Abstract
Purpose
To describe a new species of a parasitic copepod, Rhinergasilus digitus n. sp. (Cyclopoida, Ergasilidae), collected from the gills of the red-tailed lambari Astyanax fasciatus (Characiformes, Characidae) in two tributaries of the Jurumirim Reservoir (Upper Paranapanema River), São Paulo State, Brazil: Ribeirão dos Veados and Paranapanema River.
Methods
Fish were collected using multi-panel gills nets. The gill of each fish was washed and examined in a stereo microscope for copepods. The copepods found were stored in 70% ethanol, cleared in lactic acid, and mounted in Hoyer’s medium. Drawings were made with the aid of a Leica microscope DMLS equipped with a drawing tube.
Results
The new species differs from its congeneric species, Rhinergasilus piranhus (type-species), in having comparatively biggest body size (body length: 535–598 µm in the new species vs. 237–282 µm in R. piranhus); second antennary segment armed with a minute sensillum near middle of inner margin and a row of spinules on outer margin; third exopodal segment of leg 1 with digitiform process; interpodal plates two and three both ornamented with spinules along posterior margin; leg 5 reduced and represented by two unequal setae.
Conclusions
Based on the morphological differences described above, we erected a new species of Rhinergasilus. Rhinergasilus digitus n. sp. is the second ergasilid described from A. fasciatus, as well as it represents the first report of this genus in a characid fish.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ergasilidae Burmeister, 1835 represents one of the most important and biggest families of parasitic copepods of the order Cyclopoida Burmeister, 1834. It current comprises 28 valid genera and over than 260 species [1,2,3]. Ergasilids are recorded in all continents and zoogeographic regions excluding Antarctica, and occur in a wide variety of aquatic habitats: marine, brackish, and freshwater (the majority) [4]. Due to their unique life cycle, which is characterized by the presence of a parasitic phase (post-mated adult females) and a free-living phase (larvae ♀♂ and adult males) [1, 4], most ergasilids are only known by their parasitic adult females. Females are usually found attached to gills, fins, nasal fossae, embedded into the host tissues or in the urinary bladder of actinopterygian fishes, with few species on elasmobranchs and bivalve mollusks [3].
The Brazilian territory is inhabited by the richest biodiversity of Ergasilidae worldwide comprising over than 60 species from 17 genera [4, 5]. Most of the Brazilian ergasilids are found parasitizing the gills of freshwater fishes, with few records in other sites (e. g.: nasal fossae or urinary bladder) [3, 6]. Ergasilids of six genera are typically found associated with nasal fossae of Brazilian fishes, especially those species from the Amazon region, as follow: Brasergasilus Thatcher et Boeger, 1983; Gamidactylus Thatcher et Boeger, 1984; Gamispatulus Thatcher et Boeger, 1984; Gamispinus Thatcher et Boeger, 1984; Rhinergarsilus Boeger et Thatcher, 1988; and Vaigamus Thatcher et Robertson, 1984 [3]. Recently, Ergasilus tipurus Varella, Morey et Malta, 2019 and Therodamas elongatus (Thatcher, 1986) were recorded from nasal fossae of Amazonian fishes, increasing the number of genera in this family with species that parasitize nasal fossae of fishes in Brazil [3, 7].
Rhinergasilus is a monotypic genus described by Boeger and Thatcher [8] as a parasite of the nasal fossae of Pygocentrus nattereri Kner, 1858 from the Amazon region. The type-species, Rhinergasilus piranhus Boeger et Thatcher, 1988 can be easily distinguished from other ergasilids by the unique combination of diagnostic features, as follow: four-segmented antenna, three pairs of legs biramous, and legs 4 and 5, both reduced and represented by setae [8]. Since its description, specimens of Rhinergasilus has been reported in nasal fossae of fishes from three families (Acestrorhynchidae, Prochilodontidae, and Serrasalmidae) sampled in Amazonas, Minas Gerais and Paraná States, Brazil (see Table 4 for a list of fishes parasitized with Rhinergasilus species).
During a parasitological survey of fishes from two tributaries (Ribeirão dos Veados and Paranapanema River) of the Jurumirim Reservoir, Upper Paranapanema River, São Paulo State, Brazil, we detected several ectoparasitic ergasilids parasitizing the gills of the red-tailed lambari, Astyanax fasciatus (Cuvier, 1819). A morphological analysis of these specimens indicated that they represent a new species of Rhinergasilus, which is described herein.
Materials and Methods
Specimens of A. fasciatus were collected from April 2011 to October 2012 from two tributaries of the Jurumirim Reservoir: (1) Paranapenema River, Jurumirim Reservoir (23° 29′16.54″ S, 48° 37′12.88″ W), municipality of Angatuba, São Paulo State, Brazil; and (2) Ribeirão dos Veados River, Jurumirim Reservoir (23° 16′2.49″ S, 48° 38′15.72″ W), municipality of Itatinga, São Paulo State, Brazil.. Fish were collected using multi-panel gill nets (3–14 cm mesh) soaked for 14 h. Hosts were individually stored in plastic bags and placed in a freezer before necropsy. The gill of each fish was thawed by placing it in a Petri dish with tap water and examined for copepods using a stereomicroscope. Copepods were removed from the gill using a fine needle, stored in 70% ethanol, cleared in lactic acid, and mounted in Hoyer’s medium. Whenever necessary, some specimens were dissected in glycerol medium and then each part was mounted on individual slides. Coverslips were sealed with transparent nail varnish.
Morphological analyses and measurements of whole/dissected copepods were made using a microscope with differential interference contrast optics (Leica DMLB 5000, Leica Microsystems). Drawings were made with the aid of a microscope (Leica DMLS, Leica Microsystems, Wetzlar, Germany) equipped with a drawing tube. All measurements are in micrometers (μm) and presented as the mean and standard deviation followed by the range in parenthesis. Anatomical terms followed Boxshall and Montú [9], and that key was used to identify copepod specimens to the genus level. Abbreviations used throughout the text to refer to the name of the structures and segments present in Ergasilidae are shown in Table 1. The nomenclature used for the antennary segmentation has followed the following considerations: the ergasilid antenna is four-segmented (comprising coxobasis and three endopodal segments) and the claw is an armature element derived from the third endopodal segment [10].
Ecological descriptors such as prevalence, mean abundance, and intensity were calculated in accordance with Bush et al. [11].
Type specimens (holotype and paratypes) were deposited in the Invertebrate Collection of the Instituto Nacional de Pesquisas da Amazônia (INPA), municipality of Manaus, Amazonas State, Brazil.
Results
Familiy Ergasilidae Burmeister, 1835
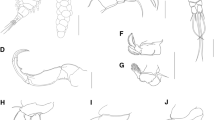
Rhinergasilus digitus n. sp. (Figs. 1–3; Tables 2, 3)
Description of adult female (based on 11 female specimens; no male observed): Body cyclopiform (Fig. 1a), comprising prosome, urosome, and caudal rami; prosome consisting of cephalosome and PS-1; PS-1 fused to cephalosome; and 3 free pedigerous somites. Cephalothorax bullet-shaped (Figs. 1a, 2a), with maximum width at level of buccal apparatus (Table 2), ornamented with two pairs of sensilla laterally (Fig. 2a). Pedigerous somites decreasing gradually in width from anterior to posterior; PS-2 narrower than cephalothorax, with paired integumental windows laterally on tergite (Fig. 1a); PS-4 reduced, smaller and thinner than other prosome somites (Fig. 1a, b).
Urosome consisting of PS-5, genital double-somite, and three free abdominal somites (Fig. 1b); PS-5 (Fig. 1b) reduced, unornamented; genital double-somite (Fig. 1b), 1.5 times wider than long, bearing paired slit-like genital apertures dorsally, ornamented with transverse row of spinules on ventral surface; abdominal somites decreasing in width from anterior to posterior, each somite ornamented with posterior spinule row along ventral margin (Fig. 1b); AS-3 (= anal somite) deeply incised posteriorly (= anus). Caudal rami (Fig. 1b), about 1.5 times longer than wide; each ramus ornamented with paired spinule rows on ventral surface and armed with four setae, all naked: seta 1 and 3 shortest, both setae inserted on ventral surface; seta 2 and 4, both setae inserted on posterior margin; seta 4 longest, about three times longer than seta 2.
Antennule six-segmented (Fig. 2b), setal formula: 1, 7, 3, 2 + 2 ae, 1 + 1 ae, 5 + 2 ae (total 24). Antenna (Fig. 2c) 4-segmented comprising coxobasis, and 3-segmented enp; coxobasis (= first segment) broad, with short naked seta; enp-1 (= second segment) ornamented with spinule row along outer margin and minute sensillum near middle of inner margin; enp-2 (= third segment) three times shorter than enp-1, unornamented; enp-3 (= fourth segment) reduced, unornamented; and terminal claw; claw curved, with fossa on concave margin.
Buccal apparatus (Fig. 1c) comprising mandible and maxilla; mandible armed with two blades (anterior and posterior blade); each blade with spinules along posterior margin; maxilla two-segmented, comprising syncoxa and basis; syncoxa broad, unornamented; basis covered with multiples spinules; maxillule vestigial, unornamented.
P1–P3 biramous (Fig. 3a–c), each comprising coxa, basis, endopod (inner ramus) and exopod (outer ramus). P1 (Fig. 3a); coxa unornamented; basis with bare outer seta and posterior margin produced posteriorly (arrowed in Fig. 3a); enp two-segmented, all segments with spinules along outer margin; enp-2 (= distal segment) with rounded end, armed with 2 setae; exp 3-segmented, all segments with spinules along outer margin; exp-1 (= proximal segment) with bristles on inner margin; exp-3 (= distal segment) with digitiform process (arrowed in Fig. a3a), armed with two serrated spines, one semi-plumose seta (serrated outer margin) and four plumose setae.
P2 (Fig. 3b); coxa unornamented; basis with bare outer seta; enp 3-segmented; enp-1 (= proximal segment) with bristles on outer margin; enp-2 and -3, both with spinules on outer margin; enp-3 (= distal segment) armed with one serrated spine and four plumose setae; exp 3-segmented, all segments with spinules along outer margin; exp-1 (= proximal segment) with bristles on inner margin; exp-3 (= distal segment) armed with one serrated spine, one semi-plumose seta (serrated outer margin) and five plumose setae. P3 (Fig. 3c) similar to P2, except for lacking spine on exp-3.
P4 absent (Fig. 1b). P5 reduced and represented by two unequal setae (Fig. 1b). Spine and setal formula of biramous swimming legs as presented in Table 3.
Intercoxal sclerites slender, unornamented, with both ends directed posteriorly (Fig. 3a–c). Interpodal plates of P2–P3 with a transverse spinule row on ventral surface (Fig. 3b, c); interpodal plate of P1, unornamented (Fig. 3a). Egg sac paired (Fig. 3d), multiseriate.
Taxonomic Summary
Type host: Astyanax fasciatus (Cuvier, 1819) (Characiformes: Characidae), red-tailed lambari.
Site of infection: Gill filaments.
Prevalence: 32.5%.
Intensity of infection: 4.3 ± 1.2 (1–39).
Type locality: Ribeirão dos Veados River, Jurumirim Reservoir, Upper Paranapanema River (23° 16′2.49″ S, 48° 38′15.72″ W), municipality of Itatinga, São Paulo State, Brazil.
Other locality: Paranapenema River, Jurumirim Reservoir, Upper Paranapanema River (23° 29′16.54″ S, 48° 37′12.88″ W), municipality of Angatuba, São Paulo State, Brazil.
Specimens deposited: The numbers of holotype and paratypes are: Holotype INPA 2515, Paratypes INPA 2516, INPA 2517, INPA 2517, INPA 2519 and INPA 2520.
Specimens examined: Rhinergasilus piranhus Boeger and Thatcher, 1988 – Holotype INPA PA 309-1, and Paratypes PA 309-2 to 309-5.
The Zoobank LSID is: urn:lsid:zoobank.org:pub:FAE7C164-50E2-4969-83D3-E30A23BB1B74.
Etymology: From Latin, the specific epithet refers to the presence of a digitiform process on exp-3 (= terminal segment) of P1.
Discussion
The present specimens were identified as members of the ergasilid genus Rhinergasilus by their possession of the diagnostic features proposed by Boeger and Thatcher [8] including the presence of three pairs of nonreduced legs, PS-4 and PS-5 greatly reduced, antennule 6-segmented and antenna 4-segmented.
The new species, R. digitus n. sp., resembles the type-species, Rhinergasilus piranhus in possessing third antennary segment (= enp-2) short, being shorter than previous antennary segments, antenna armed with a relatively long, curved claw, and P1 enp with modified distal segment (= second endopodal segment); in both species this segment lacks spines, has a rounded end and is armed with few setae, e. g., two setae in R. digitus and three setae in R. piranhus. Despite those similarities, the new species differs from the type-species in a number of features. The armature of antennary segments in R. digitus n. sp. differs from that of R. piranhus: in R. digitus n. sp. the second antennary segment (= enp-1) carries a minute sensillum near the middle of inner margin and a row of spinules on outer margin, whereas in R. piranhus this segment lacks any armature or ornament. The ornamentation of P1 is also different, R. piranhus has coxa and basis both ornamented with a row of spinules near inner posterior margin, whereas in R. digitus n. sp. those segments do not have such ornaments. Besides, the third exopodal segment of P1 in R. digitus n. sp. bears a digitiform process (located between the two distal spines) which is absent in the type-species. Another distinct difference between these two species is that in R. piranhus the first and second interpodal plates are both ornamented with spinules along posterior margin, while in R. digitus n. sp., the second and third interpodal plates have spinules (first interpodal plate without spinules in the new species). Moreover, the P4 and P5 are both represented by a seta in R. piranhus, while in the new species, the P4 is absent and P5 is represented by two unequal setae. Finally, the body size also differs, R. digitus n. sp. is about twice as long as R. piranhus: body length (less caudal setae) 535–598 in the new species vs. 237–282 in R. piranhus [8].
Based on the morphological differences described above, the present specimens were considered as new species of Rhinergasilus. Rhinergasilus digitus n. sp. is the second ergasilid described from A. fasciatus sampled in the Jurumirim reservoir (see Narciso et al. [12] and it is the first report of a Rhinergasilus species parasitizing a characid fish (Characidae). Characidae is the fourth family of Characiformes that has species parasitized by Rhinergasilus spp. A checklist of fishes parasitized by members of Rhinergasilus is provided (Table 4).
References
Boxshall GA (2016) A new species of Ergasilus von Nordmann, 1832 (Copepoda: Cyclopoida) from the gills of a dasyatid ray, Himantura oxyrhyncha (Sauvage, 1878) from West Kalimantan, Indonesia. Zootaxa 4174:93–103. https://doi.org/10.11646/zootaxa.4174.1.6
Taborda NL, Paschoal F, Luque JL (2016) altifrons and G. argyrostictus (Perciformes: Cichlidae) in the Brazilian Amazon. Acta Parasitol 61:549–555. https://doi.org/10.1515/ap-2016-0073
Varella AMB, Morey GAM, Malta JCO (2019) tipurus n. sp. (Copepoda: Ergasilidae), a parasite of Brazilian Amazon fish species. Acta Parasitol 64:187–194. https://doi.org/10.2478/s11686-018-00020-w
Boxshall GA, Defaye D (2008) Global diversity of copepods (Crustacea: Copepoda) in freshwater. Hydrobiol 595:195–207. https://doi.org/10.1007/s10750-007-9014-4
Luque JL, Pavanelli G, Vieira F, Takemoto R, Eiras J (2013) Checklist of Crustacea parasitizing fishes from Brazil. Check List 9:1449–1470. https://doi.org/10.15560/9.6.1449
Rosim DF, Boxshall GA, Ceccarelli PS (2013) A novel microhabitat for parasitic copepods: a new genus of Ergasilidae (Copepoda: Cyclopoida) from the urinary bladder of a freshwater fish. Parasitol Int 62:347–354. https://doi.org/10.1016/j.parint.2013.03.003
Morey GAM, Moreira AC, Morais AM, Atroch FMPB, Santana HP, Brandão NR, Dumbo IJC, Vital JF, Malta JCO (2016) Copepods (Crustacea: Ergasilidae) fish parasites of floodplain lakes of central Amazon, Brazil. Neotropic Helminthol 10:281–294
Boeger WA, Thatcher VE (1988) Rhinergasilus piranhus gen. et sp n (Copepoda, Poecilostomatoida, Ergasilidae) from the nasal cavities of Piranha Caju, Serrasalmus nattereri, in the Central Amazon. Proc Helminthol Soc Wash 55:5587–5590
Boxshall GA, Montú MA (1997) Copepods parasitic on Brazilian coastal fishes: a handbook. Nauplius 5:1–225
Rashidy H, Boxshall GA (1999) Ergasilid copepods (Poecilostomatoida) from the gills of primitive Mugilidae (grey mullets). Syst Parasitol 42:161–168. https://doi.org/10.1023/A:1006075223683
Bush AO, Lafferty KD, Lotz JM, Shostak AW (1997) Parasitology meets ecology on its own terms: Margolis et al. revisited. J Parasitol 83:575–583. https://doi.org/10.1645/13-394.1
Narciso RB, Brandão H, Perbiche-Neves G, Silva RJ (2019) fasciatus (Cuvier, 1819) (Actinopterygii: Characidae). Acta Parasitol. https://doi.org/10.2478/s11686-019-00108-x
Lacerda ACF, Takemoto RM, Lizama MAP, Pavanelli GC (2007) Parasitic copepods in the nasal fossae of five fish species (Characiformes) from the upper Paraná river floodplain, Paraná, Brazil. Acta Sci Biol Sci 29:429–435
Takemoto RM, Pavanelli GC, Lizama MAP, Lacerda ACF, Yamada FH, Moreira LHA, Ceschini TL, Bellay S (2009) Diversity of parasites of fish from the Upper Paraná River floodplain, Brazil. Braz J Biol 69:691–705. https://doi.org/10.1590/S1519-69842009000300023
Eiras JC, Takemoto RM, Pavanelli GC (2010) Diversidade dos parasites de peixes de água doce do Brasil. Cliche Tec, Maringá
Pavanelli GC, Takemoto RM, Eiras JC (2013) Parasitologia: Peixes de água doce do Brasil. Eduem, Maringá
Varella AMB (1992) Copépodos (Crustacea) parasitas das fossas nasais de peixes, coletados na região de Rondônia, Brasil. Dissertation, Universidade Estadual Paulista “Júlio de Mesquita Filho”
Morey GAM, Malta JCO (2016) Metazoary parasites of the nasal fossae of the Tambaqui, Colossoma macropomum (Cuvier, 1818) (Characiformes: Characidae) collected in floodplain lakes of the central Amazon, Brazil. Folia Amazónica 25:71–76. https://doi.org/10.24841/fa.v25i1.386
Thatcher VE (2006) Amazon fish parasites. Pensoft, Moscow
Luque JL, Tavares LER (2007) Checklist of Copepoda associated with fishes from Brazil. Zootaxa 1479:1–39
Morey GAM, Malta JCO (2016) The species of copepoda (Crustacea: Ergasilidae) parasites of gill filaments and nostrils of Serrasalmus altispinis (Merckx, Jégu and Santos, 2000) (Characiformes: Serrasalmidae) of central Amazon floodplain lakes, Brazil. Folia Amazónica 25:55–60. https://doi.org/10.24841/fa.v25i1.384
Arévalo EG, Morey GAM, Malta JCO (2018) Parasitic fauna of Prochilodus nigricans (Prochilodontidae) from Brazilian Amazon floodplain lakes. Biota Amazônia 8:19–21. https://doi.org/10.18561/2179-5746/biotaamazonia.v8n1p19-21
Godinho HP, Godinho AL (2003) Águas, peixes e pescadores do São Francisco das Minas Gerais. PUC Minas, Belo Horizonte
Acknowledgements
This study was supported by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) (Proc. N°: 11/24159-3). R.B.N thanks the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for the financial support given to the study (132844/2018-4). R.J.S. is supported by FAPESP #2016/50377-1; CNPq #309125/2017–0; CNPq-PROTAX #440496/2015-2.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical standards
All applicable international, national, and/or institutional guidelines for the use and care of animals were followed.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Narciso, R.B., Brandão, H., Perbiche-Neves, G. et al. A New Species of Rhinergasilus Boeger et Thatcher, 1988 (Copepoda: Ergasilidae) from Gills of Astyanax fasciatus (Cuvier, 1819) (Actinopterygii: Characidae). Acta Parasit. 65, 327–334 (2020). https://doi.org/10.2478/s11686-020-00168-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11686-020-00168-4