Abstract
Objective
To investigate the analgesic effects of electroacupuncture (EA) at 2 and 100 Hz on type 2 diabetic neuropathic pain (DNP) and on the expressions of the P2X3 receptor and calcitonin gene-related peptide (CGRP) in the dorsal root ganglion (DRG).
Methods
Rat type 2 DNP was induced by a high calorie and high sugar diet fed for 7 weeks, plus a single intraperitoneal injection of streptozotocin (STZ) after 5 weeks. EA at 2 and 100 Hz was carried out once every day after 7 weeks for 7 consecutive days. Body weight, serum fasting insulin (FINS), fasting blood glucose (FBG), insulin sensitivity index (ISI), and paw withdrawal latency (PWL) were measured. The expressions of L4–L6 DRG P2X3 receptors and CGRP were assessed by immunofluorescence.
Results
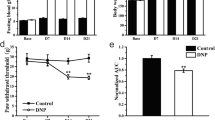
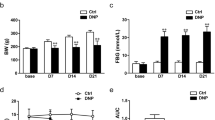
Type 2 DNP was successfully induced as shown by the increased body weight, FINS, and FBG, as well as the reduced ISI and PWL. Expressions of P2X3 receptors and CGRP in L4–L6 DRGs increased. EA at both 2 and 100 Hz relieved type 2 DNP, but the analgesic effect of EA was stronger at 2 Hz. P2X3 receptor expression decreased in L4–L6 DRGs following EA at 2 Hz and in L5 and L6 DRGs following EA at 100 Hz. EA at both 2 and 100 Hz down-regulated CGRP overexpression in L4–L6 DRGs.
Conclusions
These findings indicate that EA at 2 Hz is a good option for the management of type 2 DNP. The EA effect may be related to its down-regulation of the overexpressions of the DRG P2X3 receptors and CGRP in this condition.
概要
目的
研究2 和100 Hz 电针对2 型糖尿病神经痛(DNP)大鼠的镇痛作用,以及对DNP 大鼠背根神经节(DRG)上P2X3 受体和降钙素相关基因肽(CGRP)表达的影响。
创新点
本研究首次选用2 型DNP 大鼠模型来研究电针治疗DNP 的优势频率。研究证明2 Hz 电针对2 型DNP 大鼠的镇痛作用以及对DRG 上高表达的P2X3 抑制作用优于100 Hz,为电针治疗DNP 及其频率选择提供科学依据和阐释。
方法
将雄性SD 大鼠分为对照组(腹腔注射柠檬酸钠缓冲液)和模型组(高脂高糖饲养联合腹腔注射35 mg/kg 链脲佐菌素(STZ))。模型组根据采用不同的电针频率分为以下三组:DNP 组、DNP+2 Hz 电针组、DNP+100 Hz 电针组。采用足底测试仪检测大鼠足跖热痛阈(PWL),用酶联免疫吸附测定(ELISA)试剂盒检测大鼠空腹胰岛素(FINS)含量,用免疫荧光法检测DRG 上的P2X3 受体和CGRP 的表达水平。
结论
2 和100 Hz 电针对2 型DNP 均有明显的镇痛作用,且2 Hz 电针对2 型DNP 的镇痛作用优于100 Hz 电针(图2)。免疫荧光实验结果显示,2和100 Hz 电针均能明显下调DRG 中P2X3 受体和CGRP 的高表达(图3 和4),且2 Hz 电针对DRG 中P2X3 受体的抑制作用优于100 Hz 电针(图3)。综上所述,2 Hz 电针是治疗2 型DNP较为理想的治疗手段。
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Alberti, K.G., Zimmet, P.Z., 1998. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabet. Med., 15(7):539–553. http://dx.doi.org/10.1002/(SICI)1096-9136(199807)15:7<539::AID-DIA668>3.0.CO;2-S
Bai, J., Zheng, S., Jiang, D., et al., 2015. Oxidative stress contributes to abnormal glucose metabolism and insulin sensitivity in two hyperlipidemia models. Int. J. Clin. Exp. Pathol., 8(10):13193–13200.
Bernardini, N., Neuhuber, W., Reeh, P.W., et al., 2004. Morphological evidence for functional capsaicin receptor expression and calcitonin gene-related peptide exocytosis in isolated peripheral nerve axons of the mouse. Neuroscience, 126(3):585–590. http://dx.doi.org/10.1016/j.neuroscience.2004.03.017
Brown, M.J., Asbury, A.K., 1984. Diabetic neuropathy. Ann. Neurol., 15(1):2–12. http://dx.doi.org/10.1002/ana.410150103
Brussee, V., Guo, G., Dong, Y., et al., 2008. Distal degenerative sensory neuropathy in a long-term type 2 diabetes rat model. Diabetes, 57(6):1664–1673. http://dx.doi.org/10.2337/db07-1737
Cady, R.J., Glenn, J.R., Smith, K.M., et al., 2011. Calcitonin gene-related peptide promotes cellular changes in trigeminal neurons and glia implicated in peripheral and central sensitization. Mol. Pain, 7:94. http://dx.doi.org/10.1186/1744-8069-7-94
Chen, M., Gu, J.G., 2005. A P2X receptor-mediated nociceptive afferent pathway to lamina I of the spinal cord. Mol. Pain, 1:4. http://dx.doi.org/10.1186/1744-8069-1-4
Cheng, R.S., Pomeranz, B., 1979. Electroacupuncture analgesia could be mediated by at least two pain-relieving mechanisms; endorphin and non-endorphin systems. Life Sci., 25(23):1957–1962. http://dx.doi.org/10.1016/0024-3205(79)90598-8
Cook, S.P., Vulchanova, L., Hargreaves, K.M., et al., 1997. Distinct ATP receptors on pain-sensing and stretchsensing neurons. Nature, 387(6632):505–508. http://dx.doi.org/10.1038/387505a0
Costigan, M., Scholz, J., Woolf, C.J., 2009. Neuropathic pain: a maladaptive response of the nervous system to damage. Annu. Rev. Neurosci., 32:1–32. http://dx.doi.org/10.1146/annurev.neuro.051508.135531
Dang, J.K., Wu, Y., Cao, H., et al., 2014. Establishment of a rat model of type II diabetic neuropathic pain. Pain Med., 15(4):637–646. http://dx.doi.org/10.1111/pme.12387_1
Dyck, P.J., Kratz, K.M., Karnes, J.L., et al., 1993. The prevalence by staged severity of various types of diabetic neuropathy, retinopathy, and nephropathy in a populationbased cohort: the Rochester Diabetic Neuropathy Study. Neurology, 43(4):817–824. http://dx.doi.org/10.1212/WNL.43.4.817
El-Moselhy, M.A., Taye, A., Sharkawi, S.S., et al., 2011. The antihyperglycemic effect of curcumin in high fat diet fed rats. Role of TNF-α and free fatty acids. Food Chem. Toxicol., 49(5):1129–1140. http://dx.doi.org/10.1016/j.fct.2011.02.004
Fang, J.Q., Du, J.Y., Liang, Y., et al., 2013a. Intervention of electroacupuncture on spinal p38 MAPK/ATF-2/VR-1 pathway in treating inflammatory pain induced by CFA in rats. Mol. Pain, 9:13. http://dx.doi.org/10.1186/1744-8069-9-13
Fang, J.Q., Jiang, Y.L., Qiu, S.C., et al., 2013b. Involvement of peripheral beta-endorphin and mu, delta, kappa opioid receptors in electro acupuncture analgesia for prolonged inflammatory pain of rats. Eur. J. Inflamm., 11(2):375–383. http://dx.doi.org/10.1177/1721727X1301100208
Gallou-Kabani, C., Vigé, A., Gross, M.S., et al., 2007. C57BL/6J and A/J mice fed a high-fat diet delineate components of metabolic syndrome. Obesity, 15(8):1996–2005. http://dx.doi.org/10.1038/oby.2007.238
Gooch, C., Podwall, D., 2004. The diabetic neuropathies. Neurologist, 10(6):311–322. http://dx.doi.org/10.1097/01.nrl.0000144733.61110.25
Harati, Y., 1996. Diabetes and the nervous system. Endocrinol. Metab. Clin. North Am., 25(2):325–359. http://dx.doi.org/10.1016/S0889-8529(05)70327-3
Hayden, M.R., Tyagi, S.C., Kerklo, M.M., et al., 2005. Type 2 diabetes mellitus as a conformational disease. JOP, 6(4):287–302.
Hong, S., Morrow, T.J., Paulson, P.E., et al., 2004. Early painful diabetic neuropathy is associated with differential changes in tetrodotoxin-sensitive and-resistant sodium channels in dorsal root ganglion neurons in the rat. J. Biol. Chem., 279(28):29341–29350. http://dx.doi.org/10.1074/jbc.M404167200
Hwang, H.S., Yang, E.J., Lee, S.M., et al., 2011. Antiallodynic effects of electroacupuncture combined with MK-801 treatment through the regulation of p35/p25 in experimental diabetic neuropathy. Exp. Neurobiol., 20(3):144–152. http://dx.doi.org/10.5607/en.2011.20.3.144
Inoue, K., 2006. The function of microglia through purinergic receptors: neuropathic pain and cytokine release. Pharmacol. Ther., 109(1–2):210–226. http://dx.doi.org/10.1016/j.pharmthera.2005.07.001
Ishiko, N., Yamamoto, T., Murayama, N., et al., 1978. Electroacupuncture: current strength-duration relationship for initiation of hypesthesia in man. Neurosci. Lett., 8(4):273–276.
Jagodic, M.M., Pathirathna, S., Nelson, M.T., et al., 2007. Cell-specific alterations of T-type calcium current in painful diabetic neuropathy enhance excitability of sensory neurons. J. Neurosci., 27(12):3305–3316. http://dx.doi.org/10.1523/JNEUROSCI.4866-06.2007
Jiang, Y.L., Ning, Y., Liu, Y.Y., et al., 2011. Effects of preventive acupuncture on streptozotocin-induced hyperglycemia in rats. J. Endocrinol. Invest., 34(10):e355–e361. http://dx.doi.org/10.3275/7859
Jiang, Y.L., Yin, X.H., Shen, Y.F., et al., 2013. Low frequency electroacupuncture alleviated spinal nerve ligation induced mechanical allodynia by inhibiting TRPV1 upregulation in ipsilateral undamaged dorsal root ganglia in rats. Evid. Based Complement. Alternat. Med., 2013:170910. http://dx.doi.org/10.1155/2013/170910
Jiang, Y.L., He, X.F., Shen, Y.F., et al., 2015. Analgesic roles of peripheral intrinsic met-enkephalin and dynorphin A in long-lasting inflammatory pain induced by complete Freund’s adjuvant in rats. Exp. Ther. Med., 9(6):2344–2348. http://dx.doi.org/10.3892/etm.2015.2407
Jimenez-Andrade, J.M., Bloom, A.P., Stake, J.I., et al., 2010. Pathological sprouting of adult nociceptors in chronic prostate cancer-induced bone pain. J. Neurosci., 30(44):14649–14656. http://dx.doi.org/10.1523/JNEUROSCI.3300-10.2010
Kahn, B.B., 1998. Type 2 diabetes: when insulin secretion fails to compensate for insulin resistance. Cell, 92(5):593–596. http://dx.doi.org/10.1016/S0092-8674(00)81125-3
Khan, G.M., Chen, S.R., Pan, H.L., 2002. Role of primary afferent nerves in allodynia caused by diabetic neuropathy in rats. Neuroscience, 114(2):291–299. http://dx.doi.org/10.1016/S0306-4522(02)00372-X
Kobayashi, M., Ohno, T., Tsuchiya, T., et al., 2004. Characterization of diabetes-related traits in MSM and JF1 mice on high-fat diet. J. Nutr. Biochem., 15(10):614–621. http://dx.doi.org/10.1016/j.jnutbio.2004.05.001
Lin, Y., Sun, Z., 2010. Current views on type 2 diabetes. J. Endocrinol., 204(1):1–11. http://dx.doi.org/10.1677/JOE-09-0260
Manni, L., Florenzano, F., Aloe, L., 2011. Electroacupuncture counteracts the development of thermal hyperalgesia and the alteration of nerve growth factor and sensory neuromodulators induced by streptozotocin in adult rats. Diabetologia, 54(7):1900–1908. http://dx.doi.org/10.1007/s00125-011-2117-5
Maser, R.E., Steenkiste, A.R., Dorman, J.S., et al., 1989. Epidemiological correlates of diabetic neuropathy: report from Pittsburgh epidemiology of diabetes complications study. Diabetes, 38(11):1456–1461. http://dx.doi.org/10.2337/diab.38.11.1456
Migita, K., Moriyama, T., Koguchi, M., et al., 2009. Modulation of P2X receptors in dorsal root ganglion neurons of streptozotocin-induced diabetic neuropathy. Neurosci. Lett., 452(2):200–203. http://dx.doi.org/10.1016/j.neulet.2009.01.048
Muniyappa, R., Lee, S., Chen, H., et al., 2008. Current approaches for assessing insulin sensitivity and resistance in vivo: advantages, limitations, and appropriate usage. Am. J. Physiol. Endocrinol. Metab., 294(1):e15-E26. http://dx.doi.org/10.1152/ajpendo.00645.2007
Muoio, D.M., Newgard, C.B., 2008. Molecular and metabolic mechanisms of insulin resistance and β-cell failure in type 2 diabetes. Nat. Rev. Mol. Cell Biol., 9(3):193–205. http://dx.doi.org/10.1038/nrm2327
Nori, S.L., Rocco, M.L., Florenzano, F., et al., 2013. Increased nerve growth factor signaling in sensory neurons of early diabetic rats is corrected by electroacupuncture. Evid. Based Complement. Alternat. Med., 2013(12):652735. http://dx.doi.org/10.1155/2013/652735
Pabbidi, R.M., Cao, D.S., Parihar, A., et al., 2008. Direct role of streptozotocin in inducing thermal hyperalgesia by enhanced expression of transient receptor potential vanilloid 1 in sensory neurons. Mol. Pharmacol., 73(3):995–1004. http://dx.doi.org/10.1124/mol.107.041707
Petruska, J.C., Cooper, B.Y., Johnson, R.D., et al., 2000. Distribution patterns of different P2x receptor phenotypes in acutely dissociated dorsal root ganglion neurons of adult rats. Exp. Brain Res., 134(1):126–132. http://dx.doi.org/10.1007/s002210000414
Price, T.J., Flores, C.M., 2007. Critical evaluation of the colocalization between calcitonin gene-related peptide, substance P, transient receptor potential vanilloid subfamily type 1 immunoreactivities, and isolectin B4 binding in primary afferent neurons of the rat and mouse. J. Pain, 8(3):263–272. http://dx.doi.org/10.1016/j.jpain.2006.09.005
Raddant, A.C., Russo, A.F., 2011. Calcitonin gene-related peptide in migraine: intersection of peripheral inflammation and central modulation. Expert Rev. Mol. Med., 13(201):e36. http://dx.doi.org/10.1017/S1462399411002067
Sharma, S., Kulkarni, S.K., Agrewala, J.N., et al., 2006. Curcumin attenuates thermal hyperalgesia in a diabetic mouse model of neuropathic pain. Eur. J. Pharmacol., 536(3):256–261. http://dx.doi.org/10.1016/j.ejphar.2006.03.006
Silva, J.R.T., Silva, M.L., Prado, W.A., 2011. Analgesia induced by 2-or 100-Hz electroacupuncture in the rat tail-flick test depends on the activation of different descending pain inhibitory mechanisms. J. Pain, 12(1):51–60. http://dx.doi.org/10.1016/j.jpain.2010.04.008
Simonetti, M., Giniatullin, R., Fabbretti, E., 2008. Mechanisms mediating the enhanced gene transcription of P2X3 receptor by calcitonin gene-related peptide in trigeminal sensory neurons. J. Biol. Chem., 283(27):18743–18752. http://dx.doi.org/10.1074/jbc.M800296200
Surwit, R.S., Kuhn, C.M., Cochrane, C., et al., 1988. Dietinduced type II diabetes in C57BL/6J mice. Diabetes, 37(9):1163–1167. http://dx.doi.org/10.2337/diab.37.9.1163
Weisberg, S.P., Leibel, R., Tortoriello, D.V., 2008. Dietary curcumin significantly improves obesity-associated inflammation and diabetes in mouse models of diabesity. Endocrinology, 149(7):3549–3558. http://dx.doi.org/10.1210/en.2008-0262
Wild, S., Roglic, G., Green, A., et al., 2004. Global prevalence of diabetes: estimates for the year 2000 and projections for 2030. Diabetes Care, 27(5):1047–1053. http://dx.doi.org/10.2337/diacare.27.5.1047
Winzell, M.S., Ahrén, B., 2004. The high-fat diet-fed mouse: a model for studying mechanisms and treatment of impaired glucose tolerance and type 2 diabetes. Diabetes, 53(Suppl. 3):S215–S219. http://dx.doi.org/10.2337/diabetes.53.suppl_3.S215
Xu, G.Y., Li, G., Liu, N., et al., 2011. Mechanisms underlying purinergic P2X3 receptor-mediated mechanical allodynia induced in diabetic rats. Mol. Pain, 7(1):60. http://dx.doi.org/10.1186/1744-8069-7-60
Zimmet, P., Alberti, K.G., Shaw, J., 2001. Global and societal implications of the diabetes epidemic. Nature, 414(6865):782–787. http://dx.doi.org/10.1038/414782a
Acknowledgements
We would like to thank all the rats for giving their lives. We thank Qi-yang SHOU (Zhejiang Chinese Medical University, Hangzhou, China) for technical assistance.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Project supported by the National Natural Science Foundation of China (No. 81303039), the Specialized Research Fund for the Doctoral Program of Higher Education (No. 20133322120001), the Zhejiang Postdoctoral Science Foundation (No. BSH1302083), the Zhejiang Province Top Key Discipline of Chinese Medicine-Acupuncture & Tuina (No. [2012]80), and the Key Science and Technology Innovation Team of Zhejiang Province (No. 2013TD15), China
ORCID: Yong-liang JIANG, http://orcid.org/0000-0002-9937-7398
Compliance with ethics guidelines
Xiao-fen HE, Jun-jun WEI, Sheng-yun SHOU, Jian-qiao FANG, and Yong-liang JIANG declare that they have no conflict of interest.
All institutional and national guidelines for the care and use of laboratory animals were followed.
Rights and permissions
About this article
Cite this article
He, Xf., Wei, Jj., Shou, Sy. et al. Effects of electroacupuncture at 2 and 100 Hz on rat type 2 diabetic neuropathic pain and hyperalgesia-related protein expression in the dorsal root ganglion. J. Zhejiang Univ. Sci. B 18, 239–248 (2017). https://doi.org/10.1631/jzus.B1600247
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B1600247
Key words
- Electroacupuncture
- Type 2 diabetic neuropathic pain
- Dorsal root ganglion
- P2X3 receptor
- Calcitonin gene-related peptide