. 119, pp. 1009–1012.
Hou, F., Wang, W., and Guo, H., Appl. Surf. Sci., 2006, vol. 252, pp. 3812–3817.
Mulder, W.H. and Sluyters, J.H. Electrochim. Acta, 1988, vol. 33, pp. 303–310.
Rammelt, U. and Reinhard, G. Electrochim. Acta, 1990, vol. 35, pp. 1045–1049.
Ranganatha, S., Venkatesha, T.V., and Vathsala, K., Appl. Surf. Sci., 2010, vol. 256, pp. 7377–7383.
Ahmad, R., Ahmad, Z., Khan, A.U., Mastoi, N.R., et al., J. Environ. Chem. Eng., 2016, vol. 4, pp. 4143–4164.
Shan, A.Y., Ghazi, T.I.M., and Rashid, S.A., Appl. Catal., A, 2010, vol. 389, pp. 1–8.
Houas, A., Lachheb, H., Ksibi, M., Elaloui, E., et al., Appl. Catal., B, 2001, vol. 31, pp. 145–157.
Author information
Authors and Affiliations
Corresponding author
Additional information
Russian Text © F.I. Danilov, A.A. Kityk, D.A. Shaiderov, D.A. Bogdanov, S.A. Korniy, V.S. Protsenko, 2018, published in Elektronnaya Obrabotka Materialov, 2018, No. 3, pp. 21–33.
The article was translated by the authors.
About this article
Cite this article
Danilov, F.I., Kityk, A.A., Shaiderov, D.A. et al. Electrodeposition of Ni–TiO2 Composite Coatings Using Electrolyte Based on a Deep Eutectic Solvent. Surf. Engin. Appl.Electrochem. 55, 138–149 (2019). https://doi.org/10.3103/S106837551902008X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S106837551902008X