Abstract
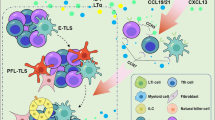
Tertiary lymphoid structures (TLSs) are formations at sites with persistent inflammatory stimulation, including tumors. These ectopic lymphoid organs mainly consist of chemo-attracting B cells, T cells, and supporting dendritic cells (DCs). Mature TLSs exhibit functional organization for the optimal development and collaboration of adaptive immune response, delivering an augmented effect on the tumor microenvironment (TME). The description of the positive correlation between TLSs and tumor prognosis is reliable only under a certain condition involving the localization and maturation of TLSs. Emerging evidence suggests that underlying mechanisms of the anti-tumor effect of TLSs pave the way for novel immunotherapies. Several approaches have been developed to take advantage of intratumoral TLSs, either by combining it with therapeutic agents or by inducing the neogenesis of TLSs.
概要
三级淋巴结构主要在包括肿瘤在内的慢性炎症部位的非淋巴组织中发展形成。这一异位形成的淋巴结构主要包含趋化因子招募而来的B细胞、T细胞以及起支持作用的树突状细胞(DC)。成熟的三级淋巴结构有利于适应性免疫细胞的发育与互作,进而增强其抗肿瘤免疫应答。但该抗肿瘤效应取决于三级淋巴结构的具体定位以及成熟阶段。目前有越来越多的研究表明三级淋巴结构的抗肿瘤机制将有助于新型免疫疗法的探索。现有的治疗策略研究方案包括联合瘤内三级淋巴结构与药物治疗或诱导瘤内三级淋巴结构的形成,从而实现提升肿瘤治疗疗效和改善肿瘤患者预后的目的。
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ahrends T, Spanjaard A, Pilzecker B, et al., 2017. CD4+ T cell help confers a cytotoxic T cell effector program including coinhibitory receptor downregulation and increased tissue invasiveness. Immunity, 47(5):848–861.e5. https://doi.org/10.1016/j.immuni.2017.10.009
Allen CDC, Okada T, Tang HL, et al., 2007. Imaging of germinal center selection events during affinity maturation. Science, 315(5811):528–531. https://doi.org/10.1126/science.1136736
Allen E, Jabouille A, Rivera LB, et al., 2017. Combined anti-angiogenic and anti-PD-L1 therapy stimulates tumor immunity through HEV formation. Sci Transl Med, 9(385): eaak9679. https://doi.org/10.1126/scitranslmed.aak9679
Aloisi F, Pujol-Borrell R, 2006. Lymphoid neogenesis in chronic inflammatory diseases. Nat Rev Immunol, 6(3): 205–217. https://doi.org/10.1038/nri1786
Amaria RN, Reddy SM, Tawbi HA, et al., 2018. Neoadjuvant immune checkpoint blockade in high-risk resectable melanoma. Nat Med, 24(11):1649–1654. https://doi.org/10.1038/s41591-018-0197-1
Ansel KM, Ngo VN, Hyman PL, et al., 2000. A chemokine-driven positive feedback loop organizes lymphoid follicles. Nature, 406(6793):309–314. https://doi.org/10.1038/35018581
Asrir A, Tardiveau C, Coudert J, et al., 2022. Tumor-associated high endothelial venules mediate lymphocyte entry into tumors and predict response to PD-1 plus CTLA-4 combination immunotherapy. Cancer Cell, 40(3):318–334.e9. https://doi.org/10.1016/j.ccell.2022.01.002
Barone F, Gardner DH, Nayar S, et al., 2016. Stromal fibroblasts in tertiary lymphoid structures: a novel target in chronic inflammation. Front Immunol, 7:477. https://doi.org/10.3389/fimmu.2016.00477
Bénézech C, White A, Mader E, et al., 2010. Ontogeny of stromal organizer cells during lymph node development. J Immunol, 184(8):4521–4530. https://doi.org/10.4049/jimmunol.0903113
Bénézech C, Luu NT, Walker JA, et al., 2015. Inflammation-induced formation of fat-associated lymphoid clusters. Nat Immunol, 16(8):819–828. https://doi.org/10.1038/ni.3215
Bennett SRM, Carbone FR, Karamalis F, et al., 1997. Induction of a CD8+ cytotoxic T lymphocyte response by cross-priming requires cognate CD4+ T cell help. J Exp Med, 186(1):65–70. https://doi.org/10.1084/jem.186.1.65
Berek C, Berger A, Apel M, 1991. Maturation of the immune response in germinal centers. Cell, 67(6):1121–1129. https://doi.org/10.1016/0092-8674(91)90289-b
Bindea G, Mlecnik B, Tosolini M, et al., 2013. Spatiotemporal dynamics of intratumoral immune cells reveal the immune landscape in human cancer. Immunity, 39(4): 782–795. https://doi.org/10.1016/j.immuni.2013.10.003
Braun A, Worbs T, Moschovakis GL, et al., 2011. Afferent lymph-derived T cells and DCs use different chemokine receptor CCR7-dependent routes for entry into the lymph node and intranodal migration. Nat Immunol, 12(9):879–887. https://doi.org/10.1038/ni.2085
Buisseret L, Garaud S, de Wind A, et al., 2017. Tumor-infiltrating lymphocyte composition, organization and PD-1/PD-L1 expression are linked in breast cancer. OncoImmunology, 6(1):e1257452. https://doi.org/10.1080/2162402X.2016.1257452
Cabrita R, Lauss M, Sanna A, et al., 2020. Tertiary lymphoid structures improve immunotherapy and survival in melanoma. Nature, 577(7791):561–565. https://doi.org/10.1038/s41586-019-1914-8
Calderaro J, Petitprez F, Becht E, et al., 2019. Intra-tumoral tertiary lymphoid structures are associated with a low risk of early recurrence of hepatocellular carcinoma. J Hepatol, 70(1):58–65. https://doi.org/10.1016/j.jhep.2018.09.003
Carmi Y, Spitzer MH, Linde IL, et al., 2015. Allogeneic IgG combined with dendritic cell stimuli induce antitumour T-cell immunity. Nature, 521(7550):99–104. https://doi.org/10.1038/nature14424
Carrega P, Loiacono F, di Carlo E, et al., 2015. NCR+ILC3 concentrate in human lung cancer and associate with intratumoral lymphoid structures. Nat Commun, 6:8280. https://doi.org/10.1038/ncomms9280
Castellino F, Huang AY, Altan-Bonnet G, et al., 2006. Chemokines enhance immunity by guiding naive CD8+ T cells to sites of CD4+ T cell-dendritic cell interaction. Nature, 440(7086):890–895. https://doi.org/10.1038/nature04651
Castino GF, Cortese N, Capretti G, et al., 2016. Spatial distribution of B cells predicts prognosis in human pancreatic adenocarcinoma. OncoImmunology, 5(4):e1085147. https://doi.org/10.1080/2162402X.2015.1085147
Choi YS, Kageyama R, Eto D, et al., 2011. ICOS receptor instructs T follicular helper cell versus effector cell differentiation via induction of the transcriptional repressor Bcl6. Immunity, 34(6):932–946. https://doi.org/10.1016/j.immuni.2011.03.023
Cillo AR, Kurten CHL, Tabib T, et al., 2020. Immune landscape of viral- and carcinogen-driven head and neck cancer. Immunity, 52(1):183–199.e9. https://doi.org/10.1016/j.immuni.2019.11.014
Colbeck EJ, Jones E, Hindley JP, et al., 2017. Treg depletion licenses T cell-driven HEV neogenesis and promotes tumor destruction. Cancer Immunol Res, 5(11):1005–1015. https://doi.org/10.1158/2326-6066.CIR-17-0131
Cui C, Wang JW, Fagerberg E, et al., 2021. Neoantigendriven B cell and CD4 T follicular helper cell collaboration promotes anti-tumor CD8 T cell responses. Cell, 184(25):6101–6118.e13. https://doi.org/10.1016/j.cell.2021.11.007
Dai SY, Zeng H, Liu ZP, et al., 2021. Intratumoral CXCL13+ CD8+T cell infiltration determines poor clinical outcomes and immunoevasive contexture in patients with clear cell renal cell carcinoma. J Immunother Cancer, 9(2): e001823. https://doi.org/10.1136/jitc-2020-001823
Dejardin E, Droin NM, Delhase M, et al., 2002. The lymphotoxin-β receptor induces different patterns of gene expression via two NF-κB pathways. Immunity, 17(4): 525–535. https://doi.org/10.1016/s1074-7613(02)00423-5
Denkert C, von Minckwitz G, Darb-Esfahani S, et al., 2018. Tumour-infiltrating lymphocytes and prognosis in different subtypes of breast cancer: a pooled analysis of 3771 patients treated with neoadjuvant therapy. Lancet Oncol, 19(1):40–50. https://doi.org/10.1016/S1470-2045(17)30904-X
Depil S, Duchateau P, Grupp SA, et al., 2020. ‘Off-the-shelf’ allogeneic CAR T cells: development and challenges. Nat Rev Drug Discov, 19(3):185–199. https://doi.org/10.1038/s41573-019-0051-2
de Togni P, Goellner J, Ruddle NH, et al., 1994. Abnormal development of peripheral lymphoid organs in mice deficient in lymphotoxin. Science, 264(5159):703–707. https://doi.org/10.1126/science.8171322
di Pucchio T, Chatterjee B, Smed-Sörensen A, et al., 2008. Direct proteasome-independent cross-presentation of viral antigen by plasmacytoid dendritic cells on major histocompatibility complex class I. Nat Immunol, 9(5): 551–557. https://doi.org/10.1038/ni.1602
Drayton DL, Liao S, Mounzer RH, et al., 2006. Lymphoid organ development: from ontogeny to neogenesis. Nat Immunol, 7(4):344–353. https://doi.org/10.1038/ni1330
Eickhoff S, Brewitz A, Gerner MY, et al., 2015. Robust anti-viral immunity requires multiple distinct T cell-dendritic cell interactions. Cell, 162(6):1322–1337. https://doi.org/10.1016/j.cell.2015.08.004
Furtado GC, Pacer ME, Bongers G, et al., 2014. TNFα-dependent development of lymphoid tissue in the absence of RORγt+ lymphoid tissue inducer cells. Mucosal Immunol, 7(3):602–614. https://doi.org/10.1038/mi.2013.79
Garner H, de Visser KE, 2020. Immune crosstalk in cancer progression and metastatic spread: a complex conversation. Nat Rev Immunol, 20(8):483–497. https://doi.org/10.1038/s41577-019-0271-z
Gee MH, Han A, Lofgren SM, et al., 2018. Antigen identification for orphan T cell receptors expressed on tumor-infiltrating lymphocytes. Cell, 172(3):549–563.e16. https://doi.org/10.1016/j.cell.2017.11.043
Germain C, Gnjatic S, Tamzalit F, et al., 2014. Presence of B cells in tertiary lymphoid structures is associated with a protective immunity in patients with lung cancer. Am J Respir Crit Care Med, 189(7):832–844. https://doi.org/10.1164/rccm.201309-1611OC
Geurtsvankessel CH, Willart MAM, Bergen IM, et al., 2009. Dendritic cells are crucial for maintenance of tertiary lymphoid structures in the lung of influenza virus-infected mice. J Exp Med, 206(11):2339–2349. https://doi.org/10.1084/jem.20090410
Giraldo NA, Sanchez-Salas R, Peske JD, et al., 2019. The clinical role of the TME in solid cancer. Br J Cancer, 120(1): 45–53. https://doi.org/10.1038/s41416-018-0327-z
Goc J, Germain C, Vo-Bourgais TKD, et al., 2014. Dendritic cells in tumor-associated tertiary lymphoid structures signal a Th1 cytotoxic immune contexture and license the positive prognostic value of infiltrating CD8+ T cells. Cancer Res, 74(3):705–715. https://doi.org/10.1158/0008-5472.CAN-13-1342
Goenka R, Barnett LG, Silver JS, et al., 2011. Cutting edge: dendritic cell-restricted antigen presentation initiates the follicular helper T cell program but cannot complete ultimate effector differentiation. J Immunol, 187(3):1091–1095. https://doi.org/10.4049/jimmunol.1100853
Groeneveld CS, Fontugne J, Cabel L, et al., 2021. Tertiary lymphoid structures marker CXCL13 is associated with better survival for patients with advanced-stage bladder cancer treated with immunotherapy. Eur J Cancer, 148: 181–189. https://doi.org/10.1016/j.ejca.2021.01.036
Gu-Trantien C, Loi S, Garaud S, et al., 2013. CD4+ follicular helper T cell infiltration predicts breast cancer survival. J Clin Invest, 123(7):2873–2892. https://doi.org/10.1172/JCI67428
Guedj K, Khallou-Laschet J, Clement M, et al., 2014. M1 macrophages act as LTβR-independent lymphoid tissue inducer cells during atherosclerosis-related lymphoid neogenesis. Cardiovasc Res, 101(3):434–443. https://doi.org/10.1093/cvr/cvt263
Gunn MD, Ngo VN, Ansel KM, et al., 1998. A B-cell-homing chemokine made in lymphoid follicles activates Burkitt’s lymphoma receptor-1. Nature, 391(6669):799–803. https://doi.org/10.1038/35876
Halle S, Dujardin HC, Bakocevic N, et al., 2009. Induced bronchus-associated lymphoid tissue serves as a general priming site for T cells and is maintained by dendritic cells. J Exp Med, 206(12):2593–2601. https://doi.org/10.1084/jem.20091472
He JJ, Xiong XX, Yang H, et al., 2022. Defined tumor antigen-specific T cells potentiate personalized TCR-T cell therapy and prediction of immunotherapy response. Cell Res, 32(6): 530–542. https://doi.org/10.1038/s41422-022-00627-9
Helmink BA, Reddy SM, Gao JJ, et al., 2020. B cells and tertiary lymphoid structures promote immunotherapy response. Nature, 577(7791):549–555. https://doi.org/10.1038/s41586-019-1922-8
Hinshaw DC, Shevde LA, 2019. The tumor microenvironment innately modulates cancer progression. Cancer Res, 79(18):4557–4566. https://doi.org/10.1158/0008-5472.CAN-18-3962
Hiraoka N, Ino Y, Yamazaki-Itoh R, et al., 2015. Intratumoral tertiary lymphoid organ is a favourable prognosticator in patients with pancreatic cancer. Br J Cancer, 112(11): 1782–1790. https://doi.org/10.1038/bjc.2015.145
Hodi FS, O’Day SJ, McDermott DF, et al., 2010. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med, 363(8):711–723. https://doi.org/10.1056/NEJMoa1003466
Hor JL, Whitney PG, Zaid A, et al., 2015. Spatiotemporally distinct interactions with dendritic cell subsets facilitates CD4+ and CD8+ T cell activation to localized viral infection. Immunity, 43(3):554–565. https://doi.org/10.1016/j.immuni.2015.07.020
Jansen CS, Prokhnevska N, Master VA, et al., 2019. An intra-tumoral niche maintains and differentiates stem-like CD8 T cells. Nature, 576(7787):465–470. https://doi.org/10.1038/s41586-019-1836-5
Janssen EM, Lemmens EE, Wolfe T, et al., 2003. CD4+ T cells are required for secondary expansion and memory in CD8+ T lymphocytes. Nature, 421(6925):852–856. https://doi.org/10.1038/nature01441
Jiang XG, 2020. Lymphatic vasculature in tumor metastasis and immunobiology. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 21(1):3–11. https://doi.org/10.1631/jzus.B1800633
Jin KF, Cao YF, Gu Y, et al., 2021. Poor clinical outcomes and immunoevasive contexture in CXCL13+CD8+ T cells enriched gastric cancer patients. OncoImmunology, 10(1): 1915560. https://doi.org/10.1080/2162402X.2021.1915560
Johansson-Percival A, He B, Li ZJ, et al., 2017. De novo induction of intratumoral lymphoid structures and vessel normalization enhances immunotherapy in resistant tumors. Nat Immunol, 18(11):1207–1217. https://doi.org/10.1038/ni.3836
Joshi NS, Akama-Garren EH, Lu YS, et al., 2015. Regulatory T cells in tumor-associated tertiary lymphoid structures suppress anti-tumor T cell responses. Immunity, 43(3): 579–590. https://doi.org/10.1016/j.immuni.2015.08.006
Kato Y, Zaid A, Davey GM, et al., 2015. Targeting antigen to Clec9A primes follicular Th cell memory responses capable of robust recall. J Immunol, 195(3):1006–1014. https://doi.org/10.4049/jimmunol.1500767
Kerfoot SM, Yaari G, Patel JR, et al., 2011. Germinal center B cell and T follicular helper cell development initiates in the interfollicular zone. Immunity, 34(6):947–960. https://doi.org/10.1016/j.immuni.2011.03.024
Kinker GS, Vitiello GAF, Ferreira WAS, et al., 2021. B cell orchestration of anti-tumor immune responses: a matter of cell localization and communication. Front Cell Dev Biol, 9:678127. https://doi.org/10.3389/fcell.2021.678127
Krishnaswamy JK, Gowthaman U, Zhang BY, et al., 2017. Migratory CD11b+ conventional dendritic cells induce T follicular helper cell-dependent antibody responses. Sci Immunol, 2(18):eaam9169. https://doi.org/10.1126/sciimmunol.aam9169
Kroeger DR, Milne K, Nelson BH, 2016. Tumor-infiltrating plasma cells are associated with tertiary lymphoid structures, cytolytic T-cell responses, and superior prognosis in ovarian cancer. Clin Cancer Res, 22(12):3005–3015. https://doi.org/10.1158/1078-0432.CCR-15-2762
Lee JM, Lee MH, Garon E, et al., 2017. Phase I trial of intra-tumoral injection of CCL21 gene-modified dendritic cells in lung cancer elicits tumor-specific immune responses and CD8+ T-cell infiltration. Clin Cancer Res, 23(16): 4556–4568. https://doi.org/10.1158/1078-0432.CCR-16-2821
Lee Y, Chin RK, Christiansen P, et al., 2006. Recruitment and activation of naive T cells in the islets by lymphotoxin β receptor-dependent tertiary lymphoid structure. Immunity, 25(3):499–509. https://doi.org/10.1016/j.immuni.2006.06.016
Lei X, Lei Y, Li JK, et al., 2020. Immune cells within the tumor microenvironment: biological functions and roles in cancer immunotherapy. Cancer Lett, 470:126–133. https://doi.org/10.1016/j.canlet.2019.11.009
Li HJ, van der Leun AM, Yofe I, et al., 2019. Dysfunctional CD8 T cells form a proliferative, dynamically regulated compartment within human melanoma. Cell, 176(4):775–789.e18. https://doi.org/10.1016/j.cell.2018.11.043
Li JP, Wu CY, Chen MY, et al., 2021. PD-1+CXCR5+CD4+ Th-CXCL13 cell subset drives B cells into tertiary lymphoid structures of nasopharyngeal carcinoma. J Immunother Cancer, 9(7):e002101. https://doi.org/10.1136/jitc-2020-002101
Lochner M, Ohnmacht C, Presley L, et al., 2011. Microbiota-induced tertiary lymphoid tissues aggravate inflammatory disease in the absence of RORγ T and LTi cells. J Exp Med, 208(1):125–134. https://doi.org/10.1084/jem.20100052
Luther SA, Lopez T, Bai W, et al., 2000. BLC expression in pancreatic islets causes B cell recruitment and lymphotoxin-dependent lymphoid neogenesis. Immunity, 12(5):471–481. https://doi.org/10.1016/s1074-7613(00)80199-5
Luther SA, Bidgol A, Hargreaves DC, et al., 2002. Differing activities of homeostatic chemokines CCL19, CCL21, and CXCL12 in lymphocyte and dendritic cell recruitment and lymphoid neogenesis. J Immunol, 169(1):424–433. https://doi.org/10.4049/jimmunol.169.1.424
Luther SA, Ansel KM, Cyster JG, 2003. Overlapping roles of CXCL13, interleukin 7 receptor α, and CCR7 ligands in lymph node development. J Exp Med, 197(9):1191–1198. https://doi.org/10.1084/jem.20021294
Lutz ER, Wu AA, Bigelow E, et al., 2014. Immunotherapy converts nonimmunogenic pancreatic tumors into immunogenic foci of immune regulation. Cancer Immunol Res, 2(7):616–631. https://doi.org/10.1158/2326-6066.CIR-14-0027
Maldonado L, Teague JE, Morrow MP, et al., 2014. Intramuscular therapeutic vaccination targeting HPV16 induces T cell responses that localize in mucosal lesions. Sci Transl Med, 6(221):221ra213. https://doi.org/10.1126/scitranslmed.3007323
Martinet L, Garrido I, Filleron T, et al., 2011. Human solid tumors contain high endothelial venules: association with T- and B-lymphocyte infiltration and favorable prognosis in breast cancer. Cancer Res, 71(17):5678–5687. https://doi.org/10.1158/0008-5472.CAN-11-0431
Matsubara S, Seki M, Suzuki S, et al., 2019. Tertiary lymphoid organs in the inflammatory myopathy associated with PD-1 inhibitors. J Immunother Cancer, 7(1):256. https://doi.org/10.1186/s40425-019-0736-4
Mebius RE, Rennert P, Weissman IL, 1997. Developing lymph nodes collect CD4+CD3−LTβ+ cells that can differentiate to APC, NK cells, and follicular cells but not T or B cells. Immunity, 7(4):493–504. https://doi.org/10.1016/s1074-7613(00)80371-4
Meylan M, Petitprez F, Becht E, et al., 2022. Tertiary lymphoid structures generate and propagate anti-tumor antibody-producing plasma cells in renal cell cancer. Immunity, 55(3):527–541.e5. https://doi.org/10.1016/j.immuni.2022.02.001
Morcrette G, Hirsch TZ, Badour E, et al., 2019. APC germline hepatoblastomas demonstrate cisplatin-induced intratumor tertiary lymphoid structures. OncoImmunology, 8(6):e1583547. https://doi.org/10.1080/2162402X.2019.1583547
Mounzer RH, Svendsen OS, Baluk P, et al., 2010. Lymphotoxin-alpha contributes to lymphangiogenesis. Blood, 116(12): 2173–2182. https://doi.org/10.1182/blood-2009-12-256065
Munoz-Erazo L, Rhodes JL, Marion VC, et al., 2020. Tertiary lymphoid structures in cancer—considerations for patient prognosis. Cell Mol Immunol, 17(6):570–575. https://doi.org/10.1038/s41423-020-0457-0
Ngo VN, Korner H, Gunn MD, et al., 1999. Lymphotoxin α/β and tumor necrosis factor are required for stromal cell expression of homing chemokines in B and T cell areas of the spleen. J Exp Med, 189(2):403–412. https://doi.org/10.1084/jem.189.2.403
Niogret J, Berger H, Rebe C, et al., 2021. Follicular helper-T cells restore CD8+-dependent antitumor immunity and anti-PD-L1/PD-1 efficacy. J Immunother Cancer, 9(6): e002157. https://doi.org/10.1136/jitc-2020-002157
Noël G, Fontsa ML, Garaud S, et al., 2021. Functional Th1-oriented T follicular helper cells that infiltrate human breast cancer promote effective adaptive immunity. J Clin Invest, 131(19): e139905. https://doi.org/10.1172/JCI139905
Overacre-Delgoffe AE, Bumgarner HJ, Cillo AR, et al., 2021. Microbiota-specific T follicular helper cells drive tertiary lymphoid structures and anti-tumor immunity against colorectal cancer. Immunity, 54(12):2812–2824.e4. https://doi.org/10.1016/j.immuni.2021.11.003
Pardoll DM, 2012. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer, 12(4):252–264. https://doi.org/10.1038/nrc3239
Peske JD, Thompson ED, Gemta L, et al., 2015. Effector lymphocyte-induced lymph node-like vasculature enables naive T-cell entry into tumours and enhanced anti-tumour immunity. Nat Commun, 6:7114. https://doi.org/10.1038/ncomms8114
Peters A, Pitcher LA, Sullivan JM, et al., 2011. Th17 cells induce ectopic lymphoid follicles in central nervous system tissue inflammation. Immunity, 35(6):986–996. https://doi.org/10.1016/j.immuni.2011.10.015
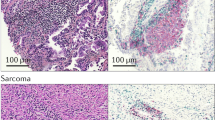
Petitprez F, de Reyniès A, Keung EZ, et al., 2020. B cells are associated with survival and immunotherapy response in sarcoma. Nature, 577(7791):556–560. https://doi.org/10.1038/s41586-019-1906-8
Pitzalis C, Jones GW, Bombardieri M, et al., 2014. Ectopic lymphoid-like structures in infection, cancer and autoimmunity. Nat Rev Immunol, 14(7):447–462. https://doi.org/10.1038/nri3700
Posch F, Silina K, Leibl S, et al., 2018. Maturation of tertiary lymphoid structures and recurrence of stage II and III colorectal cancer. OncoImmunology, 7(2):e1378844. https://doi.org/10.1080/2162402X.2017.1378844
Ridge JP, di Rosa F, Matzinger P, 1998. A conditioned dendritic cell can be a temporal bridge between a CD4+ T-helper and a T-killer cell. Nature, 393(6684):474–478. https://doi.org/10.1038/30989
Rodriguez AB, Peske JD, Woods AN, et al., 2021. Immune mechanisms orchestrate tertiary lymphoid structures in tumors via cancer-associated fibroblasts. Cell Rep, 36(3): 109422. https://doi.org/10.1016/j.celrep.2021.109422
Ruffin AT, Cillo AR, Tabib T, et al., 2021. B cell signatures and tertiary lymphoid structures contribute to outcome in head and neck squamous cell carcinoma. Nat Commun, 12:3349. https://doi.org/10.1038/s41467-021-23355-x
Sautès-Fridman C, Petitprez F, Calderaro J, et al., 2019. Tertiary lymphoid structures in the era of cancer immunotherapy. Nat Rev Cancer, 19(6):307–325. https://doi.org/10.1038/s41568-019-0144-6
Schmitt N, Morita R, Bourdery L, et al., 2009. Human dendritic cells induce the differentiation of interleukin-21-producing T follicular helper-like cells through interleukin-12. Immunity, 31(1):158–169. https://doi.org/10.1016/j.immuni.2009.04.016
Schoenberger SP, Toes REM, van der Voort EIH, et al., 1998. T-cell help for cytotoxic T lymphocytes is mediated by CD40–CD40L interactions. Nature, 393(6684):480–483. https://doi.org/10.1038/31002
Shalapour S, Lin XJ, Bastian IN, et al., 2017. Inflammation-induced IgA+ cells dismantle anti-liver cancer immunity. Nature, 551(7680):340–345. https://doi.org/10.1038/nature24302
Shedlock DJ, Shen H, 2003. Requirement for CD4 T cell help in generating functional CD8 T cell memory. Science, 300(5617):337–339. https://doi.org/10.1126/science.1082305
Siliņa K, Soltermann A, Attar FM, et al., 2018. Germinal centers determine the prognostic relevance of tertiary lymphoid structures and are impaired by corticosteroids in lung squamous cell carcinoma. Cancer Res, 78(5):1308–1320. https://doi.org/10.1158/0008-5472.CAN-17-1987
Song IH, Heo SH, Bang WS, et al., 2017. Predictive value of tertiary lymphoid structures assessed by high endothelial venule counts in the neoadjuvant setting of triple-negative breast cancer. Cancer Res Treat, 49(2):399–407. https://doi.org/10.4143/crt.2016.215
Suzuki G, Sawa H, Kobayashi Y, et al., 1999. Pertussis toxin-sensitive signal controls the trafficking of thymocytes across the corticomedullary junction in the thymus. J Immunol, 162(10):5981–5985.
Thommen DS, Koelzer VH, Herzig P, et al., 2018. A transcriptionally and functionally distinct PD-1+ CD8+ T cell pool with predictive potential in non-small-cell lung cancer treated with PD-1 blockade. Nat Med, 24(7):994–1004. https://doi.org/10.1038/s41591-018-0057-z
Tran E, Robbins PF, Lu YC, et al., 2016. T-cell transfer therapy targeting mutant KRAS in cancer. N Engl J Med, 375(23): 2255–2262. https://doi.org/10.1056/NEJMoa1609279
van de Pavert SA, Olivier BJ, Goverse G, et al., 2009. Chemokine CXCL13 is essential for lymph node initiation and is induced by retinoic acid and neuronal stimulation. Nat Immunol, 10(11): 1193–1199. https://doi.org/10.1038/ni.1789
van Dijk N, Gil-Jimenez A, Silina K, et al., 2020. Preoperative ipilimumab plus nivolumab in locoregionally advanced urothelial cancer: the NABUCCO trial. Nat Med, 26(12): 1839–1844. https://doi.org/10.1038/s41591-020-1085-z
Veiga-Fernandes H, Coles MC, Foster KE, et al., 2007. Tyrosine kinase receptor RET is a key regulator of Peyer’s Patch organogenesis. Nature, 446(7135):547–551. https://doi.org/10.1038/nature05597
Weinstein AM, Chen L, Brzana EA, et al., 2017. Tbet and IL-36γ cooperate in therapeutic DC-mediated promotion of ectopic lymphoid organogenesis in the tumor microenvironment. OncoImmunology, 6(6):e1322238. https://doi.org/10.1080/2162402X.2017.1322238
Workel HH, Lubbers JM, Arnold R, et al., 2019. A transcriptionally distinct CXCL13+ CD103+ CD8+ T-cell population is associated with B-cell recruitment and neoantigen load in human cancer. Cancer Immunol Res, 7(5):784–796. https://doi.org/10.1158/2326-6066.CIR-18-0517
Wouters MCA, Nelson BH, 2018. Prognostic significance of tumor-infiltrating B cells and plasma cells in human cancer. Clin Cancer Res, 24(24):6125–6135. https://doi.org/10.1158/1078-0432.CCR-18-1481
Wu SZ, Roden DL, Wang CF, et al., 2020. Stromal cell diversity associated with immune evasion in human triple-negative breast cancer. EMBO J, 39(19):e104063. https://doi.org/10.15252/embj.2019104063
Wykes M, Pombo A, Jenkins C, et al., 1998. Dendritic cells interact directly with naive B lymphocytes to transfer antigen and initiate class switching in a primary T-dependent response. J Immunol, 161(3):1313–1319.
Yang SC, Batra RK, Hillinger S, et al., 2006. Intrapulmonary administration of CCL21 gene-modified dendritic cells reduces tumor burden in spontaneous murine bronchoalveolar cell carcinoma. Cancer Res, 66(6):3205–3213. https://doi.org/10.1158/0008-5472.CAN-05-3619
Yeh CH, Finney J, Okada T, et al., 2022. Primary germinal center-resident T follicular helper cells are a physiologically distinct subset of CXCR5hiPD-1hi T follicular helper cells. Immunity, 55(2):272–289.e7. https://doi.org/10.1016/j.immuni.2021.12.015
Yoshida H, Naito A, Inoue J, et al., 2002. Different cytokines induce surface lymphotoxin-αβ on IL-7 receptor-α cells that differentially engender lymph nodes and Peyer’s patches. Immunity, 17(6):823–833. https://doi.org/10.1016/s1074-7613(02)00479-x
Acknowledgments
This work was supported by the Zhejiang Provincial Key Project of Research and Development (No. 2019C03043), the National Natural Science Foundation of China (Nos. 32030035, 31870874, 32000623, and 32100693), and the Zhejiang Provincial Natural Science Foundation (No. LZ21C080001) of China.
Author information
Authors and Affiliations
Contributions
Jing CHEN was responsible for reference searching and manuscript writing, and Lie WANG and Jian CHEN for
Corresponding authors
Additional information
Compliance with ethics guidelines
Jing CHEN, Jian CHEN, and Lie WANG declare that they have no conflict of interest.
This review does not contain any studies with human or animal subjects performed by any of the authors.
Rights and permissions
About this article
Cite this article
Chen, J., Chen, J. & Wang, L. Tertiary lymphoid structures as unique constructions associated with the organization, education, and function of tumor-infiltrating immunocytes. J. Zhejiang Univ. Sci. B 23, 812–822 (2022). https://doi.org/10.1631/jzus.B2200174
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B2200174