Abstract
Objectives
The objective of this study is to describe an important waterborne outbreak of gastrointestinal illness observed in a rural municipality of Quebec.
Methods
A population-based retrospective cohort study was conducted to identify risk factors associated with acute gastroenteritis. Indirect surveillance data were used to estimate the extent and the resolution of the epidemic.
Results
The cohort consisted of 140 randomly selected individuals of whom 22 met the illness case definition (15.7% attack rate). The epidemic curve was similar to the evolution of antidiarrheal products sold by the only pharmacy in town and calls made to the Health Info Line. Bivariate analysis led to identifying five risk factors of gastrointestinal illness: consumption of municipal water, contact with someone with acute gastroenteritis (within and outside of the household), contact with a child in daycare, and being less than 35 years of age. Drinking municipal water had the highest risk ratio (RR = 24.31; 95% CI = 1.50–393.4). Drinking water from a private artesian well was a protective factor (RR = 0.28; 95% CI = 0.09–0.90).
Conclusion
This study highlighted that managing the risks associated with the consumption of untreated drinking water remains an important public health challenge, particularly in small rural municipalities vulnerable to climate variability.
Résumé
Objectif
L’objectif de cette étude est de décrire une éclosion de gastroentérite aigüe d’origine hydrique survenue dans une municipalité rurale du Québec.
Méthode
Une étude de cohorte populationnelle rétrospective a été réalisée pour identifier des facteurs de risque associés au développement des gastroentérites aigües. Des données de surveillance indirecte ont été utilisées pour estimer l’étendue de la situation épidémique.
Résultats
La cohorte a été constituée de 140 individus sélectionnés aléatoirement, dont 22 répondaient à la définition de cas (taux d’attaque 15,7 %). La courbe épidémique a suivi une évolution semblable à celle de la vente de produits antidiarrhéiques vendus dans la seule pharmacie de la municipalité et aux appels faits au service Info-Santé. Des analyses bivariées ont permis d’identifier cinq facteurs de risque de gastroentérite aigüe : consommation de l’eau municipale, contact avec quelqu’un atteint de gastroentérite aiguë (dans et hors de la maisonnée), contact avec un enfant en garderie et avoir moins de 35 ans. Le fait de boire de l’eau de la municipalité non traitée a été le facteur ayant le rapport de risque le plus élevé (RR = 24,31; IC 95% = 1,50-393,4). Boire de l’eau d’un puit artésien privé a été un facteur protecteur (RR = 0,28; IC 95% = 0,09-0,90).
Conclusion
Cette étude a mis en évidence que la gestion des risques associés à la consommation de l’eau non traitée reste un défi important de santé publique, particulièrement pour les petites municipalités rurales où il existe une vulnérabilité liée aux variations climatiques.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The World Health Organization considers Campylobacter infections as the most common bacterial cause of acute gastroenteritis (AGE) worldwide (World Health Organization 2018). In Canada, it is the most commonly reported enteric disease. The peak incidence of reported cases is in July (Government of Canada 2012; Ministère de la Santé et des Services Sociaux, 2018a, b; Public Health Agency of Canada 2018; Thomas et al. 2017). In the province of Quebec, for the period 2012–2016, the average annual incidence was 33.6 cases per 100,000 persons (Ministère de la Santé et des Services Sociaux 2018c). However, outside of and to a lesser extent during an outbreak, it is assumed that cases are either underreported or underdiagnosed. In fact, it has been estimated that for every domestically acquired laboratory-confirmed case of campylobacteriosis, there are between 18 and 39 cases in the community (Thomas et al. 2013).
The most common reservoirs of Campylobacter are poultry and cattle. Humans can also be reservoirs for other humans, but due to the chronicity of infection in animal reservoirs, the main source of infection is zoonotic (Lévesque et al. 2013; Michaud et al. 2004). Although the outdoor survival time of Campylobacter appears limited, human infections are favoured by a low infectious dose required to cause the disease (Ministère de la Santé et des Services Sociaux 2018a, b, c). Sporadic cases are often associated with the ingestion of contaminated food (poultry) whereas outbreaks are generally associated with the consumption of unpasteurized raw milk or dairy products or contaminated (potable or recreational) water that is often unchlorinated (Centers for Disease Control and Prevention 2018; Government of Canada 2012; Heymann 2015).
This article presents the results of an investigation related to an AGE outbreak that occurred in the summer of 2018 in a small rural municipality (municipality A) located in the province of Quebec. The outbreak was identified by an unusual number of campylobacteriosis cases reported to the Regional Public Health Department (RPHD). The first investigations suggested a waterborne transmission of the infection. However, as the water tests did not reveal the presence of Campylobacter, a decision was made to conduct a more comprehensive epidemiological investigation to identify the source of the outbreak, ensure control, and prevent future relapses.
Methods
Context of the investigation
Between the 11 and 23 of July 2018, nine cases of Campylobacter jejuni infection were reported to the RPHD by the provincial laboratory (Laboratoire de santé publique du Québec). Eight of the nine cases came from municipality A which has 2320 inhabitants.
The outbreak was confirmed because the incidence rate of campylobacteriosis in July 2018 was found to be eight times higher than the average rate observed in the last 18 years (395.8 versus 48.3 cases per 100,000 inhabitants between 2000 and 2017). Moreover, the pharmacist from the only pharmacy in municipality A attested to counseling more than 40 clients suffering from AGE between mid-June and July 23. This information was confirmed by the Health Info Line database, where multiple calls for AGE-like symptoms from residents of municipality A were recorded. All risk factors for AGE were explored during the interviews with the first reported cases of campylobacteriosis. Because the only commonality between the reported cases was the water supply (cases reported having observed turbidity and change in taste of the drinking water), the RPHD suspected a waterborne outbreak. Also, eight of the nine first cases were concentrated close to the drinking water reservoir. The drinking water distribution system in this community serves a population of 800 people in the downtown sector (an urban-agro-industrial area) and is supplied by unfiltered and unchlorinated groundwater (five wells and one main surface water tank). The rest of the population gets their water from private artesian wells. Therefore, on July 17, 2018, a boil-water advisory was issued for municipality A.
Study design and case definitions
A population-based retrospective cohort study was designed to follow all the subjects who could have been exposed to a given factor (water, food, outdoor risk factors such as social events in the municipality, presence of manure, wild or farm animals near the house) in order to evaluate its effects on the development of AGE. Because laboratory testing was not available for the cohort population and given the fact that AGE can be caused by different pathogenic agents, the probable case definition for AGE was based on the signs and symptoms of the first reported campylobacteriosis cases. A case was defined as a resident (visitors were excluded) present in municipality A between June 23 and July 22, 2018, having reported acute gastrointestinal clinical manifestations with at least two of the following signs or symptoms: diarrhea, abdominal cramps, fever, nausea, and/or vomiting.
Study population and sampling
The required size of the random sample of the cohort was calculated at 114 subjects. The parameters used were as follows: alpha = 0.05, power 90%, ratio of sample size, unexposed/exposed = 1, and risk ratio (RR) = 5.5 (Sullivan and Soe 2007). With an expected response rate of 56% (based on a similar outbreak in Sweden by Martin et al. 2006), it was adjusted to 238 subjects. Random sampling was done using the ALEA function (Microsoft Excel 2013) from the electoral roll provided by municipality A. The names and addresses of the selected subjects were then matched with the landline numbers in the telephone directory of residents of the municipality. Five random lists were produced to reserve the option of replacing selected residents due to absence and non-response to all three call attempts or in cases where non-residential locations were selected. The replacements had to be immediate or close neighbours to the address of the chosen resident’s house. In order to question individuals from all age groups, interviewers asked to speak with the resident who most recently celebrated their birthday. For individuals less than 14 years of age, their parents or legal guardians answered on their behalf.
Collection of data
The data were collected by previously trained telephone interviewers between August 15 and 17. A Quebec standard water and foodborne disease outbreak questionnaire was reviewed by the investigation team.
Considering that AGE cases are not mandatory reportable diseases in Quebec, we collected indirect surveillance data of these illness events from two sources: the register of calls from the Health Info Line and the sales register of antidiarrheal products from the only pharmacy located in the municipality. The observation period for the pharmacy data was from May 27 to August 18, 2018. For the Health Info Line calls, the observation period was extended until October 6, 2018. This information was used as a proxy to determine the end of the outbreak.
Assessment of the quality of the water was conducted by Quebec’s Ministère de l’Agriculture, des Pêcheries et de l’Alimentation and the Ministère de l’Environnement et de la Lutte contre les changements climatiques. It was performed, between July 18 and September 12, on raw underground water from the five wells in operation as well as on 56 samples of water distributed to various locations in municipality A.
Data analysis
Representativeness of the study sample was validated according to the demographic characteristics and the urban development of the municipality and geolocalization of the selected households using Geographic Information System software (QGIS). Descriptive statistics were reported for the categorical and the continuous variables. The evolution of the indirect surveillance data was compared with the epidemic curve plotted for all observed cases of AGE (campylobacteriosis cases and probable AGE cases from the cohort). The historical moving average of Info-Santé calls over the last 5 years was calculated to draw comparative curves with the evolution of calls observed in 2018.
Bivariate analyses were conducted for all the exposure variables studied. In addition, to verify whether there was a difference in risk according to the geographical distribution of cases observed during the study period, municipality A was delineated into three geographic areas (Fig. 1): urban-agro-industrial (Zone 1), urban (Zone 2), and rural (Zone 3). For each zone, the estimated attack rates were calculated.
Ethical considerations
This investigation was conducted in accordance with the legal mandate granted to public health authorities by Quebec’s Public Health Act (R.S.Q., chapter S-2.2. Article 1. Updated July 1, 2018). The authors who were part of the investigation team were legally appointed by the director of public health of the affected region to act under this mandate that endorses the main principles of the Tri-Council Policy Statement: Ethical Conduct for Research Involving Humans (Canadian Institutes of Health Research 2018). The study did not directly involve subjects but rather the events that happened to them and these were reported by the people who gave their oral informed consent to participate in the investigation at the time of the interviewer’s phone call. Participants had the right to refuse to answer any questions and to abandon the investigation at any time. Parents or legal guardians gave consent for children under the age of 14 years.
All data were treated confidentially and analyzed without nominal identification. Authorized personnel with access to the databank were bound by oaths to comply with the confidentiality standards. Data and measures relating to manipulation, analysis, storage, and eventual destruction have been and will be processed according to the Institut national de santé publique du Québec information, protection, and security policy (PO-04-2014).
Results
Response rate and representativeness of the cohort
One hundred and forty selected individuals agreed to participate in the investigation. This represents a response rate of 58.8%. The cohort retained one of the reported cases of campylobacteriosis and one known case of AGE that was epidemiologically linked to a confirmed case of C. jejuni infection, as their telephone numbers were part of the random recruitment list.
The age of individuals in the cohort ranged from 0.1 to 87.8 years. The average and median age values of the cohort were 45.9 and 52.5 years, respectively. Table 1 shows the demographic characteristics of the study cohort and those of the population of municipality A. A higher proportion of individuals in the cohort were older, had completed high school as well as university, and were retired or stay at home as compared with the population of municipality A (mid-p exact, two-tailed p value < 0.05). However, no significant differences were observed when comparing sex and number of persons in the household.
Maps created by QGIS showed that the geographical distribution of the individuals’ houses in the cohort was comparable with that of the residents of municipality A. Figure 1 shows the distribution of cases and non-cases observed after zoning the municipality as well as the location of the five subterranean water wells, the water tank, and the locations where the environmental tests were positive.
Illness and symptoms
A total of 22 individuals in the study cohort met the case definition for AGE illness; this represents an illness attack rate of 15.7%. Table 2 shows the frequency of symptoms reported by AGE cases as well as the campylobacteriosis cases in the cohort. Campylobacteriosis cases were more severe in their presentation, where more cases presented fever and vomiting, consulted a health professional (this included nurses through the Health Info Line), or were hospitalized.
Epidemic curve
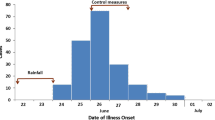
Figure 2 illustrates the weekly distribution of cohort’s AGE and campylobacteriosis cases according to the date of onset of their symptoms, the number of antidiarrheal products sold between May 27 and August 12, and the number of calls made to the Health Info Line from May 27 to October 6. These four parameters increased as of June 17 and peaked in the weeks of July 8 and 15. During these 2 weeks, there was a significant increase in the number of calls made to the Health Info Line as compared with the historical moving average calculated for the last 5 years, where 6 calls were reported for July 8 and 5 calls for July 15 versus 1.3 (95% CI = 0–3.6) and 1.1 (95% CI = 0–3.3), respectively.
Exposure variables
Table 3 shows, for the total sample, that persons less than 35 years of age, who had contact with a person with AGE within the household or outside, and with a child in daycare were associated with an increased risk of AGE. However, consumption of municipal water produced the highest risk ratio (RR = 24.31; 95% CI = 1.50–393.4). Of the 87 residents who had consumed municipal water during the study period, 21 (24.1%) developed AGE compared with 1 of the 51 residents who did not consume municipal water (2.0%). Although not significant, this increased risk was also observed for those who exclusively drank municipal water (RR = 17.0; 95% CI = 0.92–314.9). The risk of AGE was reduced by almost 70% for the individuals who drank water from a private artesian well (RR = 0.28, 95% CI 0.09–0.90). No other waterborne exposures (recreational water) were associated with an increased risk of AGE. As for the foodborne exposures, no difference in risk was observed.
Table 4 shows the results of the attack rates and the risk evaluation (risk ratio) by zone of residence. Rural Zone 3 is not connected to the municipal water distribution system and shows the lowest attack rate (10.8%) followed by urban Zone 2 (22.7%) and urban-industrial Zone 1 (31.6%). The last two are connected to the municipal water distribution system. Using Zone 3 as a constant, the risk of developing an AGE was almost four times higher among residents of Zone 1 and more than double among residents of Zone 2 (p = 0.01).
Environmental results
Escherichia coli and/or enterococci, as well as total coliforms, were detected in 6 of the 10 water samples taken between July 26 and 28 (CDC week 30). Five of the 6 contaminated samples were located on the distribution system near the main water tank. The 40 samples taken subsequently, from August 8 to September 12, did not reveal any fecal contamination. Campylobacter was not detected in any sample of the distributed or raw water supply. The epidemiological investigation showed that 5 days of rain (between 7.8 and 19 mm/day) and heat waves (between 30.5 and 33.6 °C/day for four consecutive days) were observed 10 days preceding the peak of the outbreak.
Discussion
Drinking water from the municipal system was the factor with the highest relative risk, followed by secondary contacts with another AGE. None of the potential food sources was associated with the occurrence of AGE, which gives additional importance to the water source and other identified risk factors. Multivariate analyses, and particularly the final regression model, identified drinking municipal water as the most important independent predictor of occurrence of an episode of AGE. Concerning the two other independent risk factors, although secondary cases may have occurred as a result of contact with a sick person, this does not exclude exposure to a common water source, as it can be assumed that all members of a household or neighbourhood consume the same water.
The environmental and microbiological analyses showed no type of bacterial contamination in the municipality’s groundwater wells and C. jejuni was not detected in any environmental samples obtained after confirmation of the outbreak. This can be explained by the tedious nature of these bacteria; they are able to adopt a viable but non-culturable (VNC) state, especially in water. It is also possible that the sampling was carried out outside the period of contamination with C. jejuni. However, an unusually high count of bacterial contamination indicators was identified in samples from the municipality’s main water tank and nearby distribution system (a chicken coop, located about 300 m from the tank). A report issued by a regional representative of the Ministry of Environment states that cleaning of the network through hydrant flushing was performed between June 19 and 26 in that area, just a couple of days prior to the outbreak. The report suggests that the significant decrease in pressure could have allowed an intrusion of contaminated water in the network through one of the service buildings. In fact, inspections found deficiencies in several buildings in this area, such as the lack of backflow valves at their drinking water intake. These facts highlight the vulnerability of the water distribution system and the main municipal tank to microbiological contamination and may explain the observed cases of campylobacteriosis and AGE.
Indirect surveillance data showed that AGE cases had been reported since June 24 (week CDC 26). With an overall observed attack rate of 15.7% in the cohort, it can be estimated that there were 364 people in this municipality with clinical manifestations of AGE. However, because of the significantly older age of the study cohort as compared with the population of municipality A, this risk can be an underestimation of the real risk of developing AGE. Regardless of whether it is caused by a single or multiple agents, AGE (including Campylobacter infection) is more common in younger age groups (Thomas et al. 2013).
Those who had consumed water from a private artesian well had 72% less risk of developing an AGE. This could be explained by the fact that the individuals exposed to this factor resided in the rural sector of the municipality without direct access to the city’s unchlorinated water. Similar observations have been reported in a rural population in Finland (Kuusi et al. 2004).
The attack rates observed in the three delineated areas of the municipality suggest that the place with the highest risk is located in urban-agro-industrial Zone 1, which, according to the revised QGIS maps, would be about 10 m below the level of the neighbouring urban area, which could lead to contaminants trickling downhill. In addition, this zone houses the main water tank, the chicken coop, agricultural industries, and manure storage tanks. The latter two exposure factors have already been reported to be associated with outbreaks of campylobacteriosis or other causes of AGE (Government of Canada 2014; Louis et al. 2005).
The presence of surrounding agricultural land as well as the characteristics of Zone 1 provide a favourable context in which the heavy rainfall and heat waves observed could have helped create a source of contaminated water exposure to the population. Several studies have explored the link between climate change and the incidence of AGE and campylobacteriosis (Curriero et al. 2001; Nichols et al. 2009; Thomas et al. 2006; Thomas et al. 2013). Nichols and colleagues found a significant association between waterborne outbreaks of AGE and cumulative precipitation over the previous 7 days, as well as a significant association with very high rainfall levels (Nichols et al. 2009). Studies using predictive models have found a correlation between maximum ambient temperature and the incidence of human cases of campylobacteriosis (Patrick et al. 2004; Yun et al. 2016). In England and Wales, Louis and colleagues have studied the precipitation threshold, the daily maximum and minimum temperatures, and the number of hours of natural light. Increased temperature was associated with increased incidence of campylobacteriosis (p < 0.001). This study also suggests a correlation between the incidence of campylobacteriosis, the agent’s life cycle, and agricultural activities (Louis et al. 2005).
Among the control measures taken, a boil-water advisory was issued, which was respected by the majority of the municipality’s population (80.1% in the cohort). The public health communication to the population as well as to the various stakeholders was maintained throughout this epidemic situation. The main water tank was emptied and disinfected from September 8 to 10 and corrective measures in certain sectors of the network were introduced. These measures may have substantially contributed to containing the outbreak. No new cases were reported until the extended date of the observation period (October 6, CDC week 40).
Strengths and limitations
The study had several strengths, including the representativeness of the sample and a higher than expected response rate, especially considering the summer season. In addition, the interviews were conducted shortly after the occurrence of the cases, thus decreasing a potential recall bias among the respondents. This study also showed that indirect surveillance data can provide valuable information in an outbreak investigation. Nevertheless, this study is not without its limitations. One can argue that the use of AGE as our case definition was more sensitive than using campylobacter. However, considering laboratory testing was not available for the cohort population, we cannot exclude the possibility of other microbial agents present in the drinking water. In fact, there are reports published elsewhere describing multipathogen or concomitant waterborne outbreaks (Gallay et al. 2006; Martin et al. 2006). Certain variables regarding quantity of water ingested with respect to a unique water source were missing, not allowing us to establish a dose-response relationship. Additionally, considering the attention this outbreak received from the media, information bias could have been introduced. However, this was likely limited, as sample characteristics demonstrated that our estimates were conservative.
Conclusion
This study provided a good estimate of the extent of the outbreak and its associated factors. It also highlighted that managing the risks associated with the consumption of untreated drinking water remains an important public health challenge, particularly in small rural municipalities vulnerable to climate variability. Considering the fact that numerous institutions and teams were involved in the investigation and control of this outbreak, our study also highlights the importance of communication between these stakeholders, leading to a concerted and efficient—albeit retroactive—outbreak response.
References
Canadian Institutes of Health Research. (2018). Tri-Council policy statement: ethical conduct for research involving humans. Retrieved from http://publications.gc.ca/collections/collection_2019/irsc-cihr/RR4-2-2019-eng.pdf. Accessed 9 Dec 2019.
Centers for Disease Control and Prevention. (2018). Information for health professionals | Campylobacter | CDC. Retrieved October 29, 2018, from https://www.cdc.gov/campylobacter/technical.html. Accessed 29 Oct 2018.
Curriero, F. C., Patz, J. A., Rose, J. B., & Lele, S. (2001). The association between extreme precipitation and waterborne disease outbreaks in the United States, 1948-1994. American Journal of Public Health, 91(8), 1194–1199. https://doi.org/10.2105/ajph.91.8.1194.
Gallay, A., De Valk, H., Cournot, M., Ladeuil, B., Hemery, C., Castor, C., Bon, F., Mégraud, F., Le Cann, P., Desenclos, J. C., & Outbreak Investigation Team. (2006). A large multi-pathogen waterborne community outbreak linked to faecal contamination of a groundwater system, France, 2000. Clinical Microbiology and Infection: The Official Publication of the European Society of Clinical Microbiology and Infectious Diseases, 12(6), 561–570. https://doi.org/10.1111/j.1469-0691.2006.01441.x.
Government of Canada. (2012). Campylobacter jejuni [education and sensibilisation]. Retrieved November 2, 2018, from Canada.ca website: https://www.canada.ca/en/public-health/services/food-poisoning/campylobacter-jejuni.html
Government of Canada. (2014). Agriculture et la qualité de l’eau—Agriculture et Agroalimentaire Canada (AAC). Retrieved November 19, 2018, from http://www.agr.gc.ca/fra/science-et-innovation/pratiques-agricoles/eau/protection-des-bassins-hydrographiques/agriculture-et-la-qualite-de-leau/?id=1371491033072
Heymann, D. L. (2015). Control of communicable diseases manual (20e ed.). Retrieved from https://www.apha.org/ccdm. Accessed 26 Jul 2019.
Kuusi, M., Klemets, P., Miettinen, I., Laaksonen, I., Sarkkinen, H., Hänninen, M. L., Rautelin, H., Kela, E., & Nuorti, J. P. (2004). An outbreak of gastroenteritis from a non-chlorinated community water supply. Journal of Epidemiology and Community Health, 58(4), 273–277. https://doi.org/10.1136/jech.2003.009928.
Lévesque, S., Fournier, E., Carrier, N., Frost, E., Arbeit, R. D., & Michaud, S. (2013). Campylobacteriosis in urban versus rural areas: a case-case study integrated with molecular typing to validate risk factors and to attribute sources of infection. PLoS One, 8(12), e83731. https://doi.org/10.1371/journal.pone.0083731.
Louis, V. R., Gillespie, I. A., O’Brien, S. J., Russek-Cohen, E., Pearson, A. D., & Colwell, R. R. (2005). Temperature-driven campylobacter seasonality in England and Wales. Applied and Environmental Microbiology, 71(1), 85–92. https://doi.org/10.1128/AEM.71.1.85-92.2005.
Martin, S., Penttinen, P., Hedin, G., Ljungström, M., Allestam, G., Andersson, Y., & Giesecke, J. (2006). A case-cohort study to investigate concomitant waterborne outbreaks of Campylobacter and gastroenteritis in Söderhamn, Sweden, 2002-3. Journal of Water and Health, 4(4), 417–424 Retrieved from https://www.ncbi.nlm.nih.gov/pubmed/17176812.
Michaud, S., Ménard, S., & Arbeit, R. D. (2004). Campylobacteriosis, eastern townships, Quebec. Emerging Infectious Diseases, 10(10), 1844–1847. https://doi.org/10.3201/eid1010.040228.
Ministère de la Santé et des Services Sociaux. (2018a). Campylobactériose—Professionnels de la santé—MSSS. Retrieved November 2, 2018, from http://www.msss.gouv.qc.ca/professionnels/zoonoses/campylobacteriose/
Ministère de la Santé et des Services Sociaux. (2018b). Vigie des menaces à la santé de la population. Vigie-Interventions., 13(2), 6 Retrieved from http://publications.msss.gouv.qc.ca/msss/fichiers/flashvigie/FlashVigie_vol13_no2.pdf.
Ministère de la Santé et des Services Sociaux. (2018c). Vigie et surveillance des maladies à déclaration obligatoire d’origine infectieuse—Rapport annuel 2017. Retrieved from http://publications.msss.gouv.qc.ca/msss/document-002235/?&txt=rapport%20annuel&msss_valpub&date=DESC. Accessed 24 May 2019.
Nichols, G., Lane, C., Asgari, N., Verlander, N. Q., & Charlett, A. (2009). Rainfall and outbreaks of drinking water related disease and in England and Wales. Journal of Water and Health, 7(1), 1–8. https://doi.org/10.2166/wh.2009.143.
Patrick, M. E., Christiansen, L. E., Wainø, M., Ethelberg, S., Madsen, H., & Wegener, H. C. (2004). Effects of climate on incidence of Campylobacter spp. in humans and prevalence in broiler flocks in Denmark. Applied and Environmental Microbiology, 70(12), 7474–7480. https://doi.org/10.1128/AEM.70.12.7474-7480.2004.
Public Health Agency of Canada. (2018). FoodNet Canada annual report 2017 [Transparency - other]. Retrieved from https://www.canada.ca/en/public-health/services/surveillance/foodnet-canada/publications/foodnet-canada-annual-report-2017.html. Accessed 24 May 2019.
Sullivan, M. K., & Soe, M. M. (2007). Sample size for cross-sectional, cohort, or clinical trial studies. Retrieved from https://www.openepi.com/PDFDocs/SSCohortDoc.pdf. Accessed 29 Mar 2019.
Thomas, K. M., Charron, D. F., Waltner-Toews, D., Schuster, C., Maarouf, A. R., & Holt, J. D. (2006). A role of high impact weather events in waterborne disease outbreaks in Canada, 1975—2001. International Journal of Environmental Health Research, 16(3), 167–180. https://doi.org/10.1080/09603120600641326.
Thomas, K. M., Murray, R., Flockhart, L., Pintar, K., Pollari, F., Fazil, A., Nesbitt, A., & Marshall, B. (2013). Estimates of the burden of foodborne illness in Canada for 30 specified pathogens and unspecified agents, circa 2006. Foodborne Pathogens and Disease, 10(7), 639–648 Retrieved from https://www.ncbi.nlm.nih.gov/pubmed/23659355.
Thomas, K. M., Murray, R., Nesbitt, A., & Pollari, F. (2017). The incidence of acute gastrointestinal illness in Canada, foodbook survey 2014-2015. The Canadian Journal of Infectious Diseases & Medical Microbiology = Journal Canadien Des Maladies Infectieuses Et De La Microbiologie Medicale, 2017, 5956148. https://doi.org/10.1155/2017/5956148.
World Health Organization. (2018). Campylobacter. Retrieved November 2, 2018, from World Health Organization website: https://www.who.int/news-room/fact-sheets/detail/campylobacter
Yun, J., Greiner, M., Höller, C., Messelhäusser, U., Rampp, A., & Klein, G. (2016). Association between the ambient temperature and the occurrence of human Salmonella and Campylobacter infections. Scientific Reports, 6, 28442. https://doi.org/10.1038/srep28442.
Acknowledgements
The authors acknowledge the assistance and support of all of the persons involved in the management of this outbreak investigation, including Dr. Patrick Levallois who collaborated in our study as a senior environmental health advisor. A special thanks to the staff at the Direction de santé publique de Chaudière-Appalaches and the Institut national de santé publique du Québec.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This investigation was conducted in accordance with the legal mandate granted to public health authorities by Quebec’s Public Health Act (R.S.Q., chapter S-2.2. Article 1. Updated July 1, 2018). The authors who were part of the investigation team were legally appointed by the director of public health of the affected region to act under this mandate that endorses the main principles of the Tri-Council Policy Statement: Ethical Conduct for Research Involving Humans (Canadian Institutes of Health Research 2018). The study did not directly involve subjects but rather the events that happened to them and these were reported by the people who gave their oral informed consent to participate in the investigation at the time of the interviewer’s phone call. Participants had the right to refuse to answer any questions and to abandon the investigation at any time. Parents or legal guardians gave consent for children under the age of 14 years.
All data were treated confidentially and analyzed without nominal identification. Authorized personnel with access to the databank were bound by oaths to comply with the confidentiality standards. Data and measures relating to manipulation, analysis, storage, and eventual destruction have been and will be processed according to the Institut national de santé publique du Québec information, protection, and security policy (PO-04-2014).
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Soto, J.C., Barakat, M., Drolet, MJ. et al. Waterborne outbreaks: a public health concern for rural municipalities with unchlorinated drinking water distribution systems. Can J Public Health 111, 433–442 (2020). https://doi.org/10.17269/s41997-020-00300-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.17269/s41997-020-00300-x