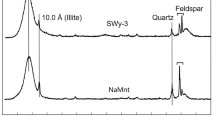
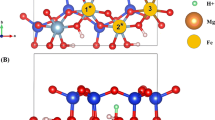
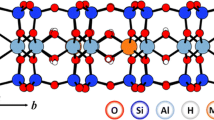
Abstract
Many important properties of clay minerals are defined by the species of charge-balancing cation. Phenomena such as clay swelling and cation exchange depend on the cation species present, and understanding how the cations bind with the mineral surface at a fundamental level is important. In the present study the binding affinities of several different charge-balancing cations with the basal surface of the smectite mineral, montmorillonite, have been calculated using molecular dynamics in conjunction with the well-tempered metadynamics algorithm. The results follow a Hofmeister series of preferred ion adsorption to the smectite basal surfaces of the form: K+ > Na+ > Ca2+ > Cs+ > Ba2+
The results also revealed the energetically favorable position of the ions above the clay basal surfaces. Key features of the free-energy profiles are illustrated by Boltzmann population inversions and analyses of the water structures surrounding the ion and clay surface. The results show that weakly hydrated cations (K+ and Cs+) preferentially form inner-sphere surface complexes (ISSC) above the ditrigonal siloxane cavities of the clay, while the more strongly hydrated cations (Na+) are able to form ISSCs above the basal O atoms of the clay surface. The strongly hydrated cations (Na+, Ca2+, and Ba2+), however, preferentially form outer-sphere surface complexes. The results provide insight into the adsorption mechanisms of several ionic species on montmorillonite and are relevant to many phenomena thought to be affected by cation exchange, such as nuclear waste disposal, herbicide/pesticide-soil interactions, and enhanced oil recovery.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.References
Aaqvist, J. (1990) Ion-water interaction potentials derived from free energy perturbation simulations. The Journal of Phyical Chemistry, 94, 8021–8024.
Barducci, A., Bussi, B., and Parrinello M. (2008) Well-tempered metadynamics: A smoothly converging and tunable free energy method. Physical Review Letters, 100, 020603.
Benson, L.V. (1982) A tabulation and evaluation of ion exchange data on smectites. Environmental Geology, 4, 23–29.
Berendsen, H.J.C, Grigera, J.R., and Straatsma, T.P. (1987) The missing term in effective pair potentials. The Journal of Physical Chemistry, 91, 6269–6271.
Bergaya, F., Theng, B., and Lagaly, G., editors (2006) Handbook of Clay Science. Elsevier Science, Amsterdam.
Boek, E.S. and Sprik, M. (2003) Ab initio molecular dynamics study of the hydration of a sodium smectite clay. The Journal of Physical Chemistry B, 107, 3251–3256.
Boek, E.S., Coveney, P.V., and Skipper, N.T. (1995) Monte Carlo molecular modeling studies of hydrated Li-, Na-, and K-smectites: Understanding the role of potassium as a clay swelling inhibitor. The Journal of the American Chemical Society, 117, 12608–12617.
Bonomi, M., Branduardi, D., Bussi, G., Camilloni, C., Provasi, D., Raiteri, P., Donadio, D., Marinelli, F., Pietrucci, F., Broglia, R.A., and Parrinello, M. (2009) PLUMED: A portable plugin for free energy calculations with molecular dynamics. Computer Physics Communications, 180, 1961–1972.
Bourg, I.C. and Sposito, G. (2011) Ion Exchange Phenomena; Handbook of Soil Science, Properties and Processes (2nd edition). CRC Press, Boca Raton, Florida, USA.
Bowers, G.M., Bish, D.L., and Kirkpatrick, R.J. (2008) H2O and cation structure and dynamics in expandable clays: 2H and 39K NMR investigations of hectorite. The Journal of Physical Chemistry C, 112, 6430–6438.
Brown, D.R. and Kevan, L. (1988) Aqueous coordination and location of exchangeable copper (2+) cations in montmorillonite clay studied by electron spin resonance and electron spin echo modulation. Journal of the American Chemical Society, 110, 2743–2748.
Chang, F.R.C., Skipper, N.T., and Sposito, G. (1995) Computer simulation of interlayer molecular structure in sodium montmorillonite hydrates. Langmuir, 11, 2734–2741.
Chen, C.C. and Hayes, K.F. (1999) X-ray absorption spectroscopy investigation of aqueous Co(II) and Sr (II) sorption at clay-water interfaces. Geochimica et Cosmochimica Acta, 63, 3205–3215.
Coveney, P.V. and Wan, S. (2016) On the calculation of equilibrium thermodynamic properties from molecular dynamics. Physical Chemistry Chemical Physics, DOI: https://doi.org/10.1039/C6CP02349E.
Cygan, R.T., Liang, J.J., and Kalinichev, A.G. (2004) Molecular models of hydroxide, oxyhydroxide, and clay phases and the development of a general force field. The Journal of Phyical Chemistry B, 108, 1255–1266.
Downs, R.T. and Hall-Wallace, M. (2003) The American Mineralogist crystal structure database. American Mineralogist, 88, 247–250.
Eisenman, G. (1962) Cation selective glass electrodes and their mode of operation. Biophysical Journal, 2, 259–323.
Gast, R.G. (1969) Standard free energies of exchange for alkali metal cations on Wyoming bentonite. Soil Science Society of America Journal, 33, 37–41.
Gast, R.G. (1972) Alkali metal cation exchange on Chambers montmorillonite. Soil Science Society of America Journal, 36, 14–19.
Greathouse, J.A., Refson, K., and Sposito, G. (2000) Molecular dynamics simulation of water mobility in magnesium-smectite hydrates. Journal of the American Chemical Society, 122, 11459–11464.
Greathouse, J.A., Hart, D.B., Bowers, G.M., Kirkpatrick, R.J., and Cygan, R.T. (2015) Molecular simulation of structure and diffusion at smectite-water interfaces: Using expanded clay interlayers as model nanopores. The Journal of Physical Chemistry C, 119, 17126–17136.
Greenwell, H.C., Jones, W., Coveney, P.V., and Stackhouse, S. (2006) On the application of computer simulation techniques to anionic and cationic clays: A materials chemistry perspective. Journal of Materials Chemistry, 16, 708–723.
Hanshaw, B.B. (1964) Cation-exchange constants for clays from electrochemical measurements. 12th Annual Meeting of the Clay Minerals Society, USA.
Heinz, H., Lin, T.-J., Mishra, R.K., and Emami, F.S. (2013) Thermodynamically consistent force fields for the assembly of inorganic, organic, and biological nanostructures: The INTERFACE force field. Langmuir, 29, 1754–1765.
Hunter, J.D. (2007) Matplotlib: A 2D graphics environment. Computing in Science and Engineering, 9, 90–95.
Koneshan, S., Lynden-Bell, R.M., and Rasaiah, J.C. (1998) Friction coefficients of ions in aqueous solution at 25°C. The Journal of the American Chemical Society, 120, 12041–12050.
Marry, V. and Turq, P. (2003) Microscopic simulations of interlayer structure and dynamics in bihydrated heteroionic montmorillonites. The Journal of Physical Chemistry B, 107, 1832–1839.
Marry, V., Dubois, E., Malikova, N., Breu, J., and Haussler, W. (2013) Anisotropy of water dynamics in clays: insights from molecular simulations for experimental QENS analysis. The Journal of Physical Chemistry C, 117, 15106–15115
Michaud-Agrawal, N., Denning, E.J., Woolf, T.B., and Beckstein, O. (2011) MDAnalysis: a toolkit for the analysis of molecular dynamics simulations. Journal of Computational Chemistry, 32, 2319–2327.
Nakano, M., Kawamura, K., and Ichikawa, Y. (2003) Local structural information of Cs in smectite hydrates by means of an EXAFS study and molecular dynamics simulations. Applied Clay Science, 23, 15–23.
Ngouana W., B.F. and Kalinichev, A.G. (2014) Structural arrangements of isomorphic substitutions in smectites: Molecular simulation of the swelling properties, interlayer structure, and dynamics of hydrated Cs-montmorillonite revisited with new clay models. The Journal of Physical Chemistry C, 118, 12758–12773.
Papelis, C. and Hayes, K.F. (1996) Distinguishing between interlayer and external sorption sites of clay minerals using X-ray absorption spectroscopy. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 107, 89–96.
Park, S.H. and Sposito, G. (2002) Structure of water adsorbed on a mica surface. Physical Review Letters, 89, 085501.
Pronk, S., Páll, S., Schulz, R., Larsson, P., Bjelkmar, P., Apostolov, R., Shirts, M.R., Smith, J.C., Kasson, P.M., van der Spoel, D., and Hess, B. (2013) GROMACS 4.5: a high-throughput and highly parallel open source molecular simulation toolkit. Bioinformatics, 29, 845–854.
Rotenberg, B., Marry, V., Vuilleumier, R., Malikova, N., Simon, C., and Turq, P. (2007) Water and ions in clays: Unraveling the interlayer/micropore exchange using molecular dynamics. Geochimica et Cosmochimica Acta, 71, 5089–5101.
Rotenberg, B., Morel, J.-P., Marry, V., Turq, P., and Morel-Desrosiers, N. (2009) On the driving force of cation exchange in clays: Insights from combined microcalorimetry experiments and molecular simulation. Geochimica et Cosmochimica Acta, 73, 4034–4044.
Rotenberg, B., Marry, V., Malikova, N., and Turq, P. (2010) Molecular simulation of aqueous solutions at clay surfaces. Journal of Physics: Condensed Matter, 22, 284114.
Shroll, R.M. and Smith, D.E. (1999) Molecular dynamics simulations in the grand canonical ensemble: Application to clay mineral swelling. The Journal of Chemical Physics, 111, 9025–9033.
Smith, D.E. and Dang, L.X. (1994a) Computer simulations of cesium-water clusters: Do ion-water clusters form gas-phase clathrates? The Journal of Chemical Physics, 101, 7873.
Smith, D.E. and Dang, L.X. (1994b) Computer simulations of NaCl association in polarizable water. The Journal of Chemical Physics, 100, 3757.
Strawn, D.G. and Sparks, D.L. (1999) The use of XAFS to distinguish between inner-and outer-sphere lead adsorption complexes on montmorillonite. Journal of Colloid and Interface Science, 216, 257–269.
Suter, J.L., Anderson, R.L., Greenwell, H.C., and Coveney, P.V. (2009) Recent advances in large-scale atomistic and coarse-grained molecular dynamics simulation of clay minerals. Journal of Materials Chemistry, 19, 2482–2493.
Swenson, J., Bergman, R., and Howells, W.S. (2000) Quasielastic neutron scattering of two-dimensional water in a vermiculite clay. The Journal of Chemical Physics, 113, 2873–2879.
Teppen, B.J. and Miller, D.M. (2006) Hydration energy determines isovalent cation exchange selectivity by clay minerals. Soil Science Society of America Journal, 70, 31–40.
Tesson, S., Salanne, M., Rotenberg, B., Tazi, S., and Marry, V. (2016) Classical polarizable force field for clays: Pyrophyllite and talc. The Journal of Physical Chemistry C, 120, 3749–3758.
Tribello, G.A., Bonomi, M., Branduardi, D., Camilloni, C., and Bussi, G. (2014) PLUMED 2: New feathers for an old bird. Computer Physics Communications, 185, 604–613.
Underwood, T., Erastova, V., Cubillas, P., and Greenwell, H.C. (2015) Molecular dynamic simulations of montmorillonite—organic interactions under varying salinity: An insight into enhanced oil recovery. The Journal of Physical Chemistry C, 119, 7282–7294.
Viani, A., Gualtieri, A.F., and Artioli, G. (2002) The nature of disorder in montmorillonite by simulation of X-ray powder patterns. American Mineralogist, 87, 966–975.
William Humphrey, A.D. (1996) VMD: visual molecular dynamics. Journal of Molecular Graphics, 14, 33–38.
Zhang, P.C., Brady, P.V., Arthur, S.E., Zhou, W.Q., Sawyer, D., and Hesterberg, D.A. (2001) Adsorption of barium (II) on montmorillonite: an EXAFS study. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 190, 239–249.
Zhang, Y. and Cremer, P.S. (2006) Interactions between macromolecules and ions: the Hofmeister series. Current Opinion in Chemical Biology, 10, 658–663.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Underwood, T., Erastova, V. & Greenwell, H.C. Ion Adsorption at Clay-Mineral Surfaces: The Hofmeister Series for Hydrated Smectite Minerals. Clays Clay Miner. 64, 472–487 (2016). https://doi.org/10.1346/CCMN.2016.0640310
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1346/CCMN.2016.0640310