Abstract
In this study, two sites with different nutritional levels at Erhai Lake were selected to perform an epilithic succession experiment in order to better understand the successional dynamic process and associated controlling factors, providing important insights into the role of epilithon in ecological functions and facilitating eutrophic lake recovery planning. The environmental parameters, epilithic biomass and epilithic diversity were qualitatively and quantitatively assessed. Our results showed that faster biomasses peaks (40–50 days colonization) were achieved at Gusheng where nutrient level was higher, comparing with that of Xiahe. A 10-day incubation period is a dividing line from an initial successional stage to the following stage during epilithon succession. The epilithic biomass and biodiversity exhibited distinctly spatial-temporal characteristics, showing higher values at the site being affected by human activities than that being under the natural condition. The spatial characteristic was associated with nitrate enrichment (the most important), and the positively biological responses of the epilithon (higher biomass and diversity) to nitrate enrichment highlight the occurrence of non-point-source pollution on the west bank of Erhai Lake. The temporal characteristic was associated with water temperature (the second important), exhibiting a high biomass accumulation in winter compared with that in summer. This may be caused by other biotic factors not measured in this study, therefore, a detailed investigation regarding the interactions among biotic components in Erhai Lake is essentially needed.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Epilithon, a kind of rock-attached algae, plays an important role in the functioning of shallow aquatic ecosystems via its impacts on primary production, nutritional cycling and food web (Sand-Jensen and Borum 1991; APHA 1998; McCollum et al. 1998; Vadeboncoeur et al. 2003; Gaiser et al. 2006). Detailed studies on the dynamics of the epilithic community will provide important insights into the role of epilithon in the functioning of ecosystems for biomonitoring, ecological status assessments and eutrophic lake recovery planning (Jöbgen et al. 2004; Roubeix et al. 2011; DeNicola and Kelly 2014; Yang et al. 2016). Epilithic dynamics can be assessed by studying succession, i.e., the process of species replacements. Succession reflects a series of interactions among habitat characteristics, allogenic and autogenic factors, and species compositions (Borduqui and Ferragut 2012).
In this study, we selected Erhai Lake to conduct the succession of epilithon. Erhai Lake is the second largest freshwater lake in Yunnan Province, southwestern China, and it has reached a meso-eutrophication level ten years ago (Peng et al. 2005). Moreover, the local water quality has deteriorated to a critical state because of increases in non-point-source pollution introduced by human activities (Zheng et al. 2004; Ni et al. 2016). This deterioration has threatened the biodiversity in the littoral zone of Erhai Lake, for instance, it increased the number of monotonous biotic communities and reduced the biotic community types (Hu et al. 2005; Tang et al. 2013). Thus, ecological assessments of the littoral ecosystem disturbed by human disturbance and a series of corresponding measures of ecological restoration must be implemented for Erhai Lake immediately. Epilithon, as one of the most important biotic components in the littoral zone of Erhai Lake, is critical to perform its function in littoral ecosystem, showing a great potential for use in bioassessment and ecological restoration. As the drastic vegetation degradation of submerged plants of Erhai Lake in recent decades (Wang et al. 2015), epilithon plays an increasingly important role in the littoral ecosystem to sustain primary production and ecosystem stability of Erhai Lake. So far, only a few studies focus on the ecology of epilithon communities in Erhai Lake (Pei et al. 2008; Lan et al. 2011; Yang et al. 2013), including spatial distributions of epilithon, and similarity among different substrata. However, there is still much ecological knowledge unknown at present, for instance, the characteristics of epilithon succession in Erhai Lake. The ecological knowledge gap may lead to an unfavorable situation in which little information is available on the functional attributes of the epilithon in Erhai Lake ecosystem, limiting the applications of epilithon on bioassessment and ecological restoration.
This study aim to describe the ecological characteristics of epilithon community by virtue of studying epilithon succession process on artificial substrata at two sites of Erhai Lake subjected to different pollution levels. It will be helpful to identify the relationships between epilithon succession and its controlling factors, and ultimately increase our understanding of the functional attributes of the epilithon in this lake.
Methods
Study site
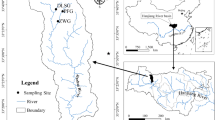
Erhai Lake (i.e., our study area) is a large freshwater body situated in Dali City of Yunnan Province of southwestern China (Fig. 1a). According to the meteorological records from Dali station, the mean annual temperature is 15.1 °C, the mean annual precipitation is 1060 mm, with more than 85% falling between May and October in the catchment (Shen et al. 2005). Erhai Lake has a length of 42 km from north to south and a maximum width of 8.4 km from east to west, and it covers an area of ~250 km2. The average depth is 10.5 m, and the maximum depth is 20.9 m (Peng et al. 2005). Erhai Lake is mainly fed by three rivers (i.e., Miju, Luoshi, and Boluo) and eighteen creeks, and it drains southwards via the natural Xi’er River. To the west of Erhai Lake, a large number of local residents are settled along the foot of the Cangshan Mountains, and a vast expanse of farmland is situated between the residential belt and Erhai Lake. Sewage and agricultural wastewater is discharged into Erhai Lake, leading to algal blooms (Yan et al. 2005; Ni et al. 2016). On the eastern side of Erhai Lake is a sparsely populated shore with a few towns, thus limited disturbances occur along most parts of the eastern shore. Two sites at Erhai aquatic-terrestrial ecotone in the west and the east are selected to perform the epilithon succession, respectively. The west one is Gusheng site (25°48″35.6‴N, 100°08″39.5‴E) which is identified to be obviously affected by human activities, and the east one is Xiahe site (25°39″25.4‴N, 100°17″09.1‴E) which is rarely affected by human activities (Fig. 1b). Both sites recorded a water depth of ~2.5 m and found widely distributed submersed macrophytes (e.g., Potamogeton maackianus, Ceratophyllum sp. and Vallisneria sp.).
Sampling methods
The experiments were performed in winter (2009.12–2010.2) and late summer (2010.8–2010.10). Epilithic colonization was conducted on artificial substrata composed of rectangular marbles (8 cm × 5 cm × 1 cm, see Fig. 1c). Thirty marble substrata were vertically immersed in water at a depth of ~0.5 m at Gusheng and Xiahe sites. Each sample was consisted of the epilithon which was randomly collected from a marble substratum using a razor blade and a toothbrush at each site every 10 days after the succession began. And four parallel epilithic samples at each site were conducted every sampling time. In addition, water samples were collected to determine the environmental parameters. Subsequently, all epilithic and water samples were stored in insulation can with ice packs and transported to the laboratory for further analysis. Each epilithic sample was divided into three parts. The first part was used to determine the chlorophyll a (Chl a) concentration, the second part was used to determine the dry weight (DW) and the ash-free dry weight (AFDW), and the third part was preserved in 4% formalin and used for epilithic enumeration.
Analytical procedures
Physical and chemical parameters of lake water were measured at each sample day. Water temperature (WT), pH and dissolved oxygen (DO) were measured in situ at a water depth of ~0.5 m. Water transparency (SD) were measured using a black and white quadrant Secchi disk of 20-cm diameter in situ. The total nitrogen (TN), total phosphorus (TP), permanganate index (CODMn), ammonia nitrogen (NH4+-N), nitrate (NO3−-N), and soluble reactive phosphate (SRP) concentrations were determined according to standard methods used to examine water and wastewater (APHA 1998).
The comprehensive trophic state index (TSI) provides insights into the nutritional status of lake water, specifically, when 30<TSI ≤ 50, the water quality is still good, indicating a mesotrophic state; and when 50<TSI ≤ 60, the water quality is polluted, indicating a eutrophic state (Li et al. 2016). The TSI was calculated based on the weighted sum of the TSIj, which was calculated using five indicators (Cj), including Chl awater, TN, TP, CODMn, and SD. More details can be found in Huang et al. (2016).
Chl a was extracted using 90% acetone for 12 h after filtering through Whatman GF/F filters. Then, the Chl a concentration was calculated according to the absorbance at 630, 647, 664 and 750 nm (Arar 1997). The DW was obtained after the epilithic sample was oven-dried at 105 °C for at least 24 h. For the AFDW analysis, the aforementioned oven-dried sample was combusted at 550 °C for 4 h to obtain the ash weight (AW), and the AFDW was calculated by subtracting the AW from the DW (APHA 1998). The DW, AW and AFDW were expressed in mg cm−2. The autotrophic index (AI) was expressed as the ratio of the AFDW to the Chl a (i.e., AFDW/Chl a), reflecting the relative importance of autotrophs and heterotrophs in the community (APHA 1998).
Epilithic enumeration of each sample was performed with a microscope at 400-fold and 1000-fold magnification. Morphotypes were identified by referencing Krammer and Lange-Bertalot (1986-1991), Zhu and Chen (2000) and Hu and Wei (2006). The epilithic cell size was estimated using an ocular micrometer. The Shannon-Wiener index (H′), the Margalef index (D), the Simpson diversity index (P), and the Pielou evenness index (J) were employed to depict variations in species biodiversity (e.g., H′ and D) and evenness (e.g., P and J) (Pandya et al. 2014).
The temporally and spatially significant differences in the water parameters, biomasses (DW, AFDW, Chl a) and biodiversities (H′, D, P, J) were analyzed by a one-way analysis of variance (ANOVA) in SPSS 19.0, followed by the Student-Neumann-Keuls posthoc (SNK) multiple comparison test to determine significance between groups. A principal component analysis (PCA) and a redundancy analysis (RDA) were conducted using Canoco 5.0 for Windows after species data were square-root transformed to satisfy the assumption of normality and the homogeneity of variance. Only the environmental variables with significant contributions to the axes were chosen for the RDA using forward selection (P < 0.05). The level of variance in the biotic data (biomass, diversity and AI) explained by the environmental variables according to the RDA was tested using Monte Carlo simulations with 999 unrestricted permutations.
Results
Water quality
The physicochemical variables at both sampling sites (i.e., Gusheng and Xiahe) during the study period are summarized in Table 1. According to the significance analysis, the WT, SD and SRP exhibited significant temporal differences, with higher values in summer. TN, NO3−-N, and Chl awater exhibited significant spatial differences, with higher values at Gusheng (P < 0.05). AI values at Xiahe were significantly higher than that at Gusheng (P < 0.01), indicating that heterotrophic organisms were more prevalent at Xiahe. The SD, TP, SRP and the comprehensive trophic state index (TSI) exhibited significant spatial-temporal differences (P < 0.01). The TSI value was higher than 50 at the Gusheng in summer, indicating a eutrophic state, whereas the others varied between 40 and 50, indicating a mesotrophic state.
Variations in the epilithic biomass
The temporal and spatial changes in epilithic biomass were assessed using the Chl a, DW and AFDW (Fig. 2). All samples were abbreviated, referring to the annotation of Fig. 2, e.g., the sample colonized for 40 days in winter at Gusheng was abbreviated as WG40d, and so on. At the Gusheng site, although faster colonization was observed in summer, the highest epilithic biomass was achieved in winter. At the Xiahe site, the epilithon biomass tended to increase with colonization time in winter and summer. The biomass at Gusheng was always higher than that at Xiahe. Relative high epilithic biomasses (Chl a, DW and AFDW) were observed in the WG40d and WG60d samples with no significant difference between each other (P > 0.05), however, both samples were significantly higher than the other samples (P < 0.05). Specifically, the WG40d biomasses were as follows: Chl a, 22.5–36.3 μg cm−2; DW, 6.9–9.7 mg cm−2; and AFDW, 1.8–2.6 mg cm−2, respectively, higher than the other biomasses colonized for the same days (i.e., WX40d, SG40d, SX40d). In general, the biomass exhibited significant spatial-temporal variations, i.e., BiomassG > BiomassX in both seasons, BiomassW ≥ BiomassS at both sites.
Variations of epilithic species
A total of 118 epilithic taxa were identified at the two sampling sites during the study period. The epilithic taxa were dominated by Bacillariophyta, Chlorophyta and Cyanophyta, and a few species of Cryptophyta, Pyrrophyta and Euglenophyta (not shown) were also observed (Fig. 3). Single-celled algae (e.g., Placoneis amphibola, Navicula sp2., Stauroneis anceps, Tetraedron sp. and Cosmarium sp.) and non-filamentous multicellular algae (e.g., Fragilaria virescens, Fragilaria lyngbye and Desmodesmus quadricauda) occurred at the beginning of succession. Some filamentous algae, such as Phormidium faveolarum and Aphanizomenon flosaquae, were also observed at the initial stage. Although a large number of cells of the filamentous algae were identified, these organisms are so tiny (~0.5 to ~2 μm) that they only accounted for a small proportion of the biovolume.
To explore the spatial-temporal variations of the epilithic communities, we adopted PCA ordination to visually characterize the epilithic successions (Fig. 4). Based on the results of the PCA, three sample groups were differentiated. The first group was composed of the initial phase samples (i.e., SG10d, SX10d, WG10d, WX10d), which were characterized by the abundant species of Bacillariophyta (e.g., F. lyngbye, P. amphibola, Rhoicosphenia abbreviata and S. anceps), as well as species of Cyanophyta and Chlorophyta (e.g., P. faveolarum, A. flosaquae, D. quadricauda, Cosmarium sp. and Tetraedron sp.). The second group (SX20d, SX30d, SX50d, WX20d, WX40d, WX50d and WX60d) was dominated by Anabaena azotica, Psephonema aenigmaticum and Calothrix sp. The third group was composed of samples colonized for 20–60 days at the Gusheng site in both seasons and was dominated by Pseudanabaena sp., Cyclotella meneghiniana, P. aenigmaticum and Cymbella ventricosa. Species of Bacillariophyta, such as Nitzschia palea, Hantzschia amphioxys, Fragilaria capucina and Navicula sp1., were frequently found in this group.
The principal component analysis (PCA) based on species data (relative abundance >2%). The numbers denote for species, corresponding to the ID number of algae in Supplementary Table S1
The PCA biplot can be used to identify the potential factors influencing the species variations. The first two axes of the PCA accounted for 42.5% of the total species variance, with the first axis (PCA 1) explaining 23.4% and the second axis (PCA 2) explaining 19.1%. Considering that all samples at the initial stage of succession (i.e., 10 d) were situated on the positive side of PCA 1 and the samples of the following successional stages (i.e., 20–60 d) were situated on the negative side of PCA 1, the succession trajectory was distinctly dependent on incubation time (Fig. 4). Thus, the first axis may be associated with the temporal effect of incubation time on the community structure of epilithon. Most samples collected from Xiahe site were located on the positive side of PCA 2, whereas samples collected from Gusheng site were located entirely on the negative side of PCA 2, indicating that PCA 2 may be associated with the effect of spatially related factors on the community structure of epilithic algae.
Variations of epilithic biodiversity
Several indices are employed to evaluate the diversity, richness and equitability of species during succession (Fig. 5). The results showed that the highest Shannon-Wiener diversity index (H′) and Margalef richness index (D) were obtained from SG samples, whereas the lowest aforementioned indices were obtained from SX samples. In general, the epilithon reached the first diversity peak within 30 days in summer, whereas the first diversity peak was reached within 50 days in winter. The Simpson evenness index (P) and Pielou evenness index (J) exhibited higher values at the initial colonization stage, followed by a sharp decline on the 20th day and a slightly increasing trend thereafter.
The biotic variables (biomass, diversity and AI) and environmental parameters (only shown the significant ones with the first two axes, P < 0.01) were subjected to a RDA to determine the relationships among the aforementioned data. According to the RDA results, the first two axes explained 52.0% of the observed variations in biotic data (Fig. 6), with the first axis (RDA 1) explaining 50.6% of the variations and second axis (RDA 2) only explaining 1.4% (Table 2). The biotic data, including the epilithic biomass (Chl a, DW and AFDW) and diversity (H′ and D), were positively correlated with RDA 1. Considering that Gusheng site were situated on the positive side of RDA 1, while Xiahe site were situated on the negative side of RDA 1, thus the first axis reflected the effects of the spatial factor on the biotic data of the epilithon. Moreover, nitrate concentration, as one of the most important environmental variables affecting the epilithon succession, was highly significant correlated with PCA 1 (r = 0.654, P < 0.01; Table 2). It implied that the spatial variability of nitrate was the spatial factor and had a striking function on epilithon succession, resulting in the spatial characteristic of epilithon communities. Similarly, the positive side of RDA 2 mainly contained biotic data in summer, and the negative side of RDA 2 contained biotic data in winter, implying that RDA 2 reflected the seasonal factor. The seasonal factor was associated with variations of WT based on the relationship between WT and RDA 2 (r = 0.415, P < 0.05; Table 2).
Discussion
Features of epilithic succession
Faster biomasses peaks were achieved at Gusheng than that at Xiahe, implying that the period of ecological restoration using epilithon colonized on artificial substrata are needed to be differentiated at different nutrient levels. For instance, 40–50 days colonization is recommended at Gusheng and more than 50 days colonization is needed at Xiahe.
The evenness of the epilithic community was initially high, lower in the intermediate phase and again slightly higher later on, being analogous with the result of others (Szabó et al. 2008). Community composition after 10 days colonization was different from following stages whereas there were only minor changes subsequently. That is, the epilithic community tended to stabilize after 20 days colonization. After 20 days, a sharply decreasing evenness index was observed, because certain successional species accounted for relatively high proportions. From 30 to 60 days, filamentous algae (e.g., Oscillatoria, Lyngbya, Phormidium, Anabaena, A. flosaquae, and P. aenigmat) increased proportionally to higher levels, implying that habitat space or light availability may be limiting resources that forced the algal mat to develop into a three-dimensional stereoscopic structure in following successional stages (Johnson et al. 1997; Jackson 2003). Nevertheless, the dispersed distribution of the samples colonized for 10 days may be associated with the randomly homogenized attachment of epilithic algae; and the samples colonized for 20–60 days were clustered on the negative side of the PCA biplot, suggesting relatively high similarities after 20 days colonization. Thus, a 10-day incubation time is a key criterion for the succession of epilithon and represents a dividing line from the initial to the following successional stage.
E. sorex and Epithemia gibba almost solely occurred at Xiahe site during the succession. These taxa are regarded as common epiphytic species (Zalat and Vildary 2005; Sıvacı et al. 2008; Malkin et al. 2009), usually accompanied by macrophytes in water environments. This situation is basically in accord with the actual distribution of macrophytes which has been found averagely wider around Xiahe site than Gusheng site by field investigations (Hu et al. 2005; Chu et al. 2014).
Factors influencing the biotic data during succession
Among the environmental parameters, the most important one influencing the biotic data (i.e., biomass, diversity and AI) is nitrate concentration, showing a significant spatial variation. The significant spatial nitrate difference may be associated with anthropogenic effects on the west lakeshore (e.g., Gusheng). In details, spring water originated from Cangshan Mountain flows through residential areas and vast areas of farmland, discharges into Erhai Lake ultimately, leading to nutrient accumulations along the west bank of the lake. Thus, Erhai Lake likely suffers from non-point-source pollution, especially in summer. Excluding nitrate, other nutrients were also highly enriched, and the highest average TSI value was also observed at Gusheng site in summer. The higher nutrients and TSI values at Gusheng in summer than in winter further highlighted the seasonal characteristics of non-point-source pollution. It is likely to be associated with high quantities of fertilizer and excrement inputs and the low use efficiency of fertilizer and living waste (Yang and Song 2006). Thus, the high nutritional load (e.g., nitrate) leads to higher biomass at Gusheng than at Xiahe, showing an obvious spatial characteristic. The diversity indices were also positively associated with the concentration of nitrate during the epilithic succession. Reports have indicated that a moderately increased nitrate concentration may improve biodiversity (Peterson and Grimm 1992; Baker et al. 2009; Shylla and Ramanujam 2013), resulting in a higher species diversity in meso-eutrophic lakes comparing with that in oligotrophic or eutrophic lakes (Jeppesen et al. 2000). Therefore, the meso-eutrophic waterbody at Gusheng may contribute to the higher biodiversity relative to that at Xiahe.
Among the environmental parameters, the second most important factor influencing the biotic data was WT, although it only accounted for 1.4% of the variations in the biotic data. The epilithic biomass was weakly negatively correlated with WT in this study, with generally higher biomass in winter than in summer. It should be pointed out that the WT is significantly higher in summer than in winter in our study area (P < 0.01). This negative correlation between epilithic biomass and WT is different from most cases because biomass accrual rates are positively correlated with WT normally (Francoeur et al. 1999). Epilithic biomass is related to many factors, such as nutrient, grazing, allelopathy and so on (Feminella and Hawkins 1995; McCollum et al. 1998; Hillebrand and Kahlert 2001; Winkelmann et al. 2014). In our study (e.g., Gusheng site), the epilithic biomass was supposed to be higher in summer than in winter because both of the WT and nutritional loads are significantly higher in summer than in winter and both factors usually lead to a higher biomass, however, a reverse result was observed. It can be inferred that certain seasonal variables that were not measured in this study, such as the effects of grazing or interactions with the macrophytes, have exerted more influences on epilithic biomass than WT and nutrients (e.g., nitrate). These undetermined factors (such as grazing intensity and interaction effects with macrophytes) should be included in future studies to further explore their roles on epilithon.
Conclusion
In this study, an epilithic successional experiment was performed at two sites with different nutritional levels in Erhai Lake. The result revealed that faster biomasses peaks (40–50 days colonization) were achieved at Gusheng where nutrient level was higher, comparing with that of Xiahe. A 10-day incubation time is a key criterion for the succession of epilithon in Erhai Lake and represents a dividing line from the initial to the following successional stage. The biomass and biodiversity of epilithic community were higher at the site being affected by human activities than that being under the natural condition. Positively biological responses of the epilithon (higher biomass and diversity) to nitrate enrichment highlight the occurrence of non-point-source pollution on the west bank of Erhai Lake. And epilithic biomass responses to water temperature call for a detailed investigation regarding the interactions among biotic components in aquatic ecosystem.
References
APHA (1998) Standard methods for the examination of water and wastewater, 20th edn. American Public Health Association, Washington
Arar EJ (1997) Method 446.0: in vitro determination of chlorophylls a, b, c1+c2 and pheopigments in marine and freshwater algae by visible spectrophotometry. United States Environmental Protection Agency, Office of Research and Development, National Exposure Research Laboratory
Baker MA, de Guzman G, Ostermiller JD (2009) Differences in nitrate uptake among benthic algal assemblages in a mountain stream. J N Am Benthol Soc 28:24–33
Borduqui M, Ferragut C (2012) Factors determining periphytic algae succession in a tropical hypereutrophic reservoir. Hydrobiologia 683:109–122
Chu ZS, Ye BB, Tian GP, Pang Y, Hu XZ (2014) Spatial distribution characteristics and estimation of submerged plant biomass in Lake Erhai. Res Environ Sci 27:1–5 (in Chinese with English abstract)
DeNicola DM, Kelly M (2014) Role of periphyton in ecological assessment of lakes. Freshw Sci 33:619–638
Feminella JW, Hawkins CP (1995) Interactions between stream herbivores and periphyton: a quantitative analysis of past experiments. J N Am Benthol Soc 14:465–509
Francoeur SN, Biggs BJ, Smith RA, Lowe RL (1999) Nutrient limitation of algal biomass accrual in streams: seasonal patterns and a comparison of methods. J N Am Benthol Soc 18:242–260
Gaiser EE, Childers DL, Jones RD, Richards JH, Scinto LJ, Trexler JC (2006) Periphyton responses to eutrophication in the Florida Everglades: cross-system patterns of structural and compositional change. Limnol Oceanogr 51:617–630
Hillebrand H, Kahlert M (2001) Effect of grazing and nutrient supply on periphyton biomass and nutrient stoichiometry in habitats of different productivity. Limnol Oceanogr 46:1881–1898
Hu HJ, Wei YX (2006) Chinese freshwater algae-system, classification and ecology. Science Press, Beijing (in Chinese)
Hu XZ, Jin XC, Du BH, Zhu J (2005) Submerged macrophyte of lake Erhai and its dynamic change. Res Environ Sci 18:1–4 (in Chinese with English abstract)
Huang J, Gao J, Zhang Y (2016) Eutrophication prediction using a markov chain model: application to lakes in the Yangtze River basin, China. Environ Model Assess 21:233–246
Jackson CR (2003) Changes in community properties during microbial succession. Oikos 101:444–448
Jeppesen E, Peder Jensen J, SØndergaard M, Lauridsen T, Landkildehus F (2000) Trophic structure, species richness and biodiversity in Danish lakes: changes along a phosphorus gradient. Freshw Biol 45:201–218
Jöbgen A, Palm A, Melkonian M (2004) Phosphorus removal from eutrophic lakes using periphyton on submerged artificial substrata. Hydrobiologia 528:123–142
Johnson RE, Tuchman NC, Peterson CG (1997) Changes in the vertical microdistribution of diatoms within a developing periphyton mat. J N Am Benthol Soc 16:503–519
Krammer K, Lange-Bertalot H (1986-1991) Bacillariophyceae. Süßwasserflora von Mitteleuropa. 2 (1-4). G. Fischer, Stuttgart
Lan B, Xiang XF, Jia YT, Jiang JH, Yang S (2011) Investigation of epilithic periphyton communities of the littoral zone of the lakes in Erhai watershed. China Environ Sci 31:1881–1887 (in Chinese with English abstract)
Li D, Erickson RA, Tang S, Zhang Y, Niu Z, Liu H, Yu H (2016) Structure and spatial patterns of macrobenthic community in tai Lake, a large shallow lake, China. Ecol Indic 61:179–187
Malkin SY, Sorichetti RJ, Wiklund JA, Hecky RE (2009) Seasonal abundance, community composition, and silica content of diatoms epiphytic on Cladophora glomerata. J Great Lakes Res 35:199–205
McCollum EW, Crowder LB, McCollum SA (1998) Complex interactions of fish, snails, and littoral zone periphyton. Ecology 79:1980–1994
Ni Z, Wang S, Zhang M (2016) Sediment amino acids as indicators of anthropogenic activities and potential environmental risk in Erhai Lake, Southwest China. Sci Total Environ 551:217–227
Pandya U, Maheshwari DK, Saraf M (2014) Assessment of ecological diversity of rhizobacterial communities in vermicompost and analysis of their potential to improve plant growth. Biologia 69:968–976
Pei GF, Hu ZY, Liu GX (2008) Benthic algal communities distribution in the littoral zone of Yunnan Plateau Lakes. J Wuhan Bit Res 26:373–378 (in Chinese with English abstract)
Peng WQ, Wang SY, Liu XB (2005) Assessment on Erhai Lake water quality. J China Inst Water Resour Hydropower Res 3:192–198 (in Chinese with English abstract)
Peterson CG, Grimm NB (1992) Temporal variation in enrichment effects during periphyton succession in a nitrogen-limited desert stream ecosystem. J N Am Benthol Soc 11:20–36
Roubeix V, Mazzella N, Schouler L, Fauvelle V, Morin S, Coste M, Delmasa F, Margoum C (2011) Variations of periphytic diatom sensitivity to the herbicide diuron and relation to species distribution in a contamination gradient: implications for biomonitoring. J Environ Monit 13:1768–1774
Sand-Jensen K, Borum J (1991) Interactions among phytoplankton, periphyton, and macrophytes in temperate freshwaters and estuaries. Aquat Bot 41:137–175
Shen J, Yang L, Yang X, Matsumoto R, Tong G, Zhu Y, Zhang Z, Wang S (2005) Lake sediment records on climate change and human activities since the Holocene in Erhai catchment, Yunnan Province, China. Sci China Ser D Earth Sci 48:353–363
Shylla O, Ramanujam P (2013) Effect of nutrients on diversity of algae in Marngar Lake, Meghalaya (India). Int J Algae 15:239–250
Sıvacı ER, Çankaya E, Kılınç S, Dere Ş (2008) Seasonal assessment of epiphytic diatom distribution and diversity in relation to environmental factors in a karstic lake (Central Turkey). Nova Hedwigia 86:215–230
Szabó KÉ, Makk J, Kiss KT, Eiler A, Ács É, Tóth B, Kiss ÁK, Bertilsson S (2008) Sequential colonization by river periphyton analysed by microscopy and molecular fingerprinting. Freshw Biol 53:1359–1371
Tang JF, Ye SW, Li W, Liu JS, Zhang TL, Guo ZQ, Zhu FY, Li ZJ (2013) Status and historical changes in the fish community in Erhai Lake. Chin J Oceanol Limnol 31:712–723
Vadeboncoeur Y, Jeppesen E, Vander Zanden MJ, Schierup HH, Christoffersen K, Lodge DM (2003) From Greenland to green lakes: cultural eutrophication and the loss of benthic pathways in lakes. Limnol Oceanogr 48:1408–1418
Wang SR, Zhang L, Ni LY, Zhao HC, Jiao LX, Yang SW, Guo LG, Shen JZ (2015) Ecological degeneration of the Erhai Lake and prevention measures. Environ Earth Sci 74:3839–3847
Winkelmann C, Schneider J, Mewes D, Schmidt SI, Worischka S, Hellmann C, Benndorf J (2014) Top-down and bottom-up control of periphyton by benthivorous fish and light supply in two streams. Freshw Biol 59:803–818
Yan CZ, Jin XC, Zhao JZ, Shen B, Li NB, Huang CZ, Xiong ZH (2005) Ecological protection and sustainable utilization of Erhai Lake, Yunnan. Environ Sci 26:38–42 (in Chinese with English abstract)
Yang SH, Song TQ (2006) Problems of pollution derived from agriculture in Erhai Lake district and countermeasures. Res Agr Moderniz 27:428–438 (in Chinese with English abstract)
Yang SY, Cai QH, Tang T, Wang XZ, Li FQ (2013) Spatial distribution of benthic algae in the Erhai basin of southwestern China. Fresenius Environ Bull 22:2731–2739
Yang J, Liu J, Wu C, Kerr PG, Wong PK, Wu Y (2016) Bioremediation of agricultural solid waste leachates with diverse species of cu (II) and cd (II) by periphyton. Bioresour Technol 221:214–221
Zalat A, Vildary SS (2005) Distribution of diatom assemblages and their relationship to environmental variables in the surface sediments of three northern Egyptian lakes. J Paleolimnol 34:159–174
Zheng GQ, Yu XX, Jiang N, He LH (2004) The cause for chang of water quality of Erhai Lake and prediction of water quality. J Northeast Forest Univ 32:99–102 (in Chinese with English abstract)
Zhu HZ, Chen JY (2000) Bacillariophyta of the Xizang plateau. Sciences Press, Beijing (in Chinese)
Acknowledgments
This research was supported by National Water Pollution Control and Management Technology Major Projects (2013ZX07105-005) and Scientific Research Foundation for High-level Talents Introducted by Chongqing Three Gorges University (NO. 17RC08). We wish to thank Yu GL for the identification of epilithic algae, Zhao QiFu and Yin YanZhen. for the sampling help and the anonymous reviewers for providing helpful suggestions.
Author information
Authors and Affiliations
Contributions
B.L. and H.L. are co-first authors with equal contribution to this work. B.L. and H.L. conducted the experiments and analyzed the data. X.X. conducted field work and offered assistance to identify the algae. S.Y. and B.L. designed the project. All authors co-wrote and co-edited the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
ESM 1
(DOCX 62 kb)
Rights and permissions
About this article
Cite this article
Lan, B., Li, H., Xiang, X. et al. Spatial-temporal characteristics of epilithic algae succession on artificial substrata in relation to water quality in Erhai Lake, Yunnan Province, China. Biologia 73, 821–830 (2018). https://doi.org/10.2478/s11756-018-0100-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11756-018-0100-z