Abstract
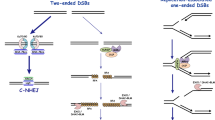
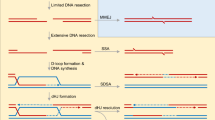
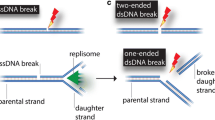
Genome stability can be threatened by both endogenous and exogenous agents. Organisms have evolved numerous mechanisms to repair DNA damage, including homologous recombination (HR) and non-homologous end joining (NHEJ). Among the factors associated with DNA repair, the MRE11-RAD50-NBS1 (MRN) complex (MRE11-RAD50-XRS2 in Saccharomyces cerevisiae) plays important roles not only in DNA damage recognition and signaling but also in subsequent HR or NHEJ repair. Upon detecting DNA damage, the MRN complex activates signaling molecules, such as the protein kinase ataxia-telangiectasia mutated (ATM), to trigger a broad DNA damage response, including cell cycle arrest. The nuclease activity of the MRN complex is responsible for DNA end resection, which guides DNA repair to HR in the presence of sister chromatids. The MRN complex is also involved in NHEJ, and has a species-specific role in hairpin repair. This review focuses on the structure of the MRN complex and its function in DNA damage repair.
概要
基因组的稳定性时刻都遭受着来自细胞内源和外源损伤因素的威胁,如果这些损伤不能够被及时、准确地修复,将会导致细胞衰老、死亡,甚至癌变的发生。因此,为了维持基因组稳定性,生物体进化出了一系列复杂而精确的修复方式以应对DNA损伤,包括同源重组修复和非同源末端连接修复。其中,MRE11-RAD50-NBS1(MRN)复合物(酿酒酵母同源物为MRE11-RAD50-XRS2)不仅参与DNA损伤识别和信号传递,还在同源重组修复和非同源末端连接修复过程中发挥着极其重要的作用。当DNA损伤被识别后,MRN复合物首先会激活信号分子(如ATM激酶等),从而引发信号级联反应放大DNA损伤应答效应,包括细胞周期阻滞。作为一个核酸酶,MRN复合物利用其核酸酶活性进行DNA末端切割,在有姐妹染色单体存在时,促使同源重组修复。此外,MRN复合物也参与了非同源末端连接修复,并在修复发卡结构方面具有物种特异性。该篇综述将主要讨论MRN复合物的结构及其在DNA损伤修复中的功能。
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Change history
08 April 2021
The typesetting format of the online version of the first issue (2021 22(01)) of Journal of Zhejiang University-SCIENCE B is different from that of the printed version (but all the text, figure and table contents in the article are correct). This is due to the new typesetting company adopted this year.
References
Bian L, Meng YL, Zhang MC, et al., 2019. MRE11-RAD50-NBS1 complex alterations and DNA damage response: implications for cancer treatment. Mol Cancer, 18:169. https://doi.org/10.1186/s12943-019-1100-5
Bugreev DV, Yu X, Egelman EH, et al., 2007. Novel pro- and anti-recombination activities of the Bloom’s syndrome helicase. Genes Dev, 21(23):3085–3094. https://doi.org/10.1101/gad.1609007
Buis J, Wu Y, Deng YB, et al., 2008. Mre11 nuclease activity has essential roles in DNA repair and genomic stability distinct from ATM activation. Cell, 135(1):85–96. https://doi.org/10.1016/j.cell.2008.08.015
Cannavo E, Reginato G, Cejka P, 2019. Stepwise 5′ DNA end-specific resection of DNA breaks by the Mre11-Rad50-Xrs2 and Sae2 nuclease ensemble. Proc Natl Acad Sci USA, 116(12):5505–5513. https://doi.org/10.1073/pnas.1820157116
Chen C, Zhang GL, Huang NJ, et al., 2013. Suppression of DNA-damage checkpoint signaling by Rsk-mediated phosphorylation of Mre11. Proc Natl Acad Sci USA, 110(51):20605–20610. https://doi.org/10.1073/pnas.1306328110
Davis AJ, Chen DJ, 2013. DNA double strand break repair via non-homologous end-joining. Transl Cancer Res, 2(3): 130–143. https://doi.org/10.3978/j.issn.2218-676X.2013.04.02
Deng SK, Yin Y, Petes TD, et al., 2015. Mre11-Sae2 and RPA collaborate to prevent palindromic gene amplification. Mol Cell, 60(3):500–508. https://doi.org/10.1016/j.molcel.2015.09.027
Deshpande RA, Myler LR, Soniat MM, et al., 2020. DNA-dependent protein kinase promotes DNA end processing by MRN and CtIP. Sci Adv, 6(2):eaay0922. https://doi.org/10.1126/sciadv.aay0922
Digweed M, Sperling K, 2004. Nijmegen breakage syndrome: clinical manifestation of defective response to DNA double-strand breaks. DNA Repair, 3(8–9): 1207–1217. https://doi.org/10.1016/j.dnarep.2004.03.004
Gao R, Singh R, Kaul Z, et al., 2015. Targeting of DNA damage signaling pathway induced senescence and reduced migration of cancer cells. J Gerontol: Ser A, 70(6): 701–713. https://doi.org/10.1093/gerona/glu019
Gobbini E, Cassani C, Vertemara J, et al., 2018. The MRX complex regulates Exo1 resection activity by altering DNA end structure. EMBO J, 37(16):e98588. https://doi.org/10.15252/embj.201798588
Ha GH, Ji JH, Chae S, et al., 2019. Pellino1 regulates reversible ATM activation via NBS1 ubiquitination at DNA double-strand breaks. Nat Commun, 10:1577. https://doi.org/10.1038/s41467-019-09641-9
Han JH, Huang J, 2020. DNA double-strand break repair pathway choice: the fork in the road. Genome Instab Dis, 1(1):10–19. https://doi.org/10.1007/s42764-019-00002-w
He YJ, Meghani K, Caron MC, et al., 2018. DYNLL1 binds to MRE11 to limit DNA end resection in BRCA1-deficient cells. Nature, 563(7732):522–526. https://doi.org/10.1038/s41586-018-0670-5
Huertas P, 2010. DNA resection in eukaryotes: deciding how to fix the break. Nat Struct Mol Biol, 17(1):11–16. https://doi.org/10.1038/nsmb.1710
Jette N, Lees-Miller SP, 2015. The DNA-dependent protein kinase: a multifunctional protein kinase with roles in DNA double strand break repair and mitosis. Prog Biophys Mol Biol, 117(2–3):194–205. https://doi.org/10.1016/j.pbiomolbio.2014.12.003
Jin MH, Oh DY, 2019. ATM in DNA repair in cancer. Pharmacol Ther, 203:107391. https://doi.org/10.1016/j.pharmthera.2019.07.002
Kowalczykowski SC, 2015. An overview of the molecular mechanisms of recombinational DNA repair. Cold Spring Harb Perspect Biol, 7(11):a016410. https://doi.org/10.1101/cshperspect.a016410
Lafrance-Vanasse J, Williams GJ, Tainer JA, 2015. Envisioning the dynamics and flexibility of Mre11-Rad50-Nbs1 complex to decipher its roles in DNA replication and repair. Prog Biophys Mol Biol, 117(2–3):182–193. https://doi.org/10.1016/j.pbiomolbio.2014.12.004
Lamarche BJ, Orazio NI, Weitzman MD, 2010. The MRN complex in double-strand break repair and telomere maintenance. FEBS Lett, 584(17):3682–3695. https://doi.org/10.1016/j.febslet.2010.07.029
Langerak P, Mejia-Ramirez E, Limbo O, et al., 2011. Release of Ku and MRN from DNA ends by Mre11 nuclease activity and Ctp1 is required for homologous recombination repair of double-strand breaks. PLoS Genet, 7(9): e1002271. https://doi.org/10.1371/journal.pgen.1002271
Lavin MF, Kozlov S, Gatei M, et al., 2015. ATM-dependent phosphorylation of all three members of the MRN complex: from sensor to adaptor. Biomolecules, 5(4):2877–2902. https://doi.org/10.3390/biom5042877
Lee KY, Im JS, Shibata E, et al., 2015. MCM8-9 complex promotes resection of double-strand break ends by MRE11-RAD50-NBS1 complex. Nat Commun, 6:7744. https://doi.org/10.1038/ncomms8744
Li YH, Wang JY, Zhou G, et al., 2017. Nonhomologous end-joining with minimal sequence loss is promoted by the Mre11-Rad50-Nbs1-Ctp1 complex in Schizosaccharomyces pombe. Genetics, 206(1):481–496. https://doi.org/10.1534/genetics.117.200972
Liu T, Huang J, 2014. Quality control of homologous recombination. Cell Mol Life Sci, 71(19):3779–3797. https://doi.org/10.1007/s00018-014-1649-5
Liu T, Huang J, 2016. DNA end resection: facts and mechanisms. Genomics Proteomics Bioinform, 14(3): 126–130. https://doi.org/10.1016/j.gpb.2016.05.002
Nakada D, Matsumoto K, Sugimoto K, 2003. ATM-related Tel1 associates with double-strand breaks through an Xrs2-dependent mechanism. Genes Dev, 17(16): 1957–1962. https://doi.org/10.1101/gad.1099003
Panier S, Boulton SJ, 2014. Double-strand break repair: 53BP1 comes into focus. Nat Rev Mol Cell Biol, 15(1): 7–18. https://doi.org/10.1038/nrm3719
Patel DS, Misenko SM, Her J, et al., 2017. BLM helicase regulates DNA repair by counteracting RAD51 loading at DNA double-strand break sites. J Cell Biol, 216(11): 3521–3534. https://doi.org/10.1083/jcb.201703144
Prakash R, Zhang Y, Feng WR, et al., 2015. Homologous recombination and human health: the roles of BRCA1, BRCA2, and associated proteins. Cold Spring Harb Perspect Biol, 7(4):a016600. https://doi.org/10.1101/cshperspect.a016600
Rass E, Grabarz A, Plo I, et al., 2009. Role of Mre11 in chromosomal nonhomologous end joining in mammalian cells. Nat Struct Mol Biol, 16(8):819–824. https://doi.org/10.1038/nsmb.1641
Rojowska A, Lammens K, Seifert FU, et al., 2014. Structure of the Rad50 DNA double-strand break repair protein in complex with DNA. EMBO J, 33(23):2847–2859. https://doi.org/10.15252/embj.201488889
Runge KW, Li YH, 2018. A curious new role for MRN in Schizosaccharomyces pombe non-homologous end-joining. Curr Genet, 64(2):359–364. https://doi.org/10.1007/s00294-017-0760-1
Rupnik A, Grenon M, Lowndes N, 2008. The MRN complex. Curr Biol, 18(11):R455–R457. https://doi.org/10.1016/j.cub.2008.03.040
Sallmyr A, Tomkinson AE, 2018. Repair of DNA doublestrand breaks by mammalian alternative end-joining pathways. J Biol Chem, 293(27):10536–10546. https://doi.org/10.1074/jbc.TM117.000375
Schiller CB, Seifert FU, Linke-Winnebeck C, et al., 2014. Structural studies of DNA end detection and resection in homologous recombination. Cold Spring Harb Perspect Biol, 6(10):a017962. https://doi.org/10.1101/cshperspect.a017962
Scully R, Panday A, Elango R, et al., 2019. DNA doublestrand break repair-pathway choice in somatic mammalian cells. Nat Rev Mol Cell Biol, 20(11):698–714. https://doi.org/10.1038/s41580-019-0152-0
Sedghi M, Salari M, Moslemi AR, et al., 2018. Ataxiatelangiectasia-like disorder in a family deficient for MRE11A, caused by a MRE11 variant. Neurol Genet, 4(6):e295. https://doi.org/10.1212/NXG.0000000000000295
Sharma A, Singh K, Almasan A, 2012. Histone H2AX phosphorylation: a marker for DNA damage. In: Bjergbæk L (Ed.), DNA Repair Protocols. Methods in Molecular Biology. Humana Press, Totowa, NJ, p.613–626. https://doi.org/10.1007/978-1-61779-998-3_40
Shibata A, Moiani D, Arvai AS, et al., 2014. DNA doublestrand break repair pathway choice is directed by distinct MRE11 nuclease activities. Mol Cell, 53(1):7–18. https://doi.org/10.1016/j.molcel.2013.11.003
Stracker TH, Petrini JHJ, 2011. The MRE11 complex: starting from the ends. Nat Rev Mol Cell Biol, 12(2):90–103. https://doi.org/10.1038/nrm3047
Tatebe H, Lim CT, Konno H, et al., 2020. Rad50 zinc hook functions as a constitutive dimerization module interchangeable with SMC hinge. Nat Commun, 11:370. https://doi.org/10.1038/s41467-019-14025-0
Tikoo S, Madhavan V, Hussain M, et al., 2013. Ubiquitin-dependent recruitment of the Bloom Syndrome helicase upon replication stress is required to suppress homologous recombination. EMBO J, 32(12):1778–1792. https://doi.org/10.1038/emboj.2013.117
Tripathi V, Agarwal H, Priya S, et al., 2018. MRN complex-dependent recruitment of ubiquitylated BLM helicase to DSBs negatively regulates DNA repair pathways. Nat Commun, 9:1016. https://doi.org/10.1038/s41467-018-03393-8
Wang QH, Goldstein M, Alexander P, et al., 2014. Rad17 recruits the MRE11-RAD50-NBS1 complex to regulate the cellular response to DNA double-strand breaks. EMBO J, 33(8):862–877. https://doi.org/10.1002/embj.201386064
Wang ZF, Gong YM, Peng B, et al., 2019. MRE11 UFMylation promotes ATM activation. Nucleic Acids Res, 47(8):4124–4135. https://doi.org/10.1093/nar/gkz110
Williams RS, Moncalian G, Williams JS, et al., 2008. Mre11 dimers coordinate DNA end bridging and nuclease processing in double-strand-break repair. Cell, 135(1):97–109. https://doi.org/10.1016/j.cell.2008.08.017
Williams RS, Dodson GE, Limbo O, et al., 2009. Nbs1 flexibly tethers Ctp1 and Mre11-Rad50 to coordinate DNA double-strand break processing and repair. Cell, 139(1): 87–99. https://doi.org/10.1016/j.cell.2009.07.033
Xie AY, Kwok A, Scully R, 2009. Role of mammalian Mre11 in classical and alternative nonhomologous end joining. Nat Struct Mol Biol, 16(8):814–818. https://doi.org/10.1038/nsmb.1640
Xie MH, Park D, You S, et al., 2015. Bcl2 inhibits recruitment of Mre11 complex to DNA double-strand breaks in response to high-linear energy transfer radiation. Nucleic Acids Res, 43(2):960–972. https://doi.org/10.1093/nar/gku1358
Zhang B, Tang ZH, Li LJ, et al., 2020. NBS1 is required for SPO11-linked DNA double-strand break repair in male meiosis. Cell Death Differ, 27(7):2176–2190. https://doi.org/10.1038/s41418-020-0493-4
Zhu Z, Chung WH, Shim EY, et al., 2008. Sgs1 helicase and two nucleases Dna2 and Exo1 resect DNA double-strand break ends. Cell, 134(6):981–94. https://doi.org/10.1016/j.cell.2008.08.037
Acknowledgments
This reasearch was supported by the National Key Research and Development Program of China (No. 2018YFC2000100), the National Natural Science Foundation of China (Nos. 31730021, 31971220, and 31961160725), the Fok Ying Tung Education Foundation, and the China’s Fundamental Research Funds for the Central Universities. We apologize to colleagues whose work could not be cited due to space limitations. We thank all our colleagues in the Huang laboratory for the insightful discussions.
Author information
Authors and Affiliations
Contributions
Shan QIU wrote the manuscript. Jun HUANG reviewed and edited the manuscript. Both authors have read and approved the final manuscript and take responsibility for its integrity.
Corresponding author
Additional information
Compliance with ethics guidelines
Shan QIU and Jun HUANG declare that they have no conflict of interest.
This article does not contain any studies with human or animal subjects performed by either of the authors.
Rights and permissions
About this article
Cite this article
Qiu, S., Huang, J. MRN complex is an essential effector of DNA damage repair. J. Zhejiang Univ. Sci. B 22, 31–37 (2021). https://doi.org/10.1631/jzus.B2000289
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B2000289