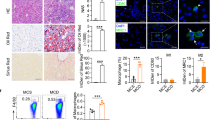
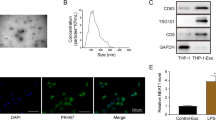
Abstract
An effective therapeutic regimen for hepatic fibrosis requires a deep understanding of the pathogenesis mechanism. Hepatic fibrosis is characterized by activated hepatic stellate cells (aHSCs) with an excessive production of extracellular matrix. Although promoted activation of HSCs by M2 macrophages has been demonstrated, the molecular mechanism involved remains ambiguous. Herein, we propose that the vitamin D receptor (VDR) involved in macrophage polarization may regulate the communication between macrophages and HSCs by changing the functions of exosomes. We confirm that activating the VDR can inhibit the effect of M2 macrophages on HSC activation. The exosomes derived from M2 macrophages can promote HSC activation, while stimulating VDR alters the protein profiles and reverses their roles in M2 macrophage exosomes. Smooth muscle cell-associated protein 5 (SMAP-5) was found to be the key effector protein in promoting HSC activation by regulating autophagy flux. Building on these results, we show that a combined treatment of a VDR agonist and a macrophage-targeted exosomal secretion inhibitor achieves an excellent anti-hepatic fibrosis effect. In this study, we aim to elucidate the association between VDR and macrophages in HSC activation. The results contribute to our understanding of the pathogenesis mechanism of hepatic fibrosis, and provide potential therapeutic targets for its treatment.
概要
肝纤维化有效治疗方案的制定需要深入了解其发病机制。肝纤维化的特征是活化的肝星状细胞(aHSC)过度产生细胞外基质。尽管已证实M2巨噬细胞能促进HSC活化,但所涉及的分子机制仍不明确。在此,我们提出参与巨噬细胞极化的维生素D受体(VDR)可能通过改变巨噬细胞外泌体的功能来调节巨噬细胞和HSC之间的通信。本研究证实,激动VDR可以抑制M2巨噬细胞对HSC活化的促进作用。源自M2巨噬细胞的外泌体可以促进HSC活化,同时激动VDR改变了M2外泌体中的蛋白质组分并逆转其激活HSC的作用。平滑肌细胞相关蛋白5(SMAP-5)被发现是M2巨噬细胞外泌体促进HSC活化的关键效应蛋白,其作用机制是通过调节HSC的自噬通量。基于这些结果表明,VDR激动剂和巨噬细胞外泌体分泌抑制剂的联合治疗可获得更加优异的抗肝纤维化效果。本研究旨在阐明VDR和巨噬细胞在HSC激活中的关联,其结果有助于对肝纤维化的发病机制理解,并为肝纤维化的治疗提供潜在的靶点。
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bernsmeier C, van der Merwe S, Périanin A, 2020. Innate immune cells in cirrhosis. J Hepatol, 73(1):186–201. https://doi.org/10.1016/j.jhep.2020.03.027
Cai XP, Cai HQ, Wang J, et al., 2022. Molecular pathogenesis of acetaminophen-induced liver injury and its treatment options. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 23(4):265–285. https://doi.org/10.1631/jzus.B2100977
Chen LS, Yao XW, Yao HB, et al., 2020. Exosomal miR-103-3p from LPS-activated THP-1 macrophage contributes to the activation of hepatic stellate cells. FASEB J, 34(4):5178–5192. https://doi.org/10.1096/fj.201902307RRR
Ding N, Yu RT, Subramaniam N, et al., 2013. A vitamin D receptor/SMAD genomic circuit gates hepatic fibrotic response. Cell, 153(3):601–613. https://doi.org/10.1016/j.cell.2013.03.028
Dong BN, Zhou Y, Wang W, et al., 2020. Vitamin D receptor activation in liver macrophages ameliorates hepatic inflammation, steatosis, and insulin resistance in mice. Hepatology, 71(5):1559–1574. https://doi.org/10.1002/hep.30937
Duran A, Hernandez ED, Reina-Campos M, et al., 2016. P62/SQSTM1 by binding to vitamin D receptor inhibits hepatic stellate cell activity, fibrosis, and liver cancer. Cancer Cell, 30(4):595–609. https://doi.org/10.1016/j.ccell.2016.09.004
Hu MY, Wang Y, Liu ZS, et al., 2021. Hepatic macrophages act as a central hub for relaxin-mediated alleviation of liver fibrosis. Nat Nanotechnol, 16(4):466–477. https://doi.org/10.1038/s41565-020-00836-6
Kalluri R, LeBleu VS, 2020. The biology, function, and biomedical applications of exosomes. Science, 367(6478): eaau6977. https://doi.org/10.1126/science.aau6977
Kisseleva T, Brenner D, 2021. Molecular and cellular mechanisms of liver fibrosis and its regression. Nat Rev Gastroenterol Hepatol, 18(3):151–166. https://doi.org/10.1038/s41575-020-00372-7
Konstantakis C, Tselekouni P, Kalafateli M, et al., 2016. Vitamin D deficiency in patients with liver cirrhosis. Ann Gastroenterol, 29(3):297–306. https://doi.org/10.20524/aog.2016.0037
Li PF, Ma C, Li J, et al., 2022. Proteomic characterization of four subtypes of M2 macrophages derived from human THP-1 cells. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 23(5):407–422. https://doi.org/10.1631/jzus.B2100930
Liu PT, Stenger S, Li HY, et al., 2006. Toll-like receptor triggering of a vitamin D-mediated human antimicrobial response. Science, 311(5768): 1770–1773. https://doi.org/10.1126/science.1123933
Lu ZN, Niu WX, Zhang N, et al., 2021. Pantoprazole ameliorates liver fibrosis and suppresses hepatic stellate cell activation in bile duct ligation rats by promoting YAP degradation. Acta Pharmacol Sin, 42(11):1808–1820. https://doi.org/10.1038/s41401-021-00754-w
Mokdad AA, Lopez AD, Shahraz S, et al., 2014. Liver cirrhosis mortality in 187 countries between 1980 and 2010: a systematic analysis. BMC Med, 12:145. https://doi.org/10.1186/s12916-014-0145-y
Pellicoro A, Ramachandran P, Iredale JP, et al., 2014. Liver fibrosis and repair: immune regulation of wound healing in a solid organ. Nat Rev Immunol, 14(3):181–194. https://doi.org/10.1038/nri3623
Qu C, Zheng DD, Li S, et al., 2018. Tyrosine kinase SYK is a potential therapeutic target for liver fibrosis. Hepatology, 68(3):1125–1139. https://doi.org/10.1002/hep.29881
Ran Y, Xiong MG, Xu ZS, et al., 2019. YIPF5 is essential for innate immunity to DNA virus and facilitates COPII-dependent STING trafficking. J Immunol, 203(6):1560–1570. https://doi.org/10.4049/jimmunol.1900387
Sassi F, Tamone C, D’Amelio P, 2018. Vitamin D: nutrient, hormone, and immunomodulator. Nutrients, 10(11):1656. https://doi.org/10.3390/nu10111656
Seki E, Schwabe RF, 2015. Hepatic inflammation and fibrosis: functional links and key pathways. Hepatology, 61(3): 1066–1079. https://doi.org/10.1002/hep.27332
Shen DF, Cheng H, Cai BZ, et al., 2022. N-n-Butyl haloperidol iodide ameliorates liver fibrosis and hepatic stellate cell activation in mice. Acta Pharmacol Sin, 43(1):133–145. https://doi.org/10.1038/s41401-021-00630-7
Shen ZR, Shao JJ, Sun JQ, et al., 2022. Exosomes released by melanocytes modulate fibroblasts to promote keloid formation: a pilot study. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 23(8):699–704. https://doi.org/10.1631/jzus.B2200036
Sica A, Invernizzi P, Mantovani A, 2014. Macrophage plasticity and polarization in liver homeostasis and pathology. Hepatology, 59(5):2034–2042. https://doi.org/10.1002/hep.26754
Soonthornsit J, Sakai N, Sasaki Y, et al., 2017. YIPF1, YIPF2, and YIPF6 are medial-/trans-Golgi and trans-Golgi network-localized Yip domain family proteins, which play a role in the Golgi reassembly and glycan synthesis. Exp Cell Res, 353(2):100–108. https://doi.org/10.1016/j.yexcr.2017.03.011
Stolle K, Schnoor M, Fuellen G, et al., 2005. Cloning, cellular localization, genomic organization, and tissue-specific expression of the TGFβ1-inducible SMAP-5 gene. Gene, 351:119–130. https://doi.org/10.1016/j.gene.2005.03.012
Sun YY, Li XF, Meng XM, et al., 2017. Macrophage phenotype in liver injury and repair. Scand J Immunol, 85(3): 166–174. https://doi.org/10.1111/sji.12468
Taguchi Y, Horiuchi Y, Kano F, et al., 2017. Novel prosurvival function of Yip1A in human cervical cancer cells: constitutive activation of the IRE1 and PERK pathways of the unfolded protein response. Cell Death Dis, 8(3):e2718. https://doi.org/10.1038/cddis.2017.147
Thery C, Witwer KW, Aikawa E, et al., 2018. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J Extracell Vesicles, 7(1):1535750. https://doi.org/10.1080/20013078.2018.1535750
Tsuchida T, Friedman SL, 2017. Mechanisms of hepatic stellate cell activation. Nat Rev Gastroenterol Hepatol, 14(7): 397–411. https://doi.org/10.1038/nrgastro.2017.38
Wang KW, 2015. Autophagy and apoptosis in liver injury. Cell Cycle, 14(11):1631–1642. https://doi.org/10.1080/15384101.2015.1038685
Wang XP, Wang GY, Qu JW, et al., 2020. Calcipotriol inhibits NLRP3 signal through YAP1 activation to alleviate cholestatic liver injury and fibrosis. Front Pharmacol, 11:200. https://doi.org/10.3389/fphar.2020.00200
Wang ZL, Yang JJ, Sun XH, et al., 2023. Exosome-mediated regulatory mechanisms in skeletal muscle: a narrative review. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 24(1): 1–14. https://doi.org/10.1631/jzus.B2200243
Yin CY, Evason KJ, Asahina K, et al., 2013. Hepatic stellate cells in liver development, regeneration, and cancer. J Clin Invest, 123(5):1902–1910. https://doi.org/10.1172/JCI66369
Zhou Y, Dong BN, Kim KH, et al., 2020. Vitamin D receptor activation in liver macrophages protects against hepatic endoplasmic reticulum stress in mice. Hepatology, 71(4): 1453–1466. https://doi.org/10.1002/hep.30887
Acknowledgments
We thank the Public Platform of the State Key Laboratory of Natural Medicines for assistance with the pathological-section imaging. This work was supported by the National Natural Science Foundation of China (Nos. 81930099, 81773664, 82130102, 92159304, 81703585, and 81903651), the Natural Science Foundation of Jiangsu Province (Nos. BK20212011 and BK20180565), the Technology Innovation Project of Nucleic Acid Drug from National Center of Technology Innovation for Biopharmaceuticals (No. NCTIB2022HS01014), the “Double First-Class” University Project (No. CPU2022QZ05), the 111 Project from the Ministry of Education of China and the State Administration of Foreign Expert Affairs of China (Nos. 111-2-07 and B17047), the Fundamental Research Funds for the Central Universities of China (No. 2632022ZD11), and the Open Project of State Key Laboratory of Natural Medicines (No. SKLNMZZ202017), China.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Materials and methods
The detailed methods are provided in the electronic supplementary materials of this paper.
Author contributions
Xuwentai LIU (Conceptualization: equal; Data curation: lead; Formal analysis: equal; Investigation: lead; Methodology: equal; Validation: lead; Visualization: lead; Writing-original draft: lead; Writing-review & editing: equal); Yue WU (Validation: supporting); Yanyi LI (Writing-review & editing: supporting); Kaiming LI (Writing-review & editing: supporting); Siyuan HOU (Conceptualization: supporting; Funding acquisition: supporting; Project administration: supporting; Resources: supporting); Ming DING (Resources: supporting; Visualization: supporting); Jingmin TAN (Methodology: supporting; Visualization: supporting); Zijing ZHU (Methodology: supporting); Yingqi TANG (Methodology: supporting); Yuming LIU (Methodology: supporting); Qianhui SUN (Investigation: supporting); Cong WANG (Conceptualization: lead; Data curation: equal; Formal analysis: lead; Funding acquisition: equal; Methodology: lead; Project administration: lead; Resources: equal; Supervision: lead; Writing-review & editing: lead); Can ZHANG (Funding acquisition: lead; Project administration: equal; Resources: lead; Supervision: equal).
Compliance with ethics guidelines
Xuwentai LIU, Yue WU, Yanyi LI, Kaiming LI, Siyuan HOU, Ming DING, Jingmin TAN, Zijing ZHU, Yingqi TANG, Yuming LIU, Qianhui SUN, Cong WANG, and Can ZHANG declare that they have no conflict of interest.
Informed consent was obtained from all the human blood donors before the experiments were carried out. All experiments were performed in compliance with the Guide for Use of Human Blood and approved by the Ethics Committee of China Pharmaceutical University. All animal experiments were approved by the Animal Experimentation Ethics Committee of China Pharmaceutical University (Approval No. 2021-10-011).
Supplementary information
Materials and methods; Table S1; Figs. S1–S7
Supplementary information
Rights and permissions
About this article
Cite this article
Liu, X., Wu, Y., Li, Y. et al. Vitamin D receptor (VDR) mediates the quiescence of activated hepatic stellate cells (aHSCs) by regulating M2 macrophage exosomal smooth muscle cell-associated protein 5 (SMAP-5). J. Zhejiang Univ. Sci. B 24, 248–261 (2023). https://doi.org/10.1631/jzus.B2200383
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B2200383
Key words
- Hepatic fibrosis
- Hepatic stellate cell (HSC)
- Macrophage
- Exosome
- Vitamin D receptor (VDR)
- Smooth muscle cell-associated protein 5 (SMAP-5)