Abstract
Immune defense often differs between sexes, where females generally exhibit higher individual immunity than males. Such sexually dimorphic immune defense stems from differential investment into reproduction as females allocate more resources into mating. Contrary to prevailing predictions, some studies have found that females exhibit a weaker immune response than males. In this study, I examined both humoral (phenoloxidase activity and lytic activity) and cellular (encapsulation ability) immune parameters including cuticular melanization in a field population of a bush-cricket (Isophya reticulata) that is an endemic species in the northeastern part of Turkey. As expected, females showed a higher phenoloxidase activity than males, whereas there were no sex differences in lytic activity. Males expressed a higher encapsulation response than females. These results indicated that sex differences in immunity may be variable and females may not always exhibit higher immunity than males. Another interesting finding of this study was a positive relationship between stridulatory file length, phenoloxidase activity, and wing melanization. This finding demonstrated a connection between size differences in stridulatory apparatus that affect sexual signaling and immune response.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Compared with jawed vertebrates, immune responses of insects have only some degree of specificity and immunological memory (Cooper and Eleftherianos 2017) and are primarily based on both humoral and cellular immune responses (Gillespie et al. 1997). Males and females express clear variations in several immune traits regarding both humoral and cellular responses (Kurtz and Sauer 2001; Gershman 2008). Such variation often includes differences in cuticular melanization, a process reliant on a pleiotropic melanin-producing enzyme cascade (True 2003; Cerenius and Soderhall 2004; Kutch et al. 2014). In some insect species, males with darker cuticle had a higher implant darkness (encapsulation), indicating a better immune response as cuticle darkness may be an indicator of male quality in terms of sexual selection (Rantala et al. 2000). Many species choose their potential mates based on traits associated with a high fecundity (Bonduriansky 2001). Hence, the most common mating preferences are fecundity indicators such as body size, mobility, mating signals or cuticular melanization that is positively correlated with immune function (Rantala et al. 2000; Bonduriansky 2001; Ahtiainen et al. 2004; Tregenza et al. 2006).
Life-history strategies of males and females can differ in terms of immunocompetence due to trade-offs based on a differential investment of limited resources (Zuk 1990; Kurtz et al. 2000; Stoehr and Kokko 2006). Although males benefit from increased investment in reproduction, females benefit more in longevity and immunity because they have to survive long enough to oviposit eggs (Zuk and McKean 1996; Nunn et al. 2009; Gershman et al. 2010a). Contrary to predominant predictions, some studies have found that females have a weaker immune system than males (Roth et al. 2011; Vincent and Gwynne 2014) and immune traits are plastic, suggesting a widespread sexual dimorphism in immune defense among insects (Rolff 2002).
In some cricket species (Gryllus (Gryllus) bimaculatus De Geer, 1773, Teleogryllus (Brachyteleogryllus) commodus (Walker, 1869), T. (Teleogryllus) oceanicus (Le Guillo, 1841)), females can use calling song as a cue to choose males with a high immunocompetence (Rantala and Kortet 2003; Simmons et al. 2005; Tregenza et al. 2006). Similarly, male Isophya reticulata Ramme, 1951 produce a calling song to attract females (Sevgili 2004) by rubbing their wings together, using specialized structures in their forewings (stridulatory file, scraper, and mirror) as in other katydids (Montealegre-Z 2009; Montealegre-Z et al. 2017). Stridulatory file length is a predictor of some characteristics of the calling song (e.g., carrier frequency) (Montealegre-Z 2009). In many genera of bush-crickets (including the genus Isophya Brunner von Wattenwyl, 1878), females answer male calls and males seek calling females to mate (Sevgili et al. 2012). Producing a calling song is known to be energetically costly (Kavanagh 1987). This cost might lead to a sex role reversal (McCartney et al. 2012) and a difference in immune response between males and females (Rantala and Kortet 2003; Simmons et al. 2005) due to a cost of secondary sexual traits which are hypothesized to compromise other costly functions such as immune response (Kotiaho 2001; Faivre et al. 2003).
Male bush-crickets may devote a large proportion of their resources into reproduction as they transfer edible spermatophore during mating, which is consumed by females after mating (Sevgili et al. 2015). If orthopteran spermatophore and its components are often considered a costly nuptial gift, it should be regarded as a trade-off between efforts devoted to immune response among sexes (Gershman et al. 2010b). Moreover, as immunological differences between males and females have a genetic basis, reproductive investment or variation in other unmeasured sex traits may mediate these differences (Gershman et al. 2010b).
Regarding immune system, prophenoloxidase (PO) activating system has important roles in invertebrate immunity and PO is a key enzyme in melanization cascade that is a common response to pathogens and parasitoids (Cerenius and Soderhall 2004). Lytic activity (LY) is another important humoral immune defense in insects and it is activated against gram-positive bacteria (Schneider 1985; Adamo 2004). Encapsulation is an effective cellular immune response in invertebrates that multiple hemocytes binding to protozoans, nematodes and other larger multicellular invaders (Strand 2008). Therefore, I predict that females should invest more in immunity than males because many insect species display female-biased sexual dimorphism in immunity response (Kurtz et al. 2000; but see Barthel et al. 2015).
To address knowledge gap in immunity differences in insects, I examined sex-based differences in terms of both humoral (total PO and LY) and cellular immune responses (encapsulation ability) including cuticular melanization between males and females at a breeding season in a field population of I. reticulata (Orthoptera: Phaneropterinae). This study also aims to understand whether stridulatory file length predicts male immunocompetence in I. reticulata.
Material and methods
Isophya reticulata is an endemic species in eastern Black Sea Region of Turkey inhabiting high altitudes of Canik and Giresun Mountains (Giresun and Ordu provinces) (Sevgili 2004; unpublished data, HS). Sexes are morphologically dimorphic; where males have a sound producing organ located under their front wings and females have smaller and reduced wings (Fig. 1a–c). For this study, 115 adult bush-crickets were collected (55 females and 60 males) during a breeding season in a single location around Çambaşı Plateau, Ordu, Turkey on July 3rd, 2014). All bush-crickets on Urtica and Rhododendron species were randomly captured using an insect net. During collection, I observed that males made calling songs and some females had spermatophore. Hence, age and mating status of the individuals were neglected in this study. Individuals were kept for one day in same size cages (27 × 34 × 38 cm) and separated by sex. Each cage contained ten individuals to avoid overcrowding. Different dietary treatments can affect to immune traits (Vincent and Gwynne 2014). Therefore, fresh apple pieces, lettuce, cucumber in addition to leaves of Urtica species were provided ad libitum, and cages were regularly misted. The laboratory was maintained between 24 and 26 °C with a 13 h:11 h light-dark photoperiod.
One day after their collection from the field, bush-crickets were randomly selected from the cages for immunological assessments, and I employed three different immunological measures, namely, PO, LY assay, and encapsulation. To collect hemolymph, bush-crickets were anesthetized for 8 s with CO2 (long enough to anesthetize bush-crickets) and 5 μL of hemolymph were removed from each bush-cricket using a microsyringe and needles (32 gauge, Hamilton Co., NV, USA) inserted between the 2nd and 3rd lateral pleural sclerite of the abdomen. The hemolymph was then dispensed into a chilled 1.5 mL microcentrifuge tube containing 17 μL of phosphate-buffered saline (PBS) and it was immediately frozen at −18 °C. However, due to clogging of needle holes, I avoided the anesthesia of some bush-crickets and therefore, hemolymph samples from 45 out of 55 females and 58 out of 60 males were used in this analysis.
Each hemolymph sample was used to estimate an individual’s PO and LY activity. To measure PO activity, 5 μL of PBS-haemolymph sample was placed onto a 96-well plate. Then, 14 μL of bovine pancreas alpha-chymotrypsin (1.3 mg mL−1; Sigma-Aldrich, MO, USA) was added using a multichannel pipette and incubated in a plate at 24 ± 1 °C in dark for 20 min. After incubation, 90 μL of 7 mM L-DOPA (Sigma-Aldrich, MO, USA) was added and the samples were placed immediately into a micro-plate reader (Multiscan™ FC Microplate Photometer, Thermo-Scientific, MA, USA). Enzyme activity was measured as optical density (OD) at 490 nm every 20 min for a total of 60 min. To estimate lytic activity, 14 μL of the PBS-haemolymph sample was placed onto a 96-well plate. I then added 90 μL of Micrococcus lysodeikticus solution (0.003 g 1 mL−1 PBS), placed the plate into a microplate reader and measured changes in OD at 450 nm every 20 min for a total of 60 min. On each plate, I included PO and LY reagent controls, which consisted of the reagents minus the hemolymph. Both PO and LY activities were estimated as the difference between the final and initial OD. To avoid any possible confusion in the interpretation of the results, lytic activity (ΔA450) was rendered positive by taking the absolute value of estimates. These procedures are similar to those used in other studies (Fedorka et al. 2004, 2013; Fedorka and Sevgili 2014) and they were tested for two bush-crickets by Sevgili (2016).
Immediately after hemolymph removal, bush-crickets were implanted with a single sterile nylon monofilament fishing line (3 mm in length and 0.25 mm in diameter). The monofilament was inserted until it was completely inside the bush-cricket’s abdominal cavity. After 36 h, implants were dissected out of the bush-crickets (after being killed with CO2) and they were photographed using Dino-Lite (Pro AM411 1 T 1.3 MP Digital Microscope). All photos were captured in a closed area where the same amount of light is provided. Encapsulation response was measured as the degree of darkness in each monofilament using an image analysis software (National Institutes of Health, http://rsb.info.nih.gov/nih-image). Both wings were removed in males, and again using Dino-Lite. Wings of males with stridulatory file and hind femur of both sexes were photographed. The degree of cuticular darkness of wings and hind femurs were measured as the mean greyscale value of pixel darkness (16 bit). Hind femur length of both sexes and length of the stridulatory file in males were measured using ImageJ software. When analyzing the implants and cuticle using ImageJ, raw measurement was completed by subtracting each ImageJ measurement from the value of 255. This step allows that melanization scores range from 0 to 255 (0: completely white and 255: completely dark).
LY and PO activity values of some bush-crickets were close to zero and therefore, removed from the analysis (remaining number of samples in PO: N = 42 females, 51 males; in LY: N = 32 females, 42 males). Previously placed implants in one female and three males could not be found during dissection. Therefore, encapsulation data for these specimens were missing (N = 44 females, 55 males). Statistical analyses were performed using R software 2.15.1 (R Development Core Team 2012). Normality of the data was evaluated with Shapiro-Wilk’s test. LY values were log transformed to meet statistical assumptions. PO activity could not be transformed and therefore, non-parametric Wilcoxon rank sum test was used to compare the PO differences between male and female individuals. To differentiate the factors those may affect some immune parameters, a linear mixed effect model (library Ime4) was used. In this analysis, individuals were considered as a random factor (to account for repeated measurements over time) and body size, sex, and femoral darkness (cuticular melanization) were considered as fixed factors. Moreover, body size (right and left hind femora), wing melanization (left and right tegmina) and cuticular melanization (darkness of both femora) were combined into a single synthetic factor via principal component analysis (PCA) (Fedorka et al. 2013). The first principal components were conducted separate PCAs for the body size, wing melanization, and melanization (the first principal component “PC1” explained 97%, 76%, and 85% of the variations, respectively).
Results
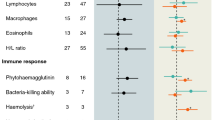
Females were found to be significantly larger than males (Welch Two Sample t-test, t = 7.91, df = 96.97, p < 0.01). There were no significant correlations between LY activity, PO activity, and encapsulation response (all p > 0.05; after controlling for variation between 96-well plates). Females had stronger total PO activity than males (W = 1583.5, p < 0.001, Fig. 2a, Table 1). Although LY activity did not differ between sexes (Fig. 2b, Table 1), encapsulation scores and overall cuticular melanization values in males were significantly greater than in females according to generalized linear mixed models (GLMs) (Fig. 2c–d, Table 1). LY activity (log-transformed) was positively correlated with cuticular melanization only in males (Male, r = 0.46, slope = 0.031, p = 0.004; Female, r = 0.11, slope = 0.006, p = 0.550, Fig. 3). Body size had no significant effect on immune parameters (i.e., PO, LY, and encapsulation) (Table 1). There was no significant relationship between femur length and femur melanization in males (r = 0.16, p = 0.260), but bigger females were much darker than smaller ones (r = −0.36, p = 0.019). Moreover, I found that larger males exhibited shorter stridulatory file (Table 1). Although the correlation between the stridulatory file length of male and PO was weak, it was strongly positive between stridulatory file length and wing melanization (Fig. 4a–b, Table 1).
Sexual dimorphism in total phenoloxidase (PO) (a), lytic activity (LY, change in the absorbance values) (b), encapsulation (c), darkness value of the implant) and cuticular melanization (PCA scores) (d) in Isophya reticulata (F = female, M = male). There was no difference in LY between sexs, but females had greater PO activity than males. Females had lower encapsulation and lighter cuticle than males. Circles represent mean and associated standard error. Asterisks denote statistical significance (***p ≤ 0.005); n. s., not significant
Discussion
In the present study, although the immunity response of females was consistently greater than that of males in PO activity, encapsulation ability of males was greater than that of females. Results of this study indicate that immunity parameters differ between sexes, across individuals within a population of bush-cricket. This finding suggests that sex differences in immunity response are often observed in populations, but it does not explain the prevailing idea of females exhibiting greater levels of immune response than males (Zuk and McKean 1996; Adamo et al. 2001; Simmons et al. 2005; Vincent and Gwynne 2014). In this study, field-collected insects were used, suggesting that sex-based immunological differences naturally occur. The sample of males was randomly selected based on the calling and breeding population. Therefore, it is possible that individuals varying in age, exposure to infections, or nutritional status were included although they were collected from a same small area (suggesting that pathogenicity and/or nutritional status might be similar across individuals).
Differences in environmental variables and ecological parameters are known to influence immune responses in many animals (Zuk et al. 2004; Boughton et al. 2011). On the other hand, costs of courtship and mating on immune traits have been known (Rolff and Siva-Jothy 2002; Gershman 2008). For example, it has been reported that only female’s immunity was affected by the number of matings (Gershman 2008). Similarly, in male drumming wolf spiders, increased display signals used to attract females were correlated with encapsulation rates and lytic activities compared to males under control treatment (Ahtiainen et al. 2005). Such costs for the bush-crickets in their natural environment were not known in this experiment due to their age and mating status unknown inaccuracies. Although there are limitations regarding the unknown life history of the bush-crickets in field populations, findings of this study represent natural variation in immune response.
Although females of I. reticulata had significantly higher PO activity, males and females did not differ in LY activity. Similar results have also been found in Glossina palpalis palpalis (Robineau-Desvoidy, 1830) (Nigam et al. 1997), Gryllus (Gryllus) texensis Cade and Otte, 2000 (Adamo et al. 2001), Gryllus (Gryllus) vocalis Scudder, 1901 (Gershman 2008), Gryllodes sigillatus (Walker, 1869) (Gershman et al. 2010a), and Culex pipiens Linnaeus, 1758 (Cornet et al. 2013). In all of these examples, including the present study, females exhibited higher PO activity than males. Male and female PO activity may vary due to several reasons. For instance, cuticles of males were darker than females in this study. Thus, selection for cuticle color can be responsible for PO differences between sexes. Although PO and melanization have been hypothesized to be correlated due to their shared biochemical cascade, LY and melanization are not expected to be physiologically correlated. However, I found that there was a positive relationship between cuticular melanization and LY in this study.
Regarding other insect species, melanic lines of Spodoptera littoralis (Boisduval, 1833) (Lepidoptera) express lower PO activity but higher LY activity than nonmelanic lines (Cotter et al. 2008). Allonemobius socius (Scudder, 1877) males exhibit darker cuticula and a stronger LY activity than females (Fedorka et al. 2013). In this study, although I. reticulata males exhibited a positive correlation between cuticular melanization and LY activity, females did not show any correlation between these traits. Furthermore, results of this study indicate no other relationships between immune parameters in I. reticulata, suggesting that immunity is not a single trait and its complex nature can lead to trade-offs between both humoral and cellular components of immunity (Rantala and Roff 2005). Additionally, LY activity which shows large amounts of variation among individuals of insect groups is thought to be predominantly environmental in origin and not heritable (Kurtz et al. 2000; Rantala and Roff 2006; Gershman et al. 2010b). Therefore, the reason for the lack of difference in LY activity between males and females may be due to specimens being collected from the same place. However, we found significant differences in LY activity between the males in lowland population of Poecilimon (Poecilimon) similis similis Retowski, 1889 (Tokgöz and Sevgili, unpublished data).
Gershman et al. (2010b) found that PO activity and encapsulation response were significantly heritable in G. sigillatus. They also found that encapsulation response is positively and genetically correlated with body size. Moreover, a larger amount of circulating hemolymph in larger crickets may allow more hemocytes to encapsulate the implant (Gershman et al. 2010b). However, in this study, although body size had no correlation with encapsulation response, sexual dimorphism indicated an effect on encapsulation ability where males showed stronger encapsulation response but lower PO than females. A similar result has been found in Mormon crickets (Anabrus simplex Haldeman, 1852) (Srygley 2012), suggesting the presence of a sex-specific trade-off between these two immune responses (LY and PO activities).
A positive relationship between wing darkness and stridulatory file length was also found in this study. Relationships between sexual signals and immune function support the immunocompetence handicap hypothesis (Ryder and Siva-Jothy 2000; Simmons et al. 2005). According to this hypothesis, correlations between sexually selected component of the calling song and some immune traits are observed. In this study, PO activity can be positively correlated with male stridulatory file length, indicating that some calling song parameters may serve as indicators of some immune functions, which are important for selective females. For example, the courtship song characters preferred by females were positively correlated with male encapsulation rate in G. bimaculatus (Rantala and Kortet 2003).
In conclusion, this is the first study demonstrating a sex-based difference in both humoral and cellular immune response in a Phaneropterinae bush-cricket species (I. reticulata). In the future, more detailed and accurate studies should be carried out to compare wing melanization, calling song parameters, the morphology of stridulatory apparatus, and immune parameters in bush-crickets.
References
Adamo SA (2004) Estimating disease resistance in insects: phenoloxidase and lysozyme-like activity and disease resistance in the cricket Gryllus texensis. J Insect Physiol 50:209–216. https://doi.org/10.1016/j.jinsphys.2003.11.011
Adamo SA, Jensen M, Younger M (2001) Changes in lifetime immunocompetence in male and female Gryllus texensis (formerly G. integer): trade-offs between immunity and reproduction. Anim Behav 62:417–425. https://doi.org/10.1006/anbe.2001.1786
Ahtiainen JJ, Alatalo RV, Kortet R, Rantala MJ (2004) Sexual advertisement and immune function in an arachnid species (Lycosidae). Behav Ecol 15:602–606. https://doi.org/10.1093/beheco/arh062
Ahtiainen JJ, Alatalo RV, Kortet R, Rantala MJ (2005) A trade-off between sexual signalling and immune function in a natural population of the drumming wolf spider Hygrolycosa rubrofasciata. J Evol Biol 18:985–991. https://doi.org/10.1111/j.1420-9101.2005.00907.x
Barthel A, Staudacher H, Schmaltz A, Heckel DG, Groot AT (2015) Sex-specific consequences of an induced immune response on reproduction in a moth. BMC Evol Biol 15:282. https://doi.org/10.1186/s12862-015-0562-3
Bonduriansky R (2001) The evolution of male mate choice in insects: a synthesis of ideas and evidence. Biol Rev 76:305–339. https://doi.org/10.1017/S1464793101005693
Boughton RK, Joop G, Armitage SAO (2011) Outdoor immunology: methodological considerations for ecologists. Funct Ecol 25:81–100. https://doi.org/10.1111/j.1365-2435.2010.01817.x
Cerenius L, Soderhall K (2004) The prophenoloxidase-activating system in invertebrates. Immunol Rev 198:116–126. https://doi.org/10.1111/j.0105-2896.2004.00116.x
Cooper D, Eleftherianos I (2017) Memory and specificity in the insect immune system: current perspectives and future challenges. Front Immunol 8:539. https://doi.org/10.3389/fimmu.2017.00539
Cornet S, Gandon S, Rivero A (2013) Patterns of phenoloxidase activity in insecticide resistant and susceptible mosquitoes differ between laboratory-selected and wild-caught individuals. Parasit Vectors 6:315. https://doi.org/10.1186/1756-3305-6-315
Cotter SC, Myatt JP, Benskin CMH, Wilson K (2008) Selection for cuticular melanism reveals immune function and life-history trade-offs in Spodoptera littoralis. J Evol Biol 21:1744–1754. https://doi.org/10.1111/j.1420-9101.2008.01587.x
Faivre B, Gregoire A, Preault M, Cezilly F, Sorci G (2003) Immune activation rapidly mirrored in a secondary sexual trait. Science 300:103–103. https://doi.org/10.1126/science.1081802
Fedorka KM, Sevgili H (2014) The influence of nuptial feeding and sperm transfer on the immunological cost of reproduction in the ground cricket Al lonemobius socius. Physiol Entomol 39:89–93. https://doi.org/10.1111/phen.12051
Fedorka KM, Zuk M, Mousseau TA (2004) Immune suppression and the cost of reproduction in the ground cricket, Allonemobius socius. Evolution 58:2478–2485. https://doi.org/10.1554/04-399
Fedorka KM, Copeland EK, Winterhalter WE (2013) Seasonality influences cuticle melanization and immune defense in a cricket: support for a temperature-dependent immune investment hypothesis in insects. J Exp Biol 216:4005–4010. https://doi.org/10.1242/jeb.091538
Gershman SN (2008) Sex-specific differences in immunological costs of multiple mating in Gryllus vocalis field crickets. Behav Ecol 19:810–815. https://doi.org/10.1093/beheco/arn040
Gershman SN, Barnett CA, Pettinger AM, Weddle CB, Hunt J, Sakaluk SK (2010a) Inbred decorated crickets exhibit higher measures of macroparasitic immunity than outbred individuals. Heredity 105:282–289. https://doi.org/10.1038/hdy.2010.1
Gershman SN, Barnett CA, Pettinger AM, Weddle CB, Hunt J, Sakaluk SK (2010b) Give 'til it hurts: trade-offs between immunity and male reproductive effort in the decorated cricket, Gryllodes sigillatus. J Evol Biol 23:829–839. https://doi.org/10.1111/j.1420-9101.2010.01951.x
Gillespie JP, Kanost MR, Trenczek T (1997) Biological mediators of insect immunity. Annu Rev Entomol 42:611–643. https://doi.org/10.1146/annurev.ento.42.1.611
Kavanagh MW (1987) The efficiency of sound production in two cricket species, Gryllotalpa australis and Teleogryllus commodus (Orthoptera, Grylloidea). J Exp Biol 130:107–119
Kotiaho JS (2001) Costs of sexual traits: a mismatch between theoretical considerations and empirical evidence. Biol Rev 76:365–376. https://doi.org/10.1017/S1464793101005711
Kurtz J, Sauer KP (2001) Gender differences in phenoloxidase activity of Panorpa vulgaris hemocytes. J Invertebr Pathol 78:53–55. https://doi.org/10.1006/jipa.2001.5040
Kurtz J, Wiesner A, Gotz P, Sauer KP (2000) Gender differences and individual variation in the immune system of the scorpionfly Panorpa vulgaris (Insecta : Mecoptera). Dev Comp Immunol 24:1–12. https://doi.org/10.1016/S0145-305X(99)00057-9
Kutch IC, Sevgili H, Wittman T, Fedorka KM (2014) Thermoregulatory strategy may shape immune investment in Drosophila melanogaster. J Exp Biol 217:3664–3669. https://doi.org/10.1242/jeb.106294
McCartney J, Kokko H, Heller KG, Gwynne DT (2012) The evolution of sex differences in mate searching when females benefit: new theory and a comparative test. Proc R Soc London Ser B 279:1225–1232. https://doi.org/10.1098/rspb.2011.1505
Montealegre-Z F (2009) Scale effects and constraints for sound production in katydids (Orthoptera: Tettigoniidae): correlated evolution between morphology and signal parameters. J Evol Biol 22:355–366. https://doi.org/10.1111/j.1420-9101.2008.01652.x
Montealegre-Z F, Ogden J, Jonsson T, Soulsbury CD (2017) Morphological determinants of signal carrier frequency in katydids (Orthoptera): a comparative analysis using biophysical evidence of wing vibration. J Evol Biol 30:2068–2078. https://doi.org/10.1111/jeb.13179
Nigam Y, Maudlin I, Welburn S, Ratcliffe NA (1997) Detection of phenoloxidase activity in the hemolymph of tsetse flies, refractory and susceptible to infection with Trypanosoma brucei rhodesiense. J Invertebr Pathol 69:279–281. https://doi.org/10.1006/jipa.1996.4652
Nunn CL, Lindenfors P, Pursall ER, Rolff J (2009) On sexual dimorphism in immune function. Philos Trans R Soc Lond Ser B Biol Sci 364:61–69. https://doi.org/10.1098/rstb.2008.0148
Rantala MJ, Kortet R (2003) Courtship song and immune function in the field cricket Gryllus bimaculatus. Biol J Linn Soc Lond 79:503–510. https://doi.org/10.1046/j.1095-8312.2003.00202.x
Rantala MJ, Roff DA (2005) An analysis of trade-offs in immune function, body size and development time in the Mediterranean field cricket, Gryllus bimaculatus. Funct Ecol 19:323–330. https://doi.org/10.1111/j.1365-2435.2005.00979.x
Rantala MJ, Roff DA (2006) Analysis of the importance of genotypic variation, metabolic rate, morphology, sex and development time on immune function in the cricket, Gryllus firmus. J Evol Biol 19:834–843. https://doi.org/10.1111/j.1420-9101.2005.01048.x
Rantala MJ, Koskimaki J, Taskinen J, Tynkkynen K, Suhonen J (2000) Immunocompetence, developmental stability and wingspot size in the damselfly Calopteryx splendens L. Proc R Soc Biol 267:2453–2457. https://doi.org/10.1098/rspb.2000.1305
Rolff J (2002) Bateman's principle and immunity. Proc R Soc London Ser B 269:867–872. https://doi.org/10.1098/rspb.2002.1959
Rolff J, Siva-Jothy MT (2002) Copulation corrupts immunity: a mechanism for a cost of mating in insects. Proc Natl Acad Sci U S A 99:9916–9918. https://doi.org/10.1073/pnas.152271999
Roth O, Scharsack JP, Keller I, Reusch TBH (2011) Bateman's principle and immunity in a sex-role reversed pipefish. J Evol Biol 24:1410–1420. https://doi.org/10.1111/j.1420-9101.2011.02273.x
Ryder JJ, Siva-Jothy MT (2000) Male calling song provides a reliable signal of immune function in a cricket. Proc R Soc London Ser B 267:1171–1175. https://doi.org/10.1098/rspb.2000.1125
Schneider PM (1985) Purification and properties of threee lysozymes from hemolymph of the cricket, Gryllus bimaculatus (Degeer). Insect Biochem 15:463–470. https://doi.org/10.1016/0020-1790(85)90058-7
Sevgili H (2004) A revision of Turkish species of Isophya Brunner von Wattenwyl (Orthoptera: Tettigoniidae: Phaneropterinae). Hacettepe University, Ankara, p 387
Sevgili H (2016) İki çalı çekirgesi üzerinde doğal bağışıklık parametrelerinden fenoloksidaz aktivitesi ile litik aktivitenin ve hemolimfteki protein konsantrasyonunun yöntemsel olarak belirlenmesi. Akademik Ziraat Dergisi 5:51–62
Sevgili H, Demirsoy A, Çıplak B (2012) Description and bioacoustics of a new species of the genus Isophya (Orthoptera: Tetigoniidae: Phaneropterinae) from Turkey. Zootaxa 3361:33–44. https://doi.org/10.5281/zenodo.209689
Sevgili H, Onal H, Yigit A (2015) Mating behavior and spermatophore characteristics in two closely related bushcricket species of the genus Phonochorion (Orthoptera: Phaneropterinae). J Insect Behav 28:369–386. https://doi.org/10.1007/s10905-015-9509-1
Simmons LW, Zuk M, Rotenberry JT (2005) Immune function reflected in calling song characteristics in a natural population of the cricket Teleogryllus commodus. Anim Behav 69:1235–1241. https://doi.org/10.1016/j.anbehav.2004.09.011
Srygley RB (2012) Ontogenetic changes in immunity and susceptibility to fungal infection in Mormon crickets Anabrus simplex. J Insect Physiol 58:342–347. https://doi.org/10.1016/j.jinsphys.2011.12.005
Stoehr AM, Kokko H (2006) Sexual dimorphism in immunocompetence: what does life-history theory predict? Behav Ecol 17:751–756. https://doi.org/10.1093/beheco/ark018
Strand MR (2008) The insect cellular immune response. Insect Sci 15:1–14. https://doi.org/10.1111/j.1744-7917.2008.00183.x
Team RC (2012) R: a language and environment for statistical computing. R Foundation Statistical Computing, Vienna, Austria
Tregenza T, Simmons LW, Wedell N, Zuk M (2006) Female preference for male courtship song and its role as a signal of immune function and condition. Anim Behav 72:809–818. https://doi.org/10.1016/j.anbehav.2006.01.019
True JR (2003) Insect melanism: the molecules matter. Trends Ecol Evol 18:640–647. https://doi.org/10.1016/j.tree.2003.09.006
Vincent CM, Gwynne DT (2014) Sex-biased immunity is driven by relative differences in reproductive investment. Proc R Soc B 281:20140333. https://doi.org/10.1098/rspb.2014.0333
Zuk M (1990) Reproductive strategies and disease susceptibility: an evolutionary viewpoint. Parasitol Today 6:231–233. https://doi.org/10.1016/0169-4758(90)90202-F
Zuk M, McKean KA (1996) Sex differences in parasite infections: patterns and processes. Int J Parasitol 26:1009–1023. https://doi.org/10.1016/S0020-7519(96)80001-4
Zuk M, Simmons LW, Rotenberry JT, Stoehr AM (2004) Sex differences in immunity in two species of field crickets. Can J Zool 82:627–634. https://doi.org/10.1139/Z04-032
Acknowledgments
I would like to thank Nilgün Tokgöz for her assistance in the laboratory work. The author is also grateful to Kenneth M. Fedorka and Ian Kutch for their comments that improved the quality of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares he has no conflict of interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sevgili, H. Sex differences in immunity in a natural population of bush-cricket (Orthoptera: Phaneropterinae). Biologia 74, 501–508 (2019). https://doi.org/10.2478/s11756-019-00193-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11756-019-00193-3