Abstract
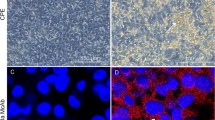
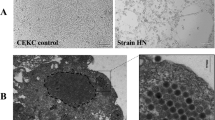
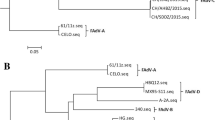
Fowl adenovirus serotype 4 (FAdV-4) strain SD1511 was isolated from chickens with severe inclusion body hepatitis and hydropericardium syndrome in Shandong Province, China. The isolate was cultured in primary chicken embryo kidney cells. A study of pathogenicity indicated that SD1511 readily infected 7–35-d-old chickens by intramuscular injection and intranasal and oral routes, causing 50%–100% mortality. The 35-d-old chickens suffered more severe infection than 7- and 21-d-old chickens with mortality highest in the intramuscular injection group. The serum from surviving chickens showed potent viral neutralizing capability. The complete genome of SD1511 was sequenced and analyzed. The strain was found to belong to the FAdV-4 cluster with more than 99% identity with the virulent FAdV-4 strains isolated in China in recent years except for some distinct variations, including deletions of open reading frame 27 (ORF27), ORF48, and part of ORF19. Our findings suggest that SD1511 might be used as a prototype strain for the study of pathogenesis and vaccine development.
抽象
目 的
了解禽腺病毒4 型 (FAdV-4) 分离株在口服、滴 鼻点眼和肌注三种感染途径下对 7∼35 日龄鸡的 致病性, 并分析该分离株与其他参考毒株在基因 组水平上的差异。
创新点
用原代鸡胚肾细胞 (CEK) 成功培养了 FAdV-4 分离株。发现除了常用的口服和肌注途径, FAdV-4 也可通过滴鼻点眼接种途径感染 7、21 和35 日龄鸡。肌注方法最为敏感, 导致感染鸡 100%死亡; 35 日龄鸡较7 和 21 日龄鸡表现出更 明显的临床症状和剖检病变。
方 法
通过口服、滴鼻点眼和肌注三种途径将 FAdV-4 分离株的 CEK 细胞培养物接种 7∼35 日龄的无特 定病原体 (SPF) 鸡。通过临床症状观察、排毒 检测、病理解剖、免疫组化和组织中毒载量测定 来判断分离株的致病性及病毒在组织脏器中的分 布情况。通过病毒中和试验检测感染存活鸡的血 清中和抗体。通过全基因组测序分析, 比较 FAdV-4 分离株与国内外参考毒株的基因组序列差异。
结 论
本研究分离鉴定的 FAdV-4 分离株 SD1511 可以 在 CEK 细胞上适应, 并可以通过口服、滴鼻点 眼和肌注三种感染途径引起 7∼35 日龄的 SPF 鸡 50%∼100% 的死亡, 其中肌注最为敏感, 死亡率 达 100%。感染鸡持续排毒至感染后的40 天, 产 生具有明显中和作用的中和抗体。全基因组测序 分析结果显示, SD1511 基因组与最近国内分离 的 FAdV-4 具有很高的同源性, 但与国外的分离 株有明显的差异。
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Alexander HS, Huber P, Cao J, et al., 1998. Growth characteristics of fowl adenovirus type 8 in a chicken hepatoma cell line. J Virol Methods, 74(1):9–14. https://doi.org/10.1016/s0166-0934(98)00062-7
Bose RTD, Grumbles LC, 1959. The relationship between quail bronchitis virus and chicken embryo lethal orphan virus. Avian Dis, 3(3):321–344. https://doi.org/10.2307/1587679
Choi KS, Kye SJ, Kim JY, et al., 2012. Epidemiological investigation of outbreaks of fowl adenovirus infection in commercial chickens in Korea. Poult Sci, 91(10):2502–2506. https://doi.org/10.3382/ps.2012-02296
Chomiak TW, Luginbuhl RE, Helmboldt CF, 1961. Tissue culture propagation and pathology of celo virus. Avian Dis, 5(3):313–320. https://doi.org/10.2307/1587640
Ganesh K, Raghavan R, 2000. Hydropericardium hepatitis syndrome of broiler poultry: current status of research. Res Vet Sci, 68(3):201–206. https://doi.org/10.1053/rvsc.1999.0365
Grgić H, Yang DH, Nagy É, 2011. Pathogenicity and complete genome sequence of a fowl adenovirus serotype 8 isolate. Virus Res, 156(1–2):91–97. https://doi.org/10.1016/j.virusres.2011.01.002
Hess M, 2000. Detection and differentiation of avian adenoviruses: a review. Avian Pathol, 29(3):195–206. https://doi.org/10.1080/03079450050045440
Jaffery MS, 1988. A treatise on angara disease (hydropericardiumpulmonary oedema-hepatonephritis syndrome). J Pak Vet Med Assoc, 34:1–33.
Kaján GL, Kecskeméti S, Harrach B, et al., 2013. Molecular typing of fowl adenoviruses, isolated in hungary recently, reveals high diversity. Vet Microbiol, 167(3–4):357–363. https://doi.org/10.1016/j.vetmic.2013.09.025
Li HX, Wang J, Qiu LY, et al., 2016. Fowl adenovirus species C serotype 4 is attributed to the emergence of hepatitishydropericardium syndrome in chickens in China. Infect Genet Evol, 45:230–241. https://doi.org/10.1016/j.meegid.2016.09.006
Li HY, Yin YB, Guo YY, et al., 2010. Isolation and PCR identification of 12 strains of inclusion body hepatitis virus from clinical broilers. Vet Sci China, 40(7):722–727 (in Chinese). https://doi.org/10.16656/j.issn.1673-4696.2010.07.013
Li PH, Zheng PP, Zhang TF, et al., 2017. Fowl adenovirus serotype 4: epidemiology, pathogenesis, diagnostic detection, and vaccine strategies. Poult Sci, 96(8):2630–2640. https://doi.org/10.3382/ps/pex087
Liu YK, Wan WY, Gao DS, et al., 2016. Genetic characterization of novel fowl aviadenovirus 4 isolates from outbreaks of hepatitis-hydropericardium syndrome in broiler chickens in China. Emerg Microbes Infect, 5(1):1–8. https://doi.org/10.1038/emi.2016.115
Lund S, Holman GD, Schmitz O, et al., 1995. Contraction stimulates translocation of glucose transporter GLUT4 in skeletal muscle through a mechanism distinct from that of insulin. Proc Natl Acad Sci USA, 92(13):5817–5821. https://doi.org/10.1073/pnas.92.13.5817
Mase M, Nakamura K, Minami F, 2012. Fowl adenoviruses isolated from chickens with inclusion body hepatitis in Japan, 2009–2010. J Vet Med Sci, 74(8):1087–1089. https://doi.org/10.1292/jvms.11-0443
Mazaheri A, Prusas C, Voß M, et al., 1998. Some strains of serotype 4 fowl adenoviruses cause inclusion body hepatitis and hydropericardium syndrome in chickens. Avian Pathol, 27(3):269–276. https://doi.org/10.1080/03079459808419335
Mittal D, Jindal N, Tiwari AK, et al., 2014. Characterization of fowl adenoviruses associated with hydropericardium syndrome and inclusion body hepatitis in broiler chickens. VirusDisease, 25(1):114–119. https://doi.org/10.1007/s13337-013-0183-7
Ojkic D, Martin E, Swinton J, et al., 2008. Genotyping of Canadian isolates of fowl adenoviruses. Avian Pathol, 37(1):95–100. https://doi.org/10.1080/03079450701805324
Park HS, Lim IS, Kim SK, et al., 2017. Molecular analysis of the hexon, penton base, and fiber-2 genes of Korean fowl adenovirus serotype 4 isolates from hydropericardium syndrome-affected chickens. Virus Genes, 53(1):111–116. https://doi.org/10.1007/s11262-016-1393-z
Schachner A, Matos M, Grafl B, et al., 2018. Fowl adenovirus-induced diseases and strategies for their control—a review on the current global situation. Avian Pathol, 47(2):111–126. https://doi.org/10.1080/03079457.2017.1385724
Schat K, Purchase HG, 1989. A Laboratory Manual for the Isolation and Identification of Avian Pathogens, 3rd Ed. American Association of Avian Pathologists, Kennett Square, PA, American, p.176–181.
Ye JQ, Liang GC, Zhang JJ, et al., 2016. Outbreaks of serotype 4 fowl adenovirus with novel genotype, China. Emerg Microbes Infect, 5(1):1–12. https://doi.org/10.1038/emi.2016.50
Zhao J, Zhong Q, Zhao Y, et al., 2015. Pathogenicity and complete genome characterization of fowl adenoviruses isolated from chickens associated with inclusion body hepatitis and hydropericardium syndrome in China. PLoS ONE, 10(7):e0133073. https://doi.org/10.1371/journal.pone.0133073
Acknowledgments
We thank Professor Guo-zhong ZHANG (Key Laboratory of Animal Epidemiology and Zoonosis, Ministry of Agriculture, College of Veterinary Medicine, China Agricultural University, Beijing, China) for providing chicken anti-FAdV hyperimmune serum.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the National Key Technology Research and Development Program of China (No. 2015BAD12B01) and the China Agriculture Research System (No. CARS-40-K13)
Compliance with ethics guidelines
Kai-kun MO, Chen-fei LYU, Shang-shang CAO, Xia LI, Gang XING, Yan YAN, Xiao-juan ZHENG, Min LIAO, and Ji-yong ZHOU declare that they have no conflict of interest.
All institutional and national guidelines for the care and use of laboratory animals were followed.
Electronic supplementary materials
Rights and permissions
About this article
Cite this article
Mo, Kk., Lyu, Cf., Cao, Ss. et al. Pathogenicity of an FAdV-4 isolate to chickens and its genomic analysis. J. Zhejiang Univ. Sci. B 20, 740–752 (2019). https://doi.org/10.1631/jzus.B1900070
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B1900070
Key words
- Fowl adenovirus serotype 4 (FAdV-4)
- Pathogenicity
- Infection way
- Virus neutralization activity
- Genome analysis