Abstract
Two new species of oripodoid mites (Oribatida, Oripodoidea) are described from the Kahurangi National Park in New Zealand. Angullozetes kahurangiensis sp. n. (Scheloribatidae) differs from Angullozetes rostratus Hammer, 1967 by the rounded rostrum and clavate bothridial setae with minute stalk and large, elongated head, and from Angullozetes arilloi Ermilov et al., Ecol Mont 18:75–81, 2018 by the presence of aggenital setae and three pairs of notogastral porose areas. Totobates elatus sp. n. (Haplozetidae) differs from Totobates elegans (Hammer, 1958) and Totobates ovalis Hammer, 1967 by the lanceolate bothridial setae. An identification key to known species of Angullozetes is presented.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
During taxonomic identification of oribatid mites from the Kahurangi National Park in New Zealand, we found two new species of the superfamily Oripodoidea, one belonging to the genus Angullozetes Hammer, 1967 (ScheloribatidaeFootnote 1), the other to Totobates Hammer, 1961 (HaplozetidaeFootnote 2). The main goal of the paper is to describe these new species.
Angullozetes was proposed by Hammer (1967) with Angullozetes rostratus Hammer, 1967 as type species. The genus comprises two species, which are known only from New Zealand (Hammer 1967; Ermilov et al. 2018). The generic diagnosis was revised by Ermilov et al. (2018).
Totobates was proposed by Hammer (1961) with Totobates discifer Hammer, 1961 as type species. The genus comprises 16 species, which are distributed in the Australian, Antarctic and Neotropical regions (Subías 2004, updated 2018). The generic characters were summarized by Hammer (1961). An identification key to species of Totobates was given by Balogh and Balogh (2002).
Material and methods
Material
The detailed collection locality and habitat for each new species are given in the “Material examined” sections.
Methods
Soil cores were collected using a stainless steel corer (5 × 5 cm); the volume collected included the ground vegetation plus 5 cm of the substrate depth. Samples were kept in the refrigerator until delivered to the lab. Mites were extracted into 75% ethanol in modified Berlese extractors for a minimum of 7 days, or longer if the soil was not fully dry.
Specimens were mounted in lactic acid on temporary cavity slides for measurement and illustration. Body length was measured in lateral view, from the tip of the rostrum to the posterior edge of the notogaster. Notogastral width refers to the maximum width of the notogaster behind pteromorphs. Lengths of body setae were measured in lateral aspect. All body measurements are presented in micrometers. Formulas for leg setation are given in parentheses according to the sequence trochanter–femur–genu–tibia–tarsus (famulus included). Formulas for leg solenidia are given in square brackets according to the sequence genu–tibia–tarsus.
Drawings were made with a camera lucida using a Leica transmission light microscope “Leica DM 2500”.
Morphological terminology used in this paper follows that of F. Grandjean: see Travé and Vachon (1975) for references, Norton (1977) for leg setal nomenclature, and Norton and Behan-Pelletier (2009), for overview.
The following abbreviations are used: lam – lamella; plam – prolamella; slam – sublamella; Al – sublamellar porose area; tu – tutorium; ar, lr – anterior and lateral ridges of prodorsum, respectively; ro, le, in, bs, ex – rostral, lamellar, interlamellar, bothridial and exobothridial setae, respectively; bo – bothridium; D – dorsophragma; P – pleurophragma; Ad – dorsosejugal porose area; Am, Ah – humeral porose areas; c, la, lm, lp, h, p – notogastral setae; Aa, A2, A3 – notogastral porose areas; ia, im, ip, ih, ips – notogastral lyrifissures; gla – opisthonotal gland opening; cs – circumgastric scissure; csb – circumgastric sigillar band; a, m, h – subcapitular setae; or – adoral seta, v, l, d, cm, acm, ul, sul, vt, lt – palp setae; ω – palp and leg solenidion; cha, chb – cheliceral setae; Tg – Trägårdh’s organ; Pd I, Pd II – pedotecta I, II, respectively; 1a, 1b, 1c, 2a, 3a, 3b, 4a, 4b – epimeral setae; cus – custodium; dis – discidium; cp – circumpedal carina; g, ag, an, ad – genital, aggenital, anal and adanal setae, respectively; iad – adanal lyrifissure; p.o. – preanal organ; p.a. – leg porose area; σ, φ – leg solenidia; ɛ – leg famulus; v, ev, bv, l, d, ft, tc, it, p, u, a, s, pv – leg setae.
Descriptions of new species
Family Scheloribatidae
GenusAngullozetes Hammer, 1967
Type speciesAngullozetes rostratus Hammer, 1967
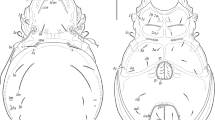
Angullozetes kahurangiensis sp. n. (Figs. 1–12)
Diagnosis. Body size: 365–398 × 166–182. Rostrum rounded. Rostral, lamellar, interlamellar, notogastral, epimeral and anogenital setae short, setiform, smooth. Bothridial setae clavate, barbed, with very small stalk and large, elongate head. Prolamellae and posterior ridges of prodorsum absent. Tutoria with small point tip. Three pairs of rounded porose areas. Aggenital setae present.
Description.Measurements. Body length: 365 (holotype, male), 381, 398 (two paratypes, two males); notogaster width: 166 (holotype, male), 174, 182 (two paratypes, two males).
Integument. Body color light brown to brown. Body surface densely microfoveolate (visible under high magnification).
Prodorsum (Figs. 1, 3). Rostrum rounded. Lamellae half of prodorsum length (measured in lateral view). Prolamellae absent. Sublamellae slightly shorter than lamellae, thin. Tutoria strong, with small pointed tip. Anterior and lateral ridges on the lateral parts of prodorsum distinct, posterior ridges absent. Rostral, lamellar and interlamellar (all 8–12) setae setiform, smooth. Exobothridial setae (2) minute, thin, smooth. Bothridial setae (20; stalks in bothridia not considered) clavate, barbed, with very small stalk and large, elongate head. Dorsosejugal porose areas oval (4 × 2).
Notogaster (Figs. 1, 3–5). Anterior margin of notogaster not developed. Pteromorphs well-developed, triangular, rounded distally. Ten pairs of notogastral setae (4–6) setiform, smooth. Three pairs (Aa, A2, A3) of rounded porose areas (6–10) developed. Lyrifissures, opisthonotal gland openings, circumgastric scissure and circumgastric sigillar band distinct.
Gnathosoma (Figs. 6–8). Subcapitulum longer than wide (82–86 × 57–61). Subcapitular setae setiform, smooth, h (6–8) shorter and thinner than a and m (10–12). Adoral setae (6–8) setiform, barbed. Palps (length 45–49) with setation 0–2–1–3–9(+ω). Postpalpal setae (2) spiniform, smooth. Chelicerae (length 82–86) with two setiform, barbed setae, cha (28–32) longer than chb (18–20). Trägårdh’s organ of chelicerae elongate triangular.
Epimeral and lateral podosomal regions (Figs. 2, 3). Epimeral setae (4–6) setiform, smooth. Humeral porose areas Am present, elongate oval, diffuse, poorly visible, Ah represented by saccules with small opening and conical channel. Pedotecta II trapezoid distally in ventral view. Discidia roundly triangular. Circumpedal carinae long, directed to triangular custodia.
Anogenital region (Figs. 2–5). Three pairs of genital, one pair of aggenital, two pairs of anal and three pairs of adanal setae (4–6) setiform, smooth. Adanal lyrifissures located close and parallel to anal plates. Postanal porose area long, band-like, poorly visible.
Legs (Figs. 9–12). All legs monodactylous, claw of each leg strong, smooth dorsally. Dorsoparaxial porose areas on femora I–IV and on trochanters III, IV slightly developed. Formulas of leg setation and solenidia: I (1–5–2–4–17) [1–2–2], II (1–5–2–4–15) [1–1–2], III (2–3–0–3–15) [1–1–0], IV (1–2–1–3–12) [0–1–0]; homology of setae and solenidia indicated in Table 1.
Material examined. Holotype (male) and two paratypes (two males): New Zealand, South Island, Nelson/Tasman region, Kahurangi National Park, Mount Arthur summit track, high alpine zone, 1521 m a.s.l., 41°12.601’ S, 172°42.099′ E, in soil under lichen, Oreobolus pectinatus (Hook. f.) and Chionochloa australis (Buchanan) Zotov, sample MtA-14, 23 January 2017 (collected by M. Minor).
Type deposition. The holotype (ethanol with a drop of glycerol) and one paratype (ethanol with a drop of glycerol) are deposited in the New Zealand National Arthropod Collection, Auckland, New Zealand. One paratype (ethanol with a drop of glycerol) is deposited in the Tyumen State University Museum of Zoology, Tyumen, Russia.
Etymology. The specific name kahurangiensis refers to the Kahurangi National Park where the species was collected.
Differential diagnosis. Distinctive characters of the new species versus other Angullozetes species can be found in the identification key below.
Key to known species of Angullozetes
-
1.
Aggenital setae absent; with four pairs of notogastral porose areas (A1 present); body size: 225–254 × 131–147 … Angullozetes arilloiErmilov et al., 2018
Aggenital setae present; with three pairs of notogastral porose areas (A1 absent) … 2
-
2.
Rostrum nasiform; bothridial setae with long stalk and small, globose head; body length: 360 … Angullozetes rostratusHammer, 1967
Rostrum not nasiform; bothridial setae with minute stalk and large, elongate head; body size: 365–398 × 166–182 … Angullozetes kahurangiensissp. n.
Family Haplozetidae
Genus Totobates Hammer, 1961
Type species Totobates discifer Hammer, 1961
Totobates elatus sp. n. (Figs. 13–24)
Diagnosis. Body size: 315–348 × 166–190. Rostrum rounded. Rostral, lamellar and interlamellar setae long, setiform, barbed. Notogastral, anal and adanal setae short, setiform, with attenuate and flexible tip, smooth. Epimeral, genital and aggenital setae short, simple. Bothridial setae lanceolate, barbed, with long stalk and elongate head. Prolamellae present. Tutoria absent. Three pairs of rounded porose areas.
Description.Measurements. Body length: 348 (holotype, female), 315–348 (three paratypes, three females); notogaster width: 190 (holotype, female), 166–190 (three paratypes, three females).
Integument (Fig. 18). Body color light brown. Body surface densely microfoveolate (visible under high magnification). Lateral parts of subcapitular mentum slightly striate.
Prodorsum (Figs. 13, 15). Rostrum slightly protruding, rounded. Lamellae half of the prodorsum length (measured in lateral view). Prolamellae present, reaching insertions of rostral setae. Sublamellae slightly longer than lamellae, thin. Sublamellar porose areas rounded (2). Tutoria absent. Rostral, lamellar and interlamellar (all 49–53) setae setiform, barbed. Exobothridial setae (4) minute, thin, smooth. Bothridial setae (32–36; stalks in bothridia not considered) lanceolate, barbed, with long stalk and elongate head. Dorsosejugal porose areas oval (4 × 2). Anterior ridges on the lateral parts of prodorsum short, lateral and posterior ridges absent.
Notogaster (Figs. 13, 15–17). Anterior margin of notogaster not developed. Pteromorphs slightly developed, broadly rounded. Ten pairs of notogastral setae (20) setiform, with attenuate and flexible tip, smooth. Three pairs (Aa, A2, A3) of rounded porose areas (6–8) developed. Lyrifissures, opisthonotal gland openings, circumgastric scissure and circumgastric sigillar band distinct.
Gnathosoma (Figs. 18–20). Subcapitulum longer than wide (73–82 × 53–57). Subcapitular setae setiform, smooth, h (16) longer than a and m (12). Adoral setae (8) setiform, hook-like distally, smooth. Palps (length 45–49) with setation 0–2–1–3–9(+ω). Postpalpal setae (2) spiniform, smooth. Chelicerae (length 73–82) with two setiform, barbed setae, cha (26–28) longer than chb (16–18). Trägårdh’s organ of chelicerae elongate triangular.
Epimeral and lateral podosomal regions (Figs. 14, 15). Epimeral setae setiform, smooth; 1c shortest (6), 1b, 3b and 4a (12) longer than others (8–10). Humeral porose areas Am present, elongate oval, diffuse, poorly visible, Ah represented by saccules with small opening and conical channel. Pedotecta II rounded distally in ventral view. Discidia roundly triangular. Circumpedal carinae long, directed to triangular custodia.
Anogenital region (Figs. 14–17). Three pairs of genital and one pair of aggenital (8–10) setae simple. Two pairs of anal and three pairs of adanal (20) setae setiform, with attenuate and flexible tip, smooth. Adanal lyrifissures located close and parallel to anal plates. Postanal porose area not absent.
Legs (Figs. 21–24). All legs monodactylous, claw of each leg strong, smooth dorsally. Dorsoparaxial porose areas on femora I–IV and on trochanters III, IV slightly developed. Formulas of leg setation and solenidia: I (1–5–2–4–17) [1–2–2], II (1–5–2–4–15) [1–1–2], III (2–3–0–3–15) [1–1–0], IV (1–2–1–3–12) [0–1–0]; homology of setae and solenidia indicated in Table 1.
Material examined. Holotype (female) and three paratypes (three females): New Zealand, South Island, Nelson/Tasman region, Kahurangi National Park, Mount Arthur summit track, high alpine zone, 1508 m a.s.l., 41°12.467’ S, 172°42.119′ E, in soil under carpet grass Chionochloa australis, sample MtA-1, 23 January 2017 (collected by M. Minor).
Type deposition. The holotype (ethanol with a drop of glycerol) and two paratypes (ethanol with a drop of glycerol) are deposited in the New Zealand National Arthropod Collection, Auckland, New Zealand. One paratype (ethanol with a drop of glycerol) is deposited in the Tyumen State University Museum of Zoology, Tyumen, Russia.
Etymology. The specific name elatus (from Latin: elevated, raised) refers to the high altitude habitat of this species.
Differential diagnosis.Totobates elatus sp. n. is morphologically most similar to Totobates elegans (Hammer, 1958) from Argentina and the Antarctic region and Totobates ovalis Hammer, 1967 from New Zealand in having an elongate body, long prodorsal setae and well-developed notogastral setae, but differs by the lanceolate bothridial setae (versus bothridial setae clavate to fusiform). In addition, T. elegans has simple prodorsal and notogastral setae (versus prodorsal setae barbed, notogastral setae with attenuate and flexible tip in Totobates elatus sp. n.), and T. ovalis has a pointed rostrum (versus rostrum rounded in Totobates elatus sp. n.).
Remarks
-
1.
Ermilov et al. (2018) presented the generic diagnosis of Angullozetes. Some of morphological traits are: (1) prolamellae present, (2) humeral porose areas Am absent. However, Angullozetes kahurangiensis sp. n. has no prolamellae, and has humeral porose areas Am present. Hence, in the future these additions should be included in the updated generic diagnosis of Angullozetes.
-
2.
The genus Totobates differs from the haplozetid genus Maculobates Hammer, 1962 by one main morphological difference: pteromorphs movable versus immovable. However, we noted that the pteromorphal hinges are often unclearly developed and sometimes represented by short dorsal ridges only, instead of true hinges, therefore, the generic identification is unclear. Thus, the systematic placement of all species belonging to Totobates and Maculobates should be revised in the future.
References
Balogh J, Balogh P (2002) Identification keys to the oribatid mites of the extra-Holarctic regions. Vol. 1. Well-Press Publishing Limited, Miskolc
Ermilov SG, Shtanchaeva UYa, Subías LS (2018) Contribution to the knowledge of the oribatid mite genus Angullozetes (Acari: Oribatida: Scheloribatidae). Ecol Mont 18:75–81
Hammer M (1958) Investigations on the Oribatid fauna of the Andes Mountains. I. The Argentine and Bolivia. Biologiske Skrifter Det Köngelige Danske Videnskabernes Selskab 10(1):1–129
Hammer M (1961) Investigations on the oribatid fauna of the Andes Mountains. II. Peru. Biologiske Skrifter Det Köngelige Danske Videnskabernes Selskab 13(1):1–157
Hammer M (1962) Investigations on the oribatid fauna of the Andes Mountains. III. Chile. Biologiske Skrifter Det Köngelige Danske Videnskabernes Selskab 13(2):1–96
Hammer M (1967) Investigations on the oribatid fauna of New Zealand. Part II. Biologiske Skrifter Det Köngelige Danske Videnskabernes Selskab 15(4):1–60
Norton RA (1977) A review of F. Grandjean's system of leg chaetotaxy in the Oribatei (Acari) and its application to the family Damaeidae. In: Dindal DL (ed) Biology of oribatid mites. SUNY College of Environmental Science and Forestry, Syracuse, pp 33–61
Norton RA, Behan-Pelletier VM (2009) Suborder Oribatida. Chapter 15. In: Krantz GW, Walter DE (eds) A manual of acarology. Texas Tech University press, Lubbock, pp 430–564
Subías LS (2004) Listado sistemático, sinonímico y biogeográfico de los ácaros oribátidos (Acariformes: Oribatida) del mundo (excepto fósiles). Graellsia 60:3–305 Online version accessed in January 2018. http://bba.bioucm.es/cont/docs/RO_1.pdf
Travé J, Vachon M (1975) François Grandjean. 1882–1975 (Notice biographique et bibliographique). Acarologia 17(1):1–19
Acknowledgements
We cordially thank two anonymous reviewers for the valuable comments, Donald and Bunty Ladley (Karanga, 88 Valley, Wakefield, NZ) for providing us a home base for this research and for their invaluable help, Alastair Robertson (School of Agriculture & Environment, Massey University, Palmerston North, NZ) for help with fieldwork and plant identification, and Peter Beveridge (The Museum of New Zealand Te Papa Tongarewa, Wellington, NZ) for identification of mosses. We also thank the New Zealand Department of Conservation for sampling permit (national authorization # 50877-GEO).
Funding
The project was supported by the Massey University Research Fund.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No potential conflict of interest was reported by the authors.
Rights and permissions
About this article
Cite this article
Ermilov, S.G., Minor, M. New Oripodoidea (Acari, Oribatida) from New Zealand. Biologia 74, 1303–1311 (2019). https://doi.org/10.2478/s11756-018-00174-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11756-018-00174-y