Abstract
Background
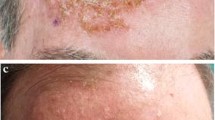
The use of ingenol mebutate (IM) as a field-directed therapy over a short period of time has been shown to be effective and well tolerated in randomized Phase III trials.
Objectives
To assess the efficacy and patient-reported outcomes for IM as treatment for actinic keratosis (AK) under daily “real-life” practice conditions.
Materials and Methods
A total of 826 adult patients with AK were enrolled by 292 dermatologists in Germany in a prospective, open, non-interventional, non-controlled, multicentre study. All patients were treated with IM and followed for eight weeks.
Results
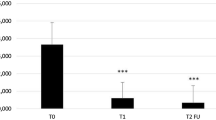
The mean number of clinically visible AK lesions decreased significantly from 7.1±6.8 to 2.8±4.5 (p<0.0001). Most dermatologists (79.0%) rated global efficacy of IM as “very good”/“good” and 82.6% of the patients were “very satisfied” or “rather satisfied” with the efficacy of IM. Patient-reported outcomes showed greater efficacy and treatment comfort with IM compared to any last previous AK treatment with a comparable tolerability profile. Skin-related QoL data revealed a significant improvement of 50.2% after IM treatment (p<0.0001). Adverse events were reported in 7.0% of all patients, which were in most cases mild in intensity.
Conclusion
Field-directed treatment with IM over a short period was associated with a high level of treatment satisfaction, as reported by dermatologists and patients. This observational study demonstrates the effectiveness and tolerability of IM in everyday clinical practice in addition to the known efficacy and safety obtained by randomized controlled clinical trials.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Werner RN, Stockfleth E, Connolly SM, et al. International League of Dermatological Societies; European Dermatology Forum. Evidenceand consensus-based (S3) Guidelines for the Treatment of Actinic Keratosis - International League of Dermatological Societies (ILDS) in cooperation with the European Dermatology Forum (EDF) - Long version (online supplement). J Eur Acad Dermatol Venereol 2015. Available at: https://doi.org/10.1111/jdv.13180.
Glogau RG. The risk of progression to invasive disease. J Am Acad Dermatol 2000; 42: 23–4.
Werner RN, Sammain A, Erdmann R, Hartmann V, Stockfleth E, Nast A. The natural history of actinic keratosis: a systematic review. Br J Dermatol 2013; 169: 502–18.
Poulin Y, Lynde CW, Barber K, et al. Canadian non-Melanoma Skin Cancer Guidelines Committee. Non-melanoma Skin Cancer in Canada. Chapter 3: Management of Actinic Keratoses. J Cutan Med Surg 2015; 19: 227–38.
Stockfleth E, Ortonne J-P, Alomar A. Actinic keratosis and field cancerisation. Eur J Dermatol 2011; 21: 3–12.
de Berker D, McGregor JM, Mohd Mustapa MF, Exton LS, Hughes BR. British Association of Dermatologists’ guidelines for the care of patients with actinic keratosis 2017. Br J Dermatol 2017; 176: 20–43.
Campione E, Ventura A, Diluvio L, et al. Current developments in pharmacotherapy for actinic keratosis. Expert Opin Pharmacother 2018; 19: 1693–704.
Thomas GJ, Herranz P, Balta Cruz S, Parodi A. Treatment of actinic keratosis through inhibition of cyclooxygenase-2: potential mechanism of action of diclofenac sodium 3% in hyaluronic acid 2.5. Dermatol Ther 2018: e12800.
Picato® (ingenol mebutate) gel, 0.015%, 0.05%. Summary of Product Characteristics. Revised: 06/2017.
Stockfleth E, Bastian M. Pharmacokinetic and pharmacodynamic evaluation of ingenol mebutate for the treatment of actinic keratosis. Expert Opin Drug Metab Toxicol 2018; 14: 911–8.
Lebwohl M, Swanson N, Anderson LL, Melgaard A, Xu Z, Berman B. Ingenol mebutate gel for actinic keratosis. N Engl J Med 2012; 366: 1010–9.
Ricci F, Tambone S, Neri L, et al. Real-life efficacy and safety of ingenol mebutate for the treatment of actinic keratosis of the face and scalp: a single arm retrospective study. J Dermatolog Treat 2016; 27: 525–30.
Chren MM, Lasek RJ, Sahay AP, Sands LP. Measurement properties of Skindex-16: a brief quality-of-life measure for patients with skin diseases. J Cutan Med Surg 2001; 5: 105–10.
Chren MM. Skindex (Skindex/Skindex-29/Skindex-16), distributed by Mapi Research Trust. Available at: https://eprovide.mapitrust.org/instruments/skindex.
Kurth BM, Hense HW. Leitlinien und Empfehlungen zur Sicherung von Guter Epidemiologischer Praxis (GEP). Arbeitsgruppe Epidemiologische Methoden der Deutschen Arbeitsgemeinschaft fu¨r Epidemiologie (DAE) in Zusammenarbeit mit der Deutschen Gesellschaft fu¨r Medizinische Informatik, Biometrie und Epidemiologie (GMDS), Deutschen Gesellschaft fu¨r Sozialmedizin und Prävention (DGSMP), Deutschen Region der Internationalen Biometrischen Gesellschaft (DR-IBS). Hoffmann (2004). Available at: http://www.gmds.de/publikationen/empfehlungen.php.
Berman B. New developments in the treatment of actinic keratosis: focus on ingenol mebutate gel. Clin Cosmet Investig Dermatol 2012; 5: 111–22.
Berman B, Marmur E, Melgaard A. Three-day topical treatment with ingenol mebutate gel 0.015% for actinic keratoses on the face and scalp: analysis of data pooled from two trials. Poster no. P5623 presented at: Annual Meeting of the American Academy of Dermatology; March 16–20, 2012; San Diego, CA.
Bettencourt MS. Tolerability of ingenol mebutate gel, 0.05%, for treating patients with actinic keratosis on the scalp in a community dermatology practice. J Clin Aesthet Dermatol 2016; 9: 20–4.
Kim YC, Yang JY, Yoon JS, et al. A multicentre, open, investigatorinitiated phase IV clinical trial to evaluate the efficacy and safety of ingenol mebutate gel, 0·015% on the face and scalp, and 0·05% on the trunk and extremities, in Korean patients with actinic keratosis (PERFECT). Br J Dermatol 2018; 179: 836–43.
Keenan CR, Allan RS. Epigenomic drivers of immune dysfunction in aging. Aging Cell 2019; 18: e12878.
Stockfleth E, Harwood CA, Serra-Guillén C, Larsson T, Østerdal ML, Skov T. Phase IV head-to-head randomized controlled trial comparing ingenol mebutate 0·015% gel with diclofenac sodium 3% gel for the treatment of actinic keratosis on the face or scalp. Br J Dermatol 2018; 178: 433–42.
Neri L, Peris K, Longo C, et al. Physician-patient communication and patient-reported outcomes in the actinic keratosis treatment adherence initiative (AK-TRAIN): a multicenter, prospective, real-life study of treatment satisfaction, quality of life and adherence to topical fielddirected therapy for the treatment of actinic keratosis in Italy. J Eur Acad Dermatol Venereol 2019; 33: 93–107.
Acknowledgements
The authors would like to acknowledge Yuri Sankawa for medical writing support that was funded by LEO Pharma in accordance with Good Publications Practice (GPP3) guidelines (http://www.ismpp.org/gpp3).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Acknowledgement and disclosures. Ethics statement: All patients provided written informed consent. The research complies with the guidelines for human studies and animal welfare regulations. Approval of the study protocol was obtained from an independent ethics committee (Freiburger Ethikkommission GmbH International). The study was conducted in accordance with the World Medical Association Declaration of Helsinki, and local ethical and legal requirements, such as the guidelines and recommendations of the German Association of Research-based Pharmaceutical Companies (Verband Forschender Arzneimittelunternehmen, VFA) to improve the quality and transparency of non-interventional studies (NIS), the joint recommendations of the Federal Institute for Drugs and Medical Devices (Bundesinstitut für Arzneimittel und Medizinprodukte, BfArM), and the Paul Ehrlich Institute on planning, conducting and evaluation of post-marketing surveillance studies, as well according to the code of conduct of the Registered Society for Medicinal Products and Cooperation in the Health Sector (Verein Arzneimittel und Kooperation im Gesundheitswesen, AKG e. V.). Financial support: This study was supported by LEO Pharma GmbH, Germany. Conflicts of interest: Thomas Diepgen has received honoraria and travelling expenses as consultant and lecturer for LEO Pharma. Christoph Eicke was commissioned by LEO Pharma GmbH for the management of this study. Mike Bastian is an employee of LEO Pharma GmbH. Data management and statistical analyses were performed by Anfomed GmbH, Möhrendorf, Germany.
About this article
Cite this article
Diepgen, T.L., Eicke, C. & Bastian, M. Ingenol mebutate as topical treatment for actinic keratosis based on a prospective, non-interventional, multicentre study of real-life clinical practice in Germany: efficacy and quality of life. Eur J Dermatol 29, 401–408 (2019). https://doi.org/10.1684/ejd.2019.3603
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1684/ejd.2019.3603