Abstract
Purpose
Light microscopic manual count is the current gold standard for parasite quantification. The ability to determine parasite density in whole blood is crucial to understanding disease pathogenesis and finding a suitable automated method of Babesia rossi parasite quantification would facilitate higher throughput and provide results that are more objective. This study investigated both peripheral capillary and central venous whole blood to estimate the correlations between light microscopy, flow cytometry and quantitative real-time polymerase chain reaction (qPCR).
Methods
Peripheral capillary and central venous blood were sampled from 40 naturally B. rossi-infected dogs and 10 healthy control dogs. Samples were analysed by reverse line blot hybridization assay to confirm a mono-B. rossi infection. Capillary blood parasite density was detected using light microscopic manual counting and venous blood parasitaemia detected by manual counts, flow cytometry and qPCR.
Results
A significant correlation was found between the venous manual counts and flow cytometry (rs = 0.465; P < 0.001), as well as qPCR (rs = − 0.500; P < 0.001). A significant correlation was also observed between the capillary manual counts compared to venous manual counts (rs = 0.793; P < 0.001), flow cytometry (rs = 0.399; P = 0.004), and qPCR (rs = − 0.526; P < 0.001).
Conclusions
The study results suggest that qPCR is of value as an alternative to the gold standard manual count for detecting B. rossi parasitaemia in canine whole blood and that flow cytometry may be useful with further refinement of issues such as background fluorescence and the influence of reticulocytes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Babesia rossi is the most prevalent species of Babesia in South Africa [23], where acute B. rossi infections can be detected and quantified accurately by light microscopic demonstration of parasitaemia on a thin stained capillary blood smear [4]. Light microscopy is utilized traditionally as the gold standard in diagnosis of B. rossi infections, but the limitations of this diagnostic tool have been reported for both malaria [29, 30, 40] and Babesia [26]. Flow cytometry [17, 21, 35] and quantitative real-time polymerase chain reaction (qPCR) [16, 28, 37] are automated diagnostic modalities that have been used for several studies in detection and quantitative analysis of malaria. Similarly, flow cytometry [2, 46] and quantitative real-time polymerase chain reaction [9, 43, 44] have proven efficient in detecting and quantifying Babesia infections, but an investigation into a comparison between these diagnostic tools and light microscopy for the detection and quantification of B. rossi parasitaemia has not been observed. Light microscopy has been compared to flow cytometry and qPCR for malaria in the same study [7]. In the study presented here, we investigated the correlation between the quantitative abilities of these three tools for the canine Babesia parasite. Flow cytometry facilitates higher throughput capabilities and less subjectivity [7], but unwanted background fluorescence may be problematic [14]. Excess background fluorescence results in discrepancies at low parasitaemias [18]. Reticulocyte interference is arguably the most important confounder in cytometric red blood cell (RBC) analysis due to the problems of non-specific staining of reticulocytes [14, 18, 45]. Other potential confounders such as normoblasts and Howell–Jolly bodies have also been recorded [38]. Conventionally, alternative staining methodologies are used to try and combat the issues of unwanted background fluorescence [21, 35]. The similarities shared between Babesia and malaria [8, 19, 34] are well documented and studies that investigated the correlation between parasite density and disease severity in malaria [11, 20] and Babesia [4] have highlighted the essential role parasite quantification plays in understanding disease pathogenesis. Investigations into the pathogenesis of malaria [25, 39] and Babesia [13, O’Connor et al. 1999] suggest that virulence is attributed to certain parasites, because of their unique ability to cytoadhere and sequester in the microvasculature [1, 15]. The hypothesis that B. rossi sequesters in the capillary endothelium of its host and that parasite density and disease severity may be related to the parasite’s pathogenesis [4] deserves further consideration and necessitate the use of modalities that facilitate efficient parasite quantification. Our study sought to investigate if statistically significant correlations exist between manual light microscopic, flow cytometric and qPCR methods for detecting and quantifying B. rossi parasitaemia in fresh whole blood from naturally infected dogs.
Materials and Methods
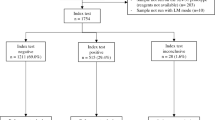
The study population consisted of 50 dogs, 40 having been confirmed infected with babesiosis and 10 healthy dogs sampled as controls. Dogs were selected for the study based on clinical findings classically suggestive of babesiosis and confirmed on a thin, stained capillary blood smear. Samples were scrutinized further by means of reverse line blot hybridization assay (RLB) to ensure a mono-infection of B. rossi. Control dogs were considered healthy based on history, clinical examination, peripheral blood smear evaluation and RLB to rule out parasitaemia.
A thin peripheral capillary blood smear was made from the ear pinna of each dog. Approximately 4 ml of venous blood was collected into ethylenediamine tetra-acetic acid (EDTA) tubes from either the jugular or cephalic vein of each animal and stored at 4 °C until processed within 12 h. A thin venous blood smear was also made from each EDTA sample.
The RLB was conducted by the Department of Veterinary Tropical Diseases (DVTD) at the Faculty of Veterinary Science, Onderstepoort, South Africa as described previously [43]. The assay ensured that the infected samples included in the dataset contained a mono-infection of B. rossi and excluded other species of Babesia, Ehrlichia, Theileria and Anaplasma, and that the control samples were free of Babesia and other pathogens. The qPCR assay was performed by the DVTD according to standard operating procedures as previously described [43].
A semi-quantitative non-volumetric method was used to quantify parasitaemias similar to what has previously been described [4]. A total of 1950 red blood cells were examined per smear. Results were calculated and for each smear expressed as a percentage parasitaemia by use of the formula: total number parasitized RBCs/total number RBCs × 100 = % parasitaemia.
Sample staining using SYBR Green I nucleic acid gel stain—10,000× concentrate in dimethyl sulfoxide (Invitrogen, catalogue no. S7563) was performed similar to what has previously been described [36], but was optimized for B. rossi. SYBR Green I working stock was prepared at a 1:1000 concentration by diluting 1 µl SYBR Green I stock with 999 µl phosphate-buffered saline (PBS). Two falcon tubes were marked ‘unstained’ and ‘stained’, respectively. 50 µl whole blood sample was pipetted into each of the two falcon tubes and cells washed with 1.5 ml PBS, vortexed for 5 s, centrifuged at 450 relative centrifugal force (rcf) (1725 revolutions per minute (rpm), where the radius (R) = 135 mm) for 5 min at 23 °C and the supernatant pipetted off. The cell pellet in the tube marked ‘unstained’ was re-suspended in 20 µl PBS, and the pellet re-suspended in 5 µl SYBR Green I dye working stock (1:1000 dilution). Both tubes were vortexed for 5 s and incubated in the dark for 30 min at 37 °C. Following incubation, the contents of each tube were washed with 1.5 ml PBS, vortexed for 5 s, centrifuged as before for 5 min at 23 °C and the supernatant pipetted off. Each tube’s cell pellet was finally re-suspended in 500 µl PBS, vortexed for 5 s and analysed by use of an Accuri C6 flow cytometer (BD Biosciences, Franklin Lakes, New Jersey, USA) interfaced with a computer and analysed using BD CSampler Software (BD Biosciences, Franklin Lakes, New Jersey, USA). Both a stained and unstained preparations were analysed. An optimized gating strategy was used as a template to gate for the red cell zone of interest. Fifty thousand events were recorded per sample. The number of gated events was recorded on a forward scatter (FSC) versus side scatter (SSC) dot plot. By use of an optimized quadrant marker on the FL-1 vs. FL-4 dot plot, the number of SYBR Green I positive events detected in the FL-1 channel in the upper left quadrant were recorded. Unadjusted parasitaemia was determined by the following formula: (SYBR Green I positive events/total gated events) × 100 = estimated % parasitaemia (unadjusted).
A correction factor methodology was designed that allowed for mathematical correction of the parasitized red cell percentage for background fluorescence and reticulocytes counted to calculate an adjusted flow cytometric result, which presented a more accurate estimate of the percentage parasitaemia (Online Resource 1). Reticulocyte interference was estimated by determining what percentage of SYBR Green I event fluorescence could likely be attributed to reticulocytes. The reticulocyte percentage was determined by the ADVIA 2120 automated cell counter (Siemens, Munich, Germany). Both infected and control samples were corrected for reticulocytes. The correction factor allowed for an adjusted flow cytometric estimate of parasitaemia, which represented a truer estimate of % parasitaemia after correction for both background and reticulocyte count.
Data analysis was performed using IBM SPSS Statistics version 24, (International Business Machines Corp., Armonk, NY, USA) and MedCalc for Windows, version 18 (MedCalc Software, Ostend, Belgium). Spearman’s rank-order correlation coefficient (rs) was used to describe the correlations among the parasite quantification methods, due to the non-normal data distribution and small sample size. The probability value (P value) was calculated based on the rs to determine significance. Scatter plots were used to graphically present correlations between the assays and a linear regression line fitted to indicate the direction of the relationship. Pearson’s correlation coefficient (rp) and the coefficient of determination (R2) was calculated for the purpose of regression analysis. Statistical results were interpreted at the 5% level of significance. A receiver-operating characteristics (ROC) curve analysis was employed to measure diagnostic test performance and the Youden J index was used to identify the most efficient cutoff value for classification of case and control dogs.
Results
Table 1 presents the Spearman’s rho correlations and the significance of the correlations for the three diagnostics tools used to detect B. rossi parasitaemia in this study. Figures 1, 2, and 3 show the scatter plots between the measurements from the diagnostic tools and include fitted linear regression lines. The linear regression lines fitted to each scatter plot in Figs. 1 and 2 demonstrate a positive correlation between the data points. The regression lines fitted to the scatter plots in Fig. 3 demonstrate a negative correlation due to the inverse proportional relationship between parasitaemia and Cq. The rp and R2 are also presented to show how close the data fits to each regression line. Capillary and venous manual parasite count had a high and significant correlation (rs = 0.793, P < 0.001, Fig. 1) for determining parasite density, as seen in Table 1. Significant correlations for parasite quantification were observed between flow cytometry and capillary manual count (rs = 0.399, P = 0.004, Fig. 2a), as well as venous manual count (rs = 0.465, P < 0.001, Fig. 2b), as seen in Table 1. Finally, significant correlations for parasite quantification are recorded in Table 1 between qPCR Cq and both log-transformed capillary manual count parasitaemia (rs = − 0.526, P < 0.001, Fig. 3a) and log-transformed venous manual count parasitaemia (rs = − 0.500, P < 0.001, Fig. 3b).
The ROC curve analysis which includes an area under the curve (AUC) and P value, as well as the Youden index J for manual counts, flow cytometry and qPCR of the study population are presented in Table 2. The AUC and significance of the ROC curves analysis allows making an assessment of the diagnostic test performance of each assay. RLB data were used as the baseline metric diagnostic test for analysis. ROC curve analysis revealed excellent test performance (AUC = 1.0, Youden J index = 1.0) for capillary manual count for diagnosis of Babesia. Likewise, good diagnostic test performance was observed for venous manual counts (AUC = 0.925, Youden J index = 0.85). This was expected, because light microscopy is currently the gold standard for parasite diagnosis and quantification [41]. ROC curve analysis provided fair diagnostic test performance for flow cytometry (AUC = 0.85, Youden J index = 0.65). ROC curve analysis revealed excellent diagnostic test performance for qPCR (AUC = 1.0, Youden J index = 1.0), the same as the gold-standard capillary manual count.
Discussion
Our study sought to investigate alternative diagnostic tools to conventional microscopy to detect and quantify the parasitaemia of B. rossi. Flow cytometry has been used in a prior study that investigated white blood cell phenotypes in B. rossi-infected whole blood [32], but has never before been used to detect and quantify B. rossi parasitaemia. SYBR Green I dye has been enlisted in several malaria studies as a stain for parasites [31, 33, 42], but its use in Babesia parasitaemia research has been limited to B. divergens [36]. Molecular diagnostics like qPCR have been used with success in investigations of B. rossi and other Babesia parasites [9, 43, 44], but comparing microscopy, flow cytometry and qPCR as a means of detecting parasite density in one study provides the opportunity to correlate their diagnostic and quantitative abilities with each other. Studies which compared these three tools as a means of diagnosis and to determine parasite density in investigations of malaria in humans [7] and to diagnose Babesia in cattle [10] prompted this study to adapt a similar approach for canine babesiosis. The success with which flow cytometry [17, 21, 35] and quantitative real-time polymerase chain reaction (qPCR) [16, 28, 37] have been used in diagnostic and quantitative analysis of malaria suggest that similar success could be achieved with the use of these modalities in canine babesiosis. More objective results facilitated by modalities that have higher throughput and that can detect and quantify parasitaemia with ease will likely aid future studies investigating the relationship between B. rossi parasite biomass, disease severity and parasite pathogenesis.
The differences in parasite density found in capillary and venous blood is documented for B. rossi [4] and other Babesia parasites [6, 24] and prompted a comparison between blood sourced from both sites. The capillary parasitaemia was significantly higher than the venous parasitaemia for the majority of the B. rossi-infected population, similar to observations made in a previous study of B. rossi [4]. Interestingly, 5/40 (12.5%) infected samples had a higher venous parasitaemia than capillary parasitaemia; a phenomenon also observed in a prior study [4] and suggest that sequestration, if indeed present in B. rossi, only occurs in some individuals or is associated with certain stages of infection. Sequestration may be associated with specific parasite genotypes [22], but further investigations into this hypothesis are needed. Capillary and venous manual parasite counts showed a high and significant correlation between the parasitaemias recorded for B. rossi in the same dog and regression analysis revealed a positive correlation between the datasets. Furthermore, results show that capillary parasitaemias are generally higher compared to venous parasitaemias, which indicate that parasite sequestration may be occurring or there may be a rheological phenomenon resulting in parasite accumulation in the microvasculature.
Flow cytometry [17, 21, 35] has been used with success for several studies on detection and quantitative analysis of malaria, but has never before been used in the quantification of B. rossi parasitaemia. Although SYBR Green I is an effective nuclear marker for the detection of babesiosis using flow cytometry [36], analysis was limited by use of SYBR Green I as a single dye reagent. Lack of a duel- or tri-staining methodology complicated distinguishing pRBCs from reticulocytes. This is of particular importance in infections of the red blood cell that result in anaemia (as in babesiosis) and thus an expected increase in the reticulocyte count. Unresolved issues of reticulocyte interference remain that relate to a lack of knowledge regarding to what extend B. rossi targets immature RBCs, as is seen with other Babesia parasites [5]. A non-conventional approach to the problem of background and reticulocyte confounders was utilized by introducing a correction factor that mathematically adjusted the raw flow cytometric results to compensate for both autofluorescence and reticulocyte interference (Online Resource 1). The results still yielded only a rough estimate of parasitaemia and the methodology would only prove valuable for routine use with further refinement. Several limitations of this study render the adjusted flow cytometry results an estimate of parasitaemia at best. Our methodology did not account for infected reticulocytes and research into the predilection of B. rossi for immature RBCs should be explored, as has been done for B. gibsoni [46] and observed for other Babesia parasites [5]. In addition, depending on an assay’s performance and limitations, it may fail to detect very low parasitaemias compared to more sensitive methods such a qPCR. The influence of other potential confounders such as normoblasts and Howell–Jolly bodies [38] was regarded as negligible compared to the issues introduced by reticulocytes in this study. Although the correction factor methodology improved the results in this study, the importance of the impact autofluorescence and reticulocyte interference have on parasite detection and quantification results need to be investigated further. Cytometric profiling can, however, be performed relatively quickly and provides the advantages of automation [7], despite the problems of background and reticulocyte interference encountered [14]. Overall, the results from the flow cytometric analysis indicate that there are statistically significant correlations between the parasitaemia of B. rossi in both capillary and venous blood determined by manual count when compared to the estimated parasite density of B. rossi in venous blood determined by flow cytometry in the same dog. Correlations between the flow cytometry and manual counts were, however, not high, despite proving statistically significant and regression analysis revealed a positive correlation between the datasets. The venous manual count outlier potentially skews the data in Fig. 2b, whereby the regression line might not necessarily be completely representative of the relationship between the data points. A possible interpretation of this phenomena might be that flow cytometric assessment of venous blood overestimates parasitaemia, compared to venous manual count, or that the venous count outlier may be due to the subjective limitations of manual parasite determination and operator error. The potential of using flow cytometry as an additional diagnostic tool to detect and quantify Babesia parasitaemia has been demonstrated. Sample storage, preparation, processing and data analysis all influence the final results and have a bearing on the accuracy of estimating the sample parasitaemia, hence the methodology presented here could be further refined by exploring these variables.
qPCR has been used successfully in the study of B. rossi and other Babesia parasites [9, 43, 44], as well as malaria [16, 28, 37]. Although conventional PCR has been employed for the detection of B. rossi to good effect [3, 12], the progressive advantages of qPCR made it attractive for use in this study. The relationship between Cq and parasitaemia is inversely proportional and this study sought to investigate the correlations between qPCR Cq and % parasitaemia as determined by capillary and venous manual count. Correlations between qPCR and manual counts were statistically significant. The qPCR findings in this study concur with previous investigations where light microscopy and PCR [10] or qPCR [7] were compared by demonstrating that qPCR is far more sensitive and reliable in parasite detection. Although high diagnostic costs limit its use as a routine screening test for B. rossi, its value in a laboratory or research setting requiring quantitative parasite analysis or detection of low-density parasitaemias is promising. The correlation results and regression analysis revealed a negative correlation between the datasets due to the inversely proportional relationship of Cq and parasitaemia. Results from the qPCR analysis indicate that there are statistically significant correlations between the parasitaemia of B. rossi in both capillary and venous blood determined by manual count when compared to the estimated parasite density of B. rossi in venous blood determined by qPCR in the same dog. The value of qPCR as an alternative molecular diagnostic technique to detect and quantify B. rossi parasitaemia in whole blood was thus demonstrated.
Conclusions
Comparative analysis in this study demonstrated statistically significant correlations between manual count, flow cytometry and qPCR for the detection of B. rossi parasitaemia in both capillary and venous canine whole blood. The well-documented differences between peripheral capillary and central venous parasitaemia may be related to the disease pathogenesis of B. rossi. The results suggest the use of qPCR for B. rossi parasite detection and quantification proved efficient; flow cytometry showed promise if problems presented by background fluorescence and reticulocyte detection can be refined; and automated methods of B. rossi parasite detection and quantification may be a potentially suitable alternative to the labour intensive gold-standard light microscopy. In the author’s opinion, the use of light microscopy remains a suitable gold-standard technique for the diagnosis of B. rossi. qPCR might, however, prove to be a more reliable and faster method for the purposes of quantitative analysis of B. rossi parasite density, especially in samples with low parasitaemia.
Data Availability
The datasets generated during and/or analysed during the current study care are available in the University of Pretoria repository, http://hdl.handle.net/2263/66680.
References
Allred DR, Al-Khedery B (2004) Antigenic variation and cytoadhesion in Babesia bovis and Plasmodium falciparum: different logics achieve the same goal. Mol Biochem Parasitol 134:27–35
Bicalho KA, Ribeiro MF, Martins-Filho OA (2004) Molecular fluorescent approach to assessing intraerythrocytic hemoprotozoan Babesia canis infection in dogs. Vet Parasitol 125:221–235
Birkenheuer AJ, Levy MG, Breitschwerdt EB (2003) Development and evaluation of a seminested PCR for detection and differentiation of Babesia gibsoni (Asian Genotype) and B. canis DNA in canine blood samples. J Clin Microbiol 41:4172–4177
Bohm M, Leisewitz AL, Thompson PN, Schoeman JP (2006) Capillary and venous Babesia canis rossi parasitaemias and their association with outcome of infection and circulatory compromise. Vet Parasitol 141:18–29
Borggraefe I, Yuan J, Telford SR et al (2006) Babesia microti primarily invades mature erythrocytes in mice. Infect Immun 74:3204–3212
Callow LL, Pepper PM (1974) Measurement of and correlations between fever, changes in the packed cell volume and parasitaemia in the evaluation of the susceptibility of cattle to infection with Babesia argentina. Aust Vet J 50(1):1–5
Campo JJ, Aponte JJ, Nhabomba AJ et al (2011) Feasibility of flow cytometry for measurements of Plasmodium falciparum parasite burden in studies in areas of malaria endemicity by use of bidimensional assessment of YOYO-1 and autofluorescence. J Clin Microbiol 49(3):968–974
Clark I, Jacobson L (1998) Do babesiosis and malaria share a common disease process? Ann Trop Med Parasitol 92(4):483–488
Costa LM Jr, Zahler-Rinder M, Ribeiro MF et al (2012) Use of a real time PCR for detecting subspecies of Babesia canis. Vet Parasitol 188:160–163
Costa-Junior LM, Rabelo EM, Martins Filho OA, Ribeiro MF (2006) Comparison of different direct diagnostic methods to identify Babesia bovis and Babesia bigemina in animals vaccinated with live attenuated parasites. Vet Parasitol 139(1–3):231–236
Cunnington AJ, Walther M, Riley EM (2013) Piecing together the puzzle of severe malaria. Sci Transl Med 5(211):211ps218
Duarte SC, Linhares GF, Romanowsky TN, da Silveira Neto OJ, Borges LM (2008) Assessment of primers designed for the subspecies-specific discrimination among Babesia canis canis, Babesia canis vogeli and Babesia canis rossi by PCR assay. Vet Parasitol 152:16–20
Gohil S, Kats LM, Sturm A, Cooke BM (2010) Recent insights into alteration of red blood cells by Babesia bovis: moovin’ forward. Trends Parasitol 26:591–599
Grimberg BT (2011) Methodology and application of flow cytometry for investigation of human malaria parasites. J Immunol Methods 367(1–2):1–16
Hutchings CL, Li A, Fernandez KM et al (2007) New insights into the altered adhesive and mechanical properties of red blood cells parasitized by Babesia bovis. Mol Microbiol 65:1092–1105
Hwang SY, Kim SH, Lee GY et al (2011) A novel real-time PCR assay for the detection of Plasmodium falciparum and Plasmodium vivax malaria in low parasitized individuals. Acta Trop 120(1–2):40–45
Izumiyama S, Omura M, Takasaki T, Ohmae H, Asahi H (2009) Plasmodium falciparum: development and validation of a measure of intraerythrocytic growth using SYBR Green I in a flow cytometer. Exp Parasitol 121(2):144–150
Jiménez-Díaz MB, Rullas J, Mulet T et al (2005) Improvement of detection specificity of Plasmodium-infected murine erythrocytes by flow cytometry using autofluorescence and YOYO-1. Cytometry Part A 67(1):27–36
Krause PJ, Daily J, Telford SR et al (2007) Shared features in the pathobiology of babesiosis and malaria. Trends Parasitol 23(12):605–610
Lyke KE, Diallo DA, Dicko A et al (2003) Association of intraleukocytic Plasmodium falciparum malaria pigment with disease severity, clinical manifestations, and prognosis in severe malaria. Am J Trop Med Hyg 69(3):253–259
Malleret B, Claser C, Ong AS et al (2011) A rapid and robust tri-color flow cytometry assay for monitoring malaria parasite development. Sci Rep 1:118
Matjila PT, Carcy B, Leisewitz AL et al (2009) Preliminary evaluation of the BrEMA1 gene as a tool for associating Babesia rossi genotypes and clinical manifestation of canine babesiosis. J Clin Microbiol 47:3586–3592
Matjila PT, Leisewitz AL, Jongejan F, Penzhorn BL (2008) Molecular detection of tick-borne protozoal and ehrlichial infections in domestic dogs in South Africa. Vet Parasitol 155:152–157
Mendis K, Carter R (1995) Clinical disease and pathogenesis in malaria. Parasitol Today 11(5):PTI1–PTI16
Miller LH, Baruch DI, Marsh K, Doumbo OK (2002) The pathogenic basis of malaria. Nature 415(6872):673–679
Mosqueda J, Olvera-Ramirez A, Aguilar-Tipacamu G, Canto GJ (2012) Current advances in detection and treatment of babesiosis. Curr Med Chem 19:1504–1518
O’Connor RM et al (1999) Cytoadherence of Babesia bovis-infected erythrocytes to bovine brain capillary endothelial cells provides an in vitro model for sequestration. Infect Immun 67(8): 3921–3928
Osoga J, Waitumbi J, Guyah B et al (2017) Comparative evaluation of fluorescent in situ hybridization and Giemsa microscopy with quantitative real-time PCR technique in detecting malaria parasites in a holoendemic region of Kenya. Malar J 16(1):297
O’meara WP, McKenzie FE, Magill AJ (2005) Sources of variability in determining malaria parasite density by microscopy. Am J Trop Med Hyg 73(3):593–598
Payne D (1988) Use and limitations of light microscopy for diagnosing malaria at the primary health care level. Bull World Health Organ 66(5):621
Philipp S, Oberg HH, Janssen O, Leippe M, Gelhaus C (2012) Isolation of erythrocytes infected with viable early stages of Plasmodium falciparum by flow cytometry. Cytometry Part A 81(12):1048–1054
Rautenbach Y, Goddard A, Thompson PN, Mellanby RJ, Leisewitz AL (2017) A flow cytometric assessment of the lymphocyte immunophenotypes in dogs naturally infected with Babesia rossi. Vet Parasitol 241:26–34
Rebelo M, Shapiro HM, Amaral T, Melo-Cristino J, Hänscheid T (2012) Haemozoin detection in infected erythrocytes for Plasmodium falciparum malaria diagnosis—prospects and limitations. Acta Trop 123(1):58–61
Reyers F, Leisewitz A, Lobetti R, Milner R, Jacobson L (1998) Canine babesiosis in South Africa: more than one disease. Does this serve as a model for falciparum malaria? Ann Trop Med Parasitol 92(4):503–511
Roobsoong W, Maher SP, Rachaphaew N et al (2014) A rapid sensitive, flow cytometry-based method for the detection of Plasmodium vivax-infected blood cells. Malar J 13(1):55
Rossouw I, Maritz-Olivier C, Niemand J et al (2015) Morphological and molecular descriptors of the developmental cycle of Babesia divergens parasites in human erythrocytes. PLoS Negl Trop Dis 9:e0003711
Schneider P, Wolters L, Schoone G et al (2005) Real-time nucleic acid sequence-based amplification is more convenient than real-time PCR for quantification of Plasmodium falciparum. J Clin Microbiol 43(1):402–405
Shapiro HM, Apte SH, Chojnowski GM et al (2013) Cytometry in malaria—a practical replacement for microscopy? Curr Protoc Cytom 65:11–20
Sherman IW, Eda S, Winograd E (2003) Cytoadherence and sequestration in Plasmodium falciparum: defining the ties that bind. Microbes Infect 5(10):897–909
Sori G, Zewdie O, Tadele G, Samuel A (2018) External quality assessment of malaria microscopy diagnosis in selected health facilities in Western Oromia, Ethiopia. Malar J 17(1):233
Tavares R, Staggemeier R, Borges A et al (2011) Molecular techniques for the study and diagnosis of parasite infection. J Venom Anim Toxins Incl Trop Dis 17(3):239–248
Theron M, Hesketh RL, Subramanian S, Rayner JC (2010) An adaptable two-color flow cytometric assay to quantitate the invasion of erythrocytes by Plasmodium falciparum parasites. Cytom Part A 77(11):1067–1074
Troskie M, De Villiers L, Leisewitz AL, Oosthuizen MC, Quan M (2019) Development and analytical validation of a multiplex, real-time PCR assay for Babesia rossi and Babesia vogeli. Ticks Tick Borne Dis 10(2):421–432
Wang C, Ahluwalia SK, Li Y et al (2010) Frequency and therapy monitoring of canine Babesia spp. infection by high-resolution melting curve quantitative FRET-PCR. Vet Parasitol 168:11–18
Wongchotigul V et al (2004) The use of flow cytometry as a diagnostic test for malaria parasites. South-east Asian J Trop Med Public Health 35(3):552–559
Yamasaki M, Otsuka Y, Yamato O, Tajima M, Maede Y (2000) The cause of the predilection of Babesia gibsoni for reticulocytes. J Vet Med Sci 62:737–741
Acknowledgements
This work was supported by the National Research Foundation of South Africa under a grant held by ALL (CPRR13080726333), as well as by the Department of Companion Animal Studies, Faculty of Veterinary Science, Onderstepoort, South Africa. We thank Dr. Jeanne Rudman from Mamelodi Animal Health Clinic and Sr. Marizelle de Clercq from the Onderstepoort Animal Blood Bank for assistance in sample collections. We also thank Prof. Marinda Oosthuizen, Prof. Geoffrey Fosgate and Mr. Pieter de Villiers for their contributions. A special thanks to Doornpoort Animal Clinic for allowing us to sample at the clinic.
Funding
This work was supported by the National Research Foundation of South Africa under a grant held by ALL (CPRR13080726333), as well as by the Department of Companion Animal Studies, Faculty of Veterinary Science, Onderstepoort, South Africa.
Author information
Authors and Affiliations
Contributions
LDV wrote the manuscript, with input from ALL, as part of fulfilment of the requirements for obtaining the degree M.Sc. Veterinary Science. LDV assisted in sample collection and performed the light microscopic and flow cytometric laboratory work. ALL supervised the study and assisted with sample collections. MQ co-supervised the study and developed the qPCR protocol along with MT, who performed all the RLB and qPCR laboratory work. JCJ performed the statistical analysis for the study, with input from LDV.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with ethical required in terms of the University of Pretoria’s Code of ethics for researchers and the Policy guidelines for responsible research and ethical clearance was granted by the Animal Ethics Committee of the Faculty of Veterinary Science, Onderstepoort (Reference: V060-16).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
de Villiers, L., Quan, M., Troskie, M. et al. A Comparison Between Manual Count, Flow Cytometry and Quantitative Real-Time Polymerase Chain Reaction as a Means of Determining Babesia rossi Parasitaemia in Naturally Infected Dogs. Acta Parasit. 65, 128–135 (2020). https://doi.org/10.2478/s11686-019-00134-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11686-019-00134-9