Abstract
Background
Most relapses in melanoma patients occur during the first five years after diagnosis. Identifying characteristics associated with recurrence after this period could help delineate guidelines, specifically for follow-up protocols.
Objectives
The aim of this study was to identify the prognostic factors for relapse and death caused by melanoma in patients who have been disease-free for five years.
Materials & Methods
We designed a longitudinal retrospective cohort to study Stage I/II cutaneous melanoma patients who have been free of disease for more than five years (late relapse cohort). Prognostic factors for disease-free and melanoma-specific survival were evaluated using the Kaplan-Meier method and Cox regression models.
Results
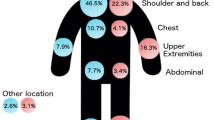
A series of 746 patients who had Stage I-II cutaneous melanoma and were free of disease for five years was selected. After a median follow-up of 64 months (124 months since melanoma diagnosis), 51 (6.8%) patients relapsed and 18 (2.4%) died from melanoma. Acral location and presence of ulceration, as well as intermediate growth rate (0.11–0.50 mm/month), were significantly associated with relapse or death due to melanoma. The initial recurrence site was associated with distant metastasis in 48% of the cases.
Conclusion
In this study, we have identified melanoma characteristics in patients who have been disease-free for five years that may allow us to establish groups at increased risk of relapse or death due to melanoma, which could be helpful for melanoma management.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Tejera-Vaquerizo A, Descalzo-Gallego MA, Otero-Rivas MM, et al. Skin cancer incidence and mortality in spain: a systematic review and meta-analysis. Actas Dermosifiliogr 2016; 107: 318–28.
Arnold M, Holterhues C, Hollestein LM, et al. Trends in incidence and predictions of cutaneous melanoma across Europe up to 2015. J Eur Acad Dermatol Venereol 2014; 28: 1170–8.
Balch CM, Soong SJ, Gershenwald JE, et al. Prognostic factors analysis of 17,600 melanoma patients: validation of the American Joint Committee on Cancer melanoma staging system. J Clin Oncol 2001; 19: 3622–34.
Tejera-Vaquerizo A, Solis-Garcia E, Rios-Martin JJ, Moreno-Ramirez D. Primary cutaneous melanoma: prognostic factors not included in the classification of the American Joint Committee on Cancer. Actas Dermo-Sifiliograficas 2011; 102: 255–63.
Nagore E, Heidenreich B, Rachakonda S, et al. TERT promoter mutations in melanoma survival. Int J Cancer 2016; 139: 75–84.
Thomas NE, Edmiston SN, Alexander A, et al. Association between NRAS and BRAF mutational status and melanoma-specific survival among patients with higher-risk primary melanoma. JAMA Oncology 2015; 1: 35948.
Moreau S, Saiag P, Aegerter P, et al. Prognostic value of BRAF(V(6)(0)(0)) mutations in melanoma patients after resection of metastatic lymph nodes. Annal Surg Oncol 2012; 19: 4314–21.
Nagore E, Requena C, Traves V, et al. Prognostic value of BRAF mutations in localized cutaneous melanoma. J Am Acad Dermatol 2014; 70: 85842e1–2.
Liu W, Dowling JP, Murray WK, et al. Rate of growth in melanomas: characteristics and associations of rapidly growing melanomas. Arch Dermatol 2006; 142: 1551–8.
Nagore E, Martorell-Calatayud A, Botella-Estrada R, Guillen C. Growth rate as an independent prognostic factor in localized invasive cutaneous melanoma. J Eur Acad Dermatol Venereol 2011; 25: 618–20.
Tejera-Vaquerizo A, Barrera-Vigo MV, Lopez-Navarro N, Herrera-Ceballos E. Growth rate as a prognostic factor in localized invasive cutaneous melanoma. J Eur Acad Dermatol Venereol 2010; 24: 147–54.
Tejera-Vaquerizo A, Nagore E, Herrera-Acosta E, et al. Prediction of sentinel lymph node positivity by growth rate of cutaneous melanoma. Arch Dermatol 2012; 148: 577–84.
Tejera-Vaquerizo A, Nagore E, Melendez JJ, et al. Chronology of metastasis in cutaneous melanoma: growth rate model. J Invest Dermatol 2012; 132: 1215–21.
Bichakjian CK, Halpern AC, Johnson TM, et al. Guidelines of care for the management of primary cutaneous melanoma. American Academy of Dermatology. J Am Acad Dermatol 2011; 65: 1032–47.
Dummer R, Hauschild A, Lindenblatt N, Pentheroudakis G, Keilholz U, Committee EG. Cutaneous melanoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Annal Oncol 2015; 26: v126–32.
Garbe C, Peris K, Hauschild A, et al. Diagnosis and treatment of melanoma. European consensus-based interdisciplinary guideline — Update 2016. Eur J Cancer 2016; 63: 201–17.
Marsden JR, Newton-Bishop JA, Burrows L, et al. Revised UK guidelines for the management of cutaneous melanoma 2010. Br J Dermatol 2010; 163: 238–56.
Crowley NJ, Seigler HF. Late recurrence of malignant melanoma. Analysis of 168 patients. Annal Surg 1990; 212: 173–7.
Osella-Abate S, Ribero S, Sanlorenzo M, et al. Risk factors related to late metastases in 1,372 melanoma patients disease free more than 10 years. Int J Cancer 2015; 136: 2453–7.
Tseng WW, Fadaki N, Leong SP. Metastatic tumor dormancy in cutaneous melanoma: does surgery induce escape? Cancers 2011; 3: 730–46.
von Elm E, Altman DG, Egger M, et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: guidelines for reporting observational studies. Int J Surg 2014; 12: 1495–9.
Tejera-Vaquerizo A, Perez-Cabello G, Marinez-Leborans L, et al. Is mitotic rate still useful in the management of patients with thin melanoma? J Eur Acad Dermatol Venereol 2017; 12: 2025–9.
Garcia-Casado Z, Traves V, Banuls J, et al. BRAF, NRAS and MC1R status in a prospective series of primary cutaneous melanoma. Br J Dermatol 2015; 172: 1128–31.
Qu K, Pan Q, Zhang X, et al. Detection of BRAF V600 mutations in metastatic melanoma: comparison of the Cobas 4800 and Sanger sequencing assays. J Mol Diagn 2013; 15: 790–5.
Grob JJ, Richard MA, Gouvernet J, et al. The kinetics of the visible growth of a primary melanoma reflects the tumor aggressiveness and is an independent prognostic marker: a prospective study. Int J Cancer 2002; 102: 34–8.
Rubin DB. Multiple Imputation for Nonresponse Surveys. 1st Ed. New York, USA: John Wiley & Sons, Inc., 1987.
Brauer JA, Wriston CC, Troxel AB, et al. Characteristics associated with early and late melanoma metastases. Cancer 2010; 116: 415–23.
Hansel G, Schonlebe J, Haroske G, Wollina U. Late recurrence (10 years or more) of malignant melanoma in south-east Germany (Saxony). A single-centre analysis of 1881 patients with a follow-up of 10 years or more. J Eur Acad Dermatol Venereol 2010; 24: 833–6.
Schmid-Wendtner MH, Baumert J, Schmidt M, et al. Late metastases of cutaneous melanoma: an analysis of 31 patients. J Am Acad Dermatol 2000; 43: 605–9.
Faries MB, Steen S, Ye X, Sim M, Morton DL. Late recurrence in melanoma: clinical implications of lost dormancy. J Am Coll Surg 2013; 217: 27–34.
Tsao H, Cosimi AB, Sober AJ. Ultra-late recurrence (15 years or longer) of cutaneous melanoma. Cancer 1997; 79: 2361–70.
Martorell-Calatayud A, Nagore E, Botella-Estrada R, et al. Defining fast-growing melanomas: reappraisal of epidemiological, clinical, and histological features. Melanoma Res 2011; 21: 131–8.
Acknowledgments and disclosures
Acknowledgments: this study was supported partially by grant 2017-109-001 from the Universidad Católica de València. Conflicts of interest: none.
Author information
Authors and Affiliations
Corresponding author
Supplementary material
About this article
Cite this article
Bancalari, B., Ferrer-Guillen, S., Tejera-Vaquerizo, A. et al. Longitudinal study of prognostic factors for localized cutaneous melanoma in patients who have been disease-free for five years. Eur J Dermatol 31, 192–198 (2021). https://doi.org/10.1684/ejd.2021.4022
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1684/ejd.2021.4022