Abstract
Background
This study investigated patient outcomes after urinary diversion in order to manage malignant ureteral obstruction caused by non-urologic cancers and to evaluate predictive factors for overall survival.
Methods
The study retrospectively reviewed patients with non-urologic malignancies who underwent ureteral stenting or percutaneous nephrostomy for ureteral obstruction between 2006 and 2014. The variables for predicting overall survival were identified by Cox regression analysis.
Results
The study enrolled 778 patients, including 522 patients who underwent ureteral stenting and 256 patients who underwent percutaneous nephrostomy. Renal function was assessed immediately and then 2 weeks after urinary diversion. The median survival period was 5 months (interquartile range [IQR] 2–12 months). A total of 708 patients died. The patients who received chemotherapy after urinary diversion had a survival gain of 7 months compared with the patients who did not receive subsequent chemotherapy (p < 0.001). The survival rate did not differ between the various types of urinary diversion (p = 0.451). In the multivariate analysis, lower survival rates were significantly associated with male sex; previous chemotherapy without radiotherapy; an increasing number of events related to malignant dissemination; low preoperative hemoglobin (< 10 mg/dL), albumin (< 3 g/dL), and estimated glomerular filtration (< 60 mL/min/1.73 m2) rates; and no subsequent chemotherapy or radiotherapy.
Conclusions
In cases of ureteral obstruction caused by non-urologic malignancies, the overall survival was poor. However, the patients who received chemotherapy after urinary diversion had a survival gain of 7 months. Therefore, urinary diversion could be considered to preserve renal function for subsequent chemotherapy, whereas patients with the poor prognostic factors should be presented with the option of no intervention.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Malignant ureteral obstruction (MUO) usually is associated with advanced cancer. The MUO may result from extrinsic compression by a primary lesion, retroperitoneal and pelvic lymphadenopathy, or direct tumor invasion. If the obstruction is not decompressed by percutaneous nephrostomy (PCN) or ureteral stent, obstructive uropathy can result in renal insufficiency, uremia, urosepsis, or even death. However, it is difficult for urologists to investigate the effectiveness of different urinary diversion methods because of the short median survival time (median, 6.4 months; range 21 h to 140 months) of patients with MUO and the high stent failure rate (range 37–51%). Moreover, due to the palliative nature of these procedures, it is necessary to consider the patient’s quality of life (QoL) or performance status after urinary diversion.1,2,3,–4
In current clinical practice, urinary diversion is performed for palliative care and to facilitate chemotherapy because advanced cancer currently is treated with a multi-disciplinary approach.5 Generally, PCN is used as a primary procedure due to the high failure rate of ureteral stents.
With modern advances in endourologic devices and materials, the ureteral stent has become the gold standard for MUO treatment.6 However, evidence that urinary diversion outcomes have significantly improved as a result of these technical advances is sparse.7 Moreover, the risks and benefits of urinary diversion must be considered on an individual basis.1 Regardless of recommendations in cancer-specific guidelines, the National Institute for Health and Care Excellence guidelines concluded that patients with obstructive uropathy secondary to prostate cancer should be offered urinary diversion by PCN or ureteral stenting. However, the option of no intervention as a treatment choice also should be discussed.8
Therefore, there is a need to clarify which method is the optimal option for patients with MUO in terms of life expectancy and treatment for primary cancer. The current study aimed to evaluate patient survival rates after urinary diversion for MUO and to identify predictive factors for overall survival (OS) in a single-center cohort of patients with non-urologic malignancy.
Patients and Methods
This retrospective study was approved by the institutional review board (IRB) of Yonsei University Severance Hospital (IRB No. 4-2020-0521). Patients were identified from our institution database under the procedures codes for PCN or ureteral stenting performed from January 2006 to December 2014. All patients provided written informed consent for participation in the study.
The study enrolled patients older than 18 years with a diagnosis of inoperable non-urological malignancies based on radiologic and pathologic findings. Ureteral obstruction was confirmed with computed tomography, magnetic resonance imaging, or ultrasound. Patients with MUO who did not undergo PCN or ureteral stenting were not enrolled. Furthermore, patients who had previously undergone urologic surgery, had bladder metastases, had undergone urinary diversion for a benign condition such as urinary stones or urinary fistula, or had incomplete clinical or follow-up data were excluded from the study.
The study collected data regarding age, sex, type of malignancy, previous treatment (surgery, chemotherapy, and radiotherapy to the retroperitoneal or pelvic area), events related to malignant dissemination (ascites, pleural effusion, metastasis), comorbidities (hypertension, diabetes mellitus, cardiovascular disease, cerebrovascular disease), diversion type (ureteral stenting or PCN), preoperative serum blood urea nitrogen, creatinine, estimated glomerular filtration rate (eGFR), hemoglobin, albumin, and subsequent therapy after urinary diversion.
Before the insertion of ureteral stents, all the patients received analgesics (pethidine HCl 50 mg) without sedation, and the male patients received 10 mL of 2% lidocaine gel, which was retained in the urethra. The stent was passed over a guidewire under both cystoscopic and fluoroscopic guidance. The wire was subsequently removed, and the coiling of both ends of the stent was confirmed through fluoroscopy. When retrograde stent insertion failed, PCN was performed with the patient under local anesthesia. A 22-G needle was used to enter the renal calyces, and correct placement was confirmed radiologically by using a dye. An 8-Fr PCN catheter was inserted after the tract had been gradually dilated. Proper placement of the stent or PCN catheter was confirmed with an abdominal x-ray image. The stent or PCN catheter was changed every 3 months. The survival time was defined as the interval from the initial diversion procedure to death.
Survival was estimated using the Kaplan–Meier method. Cox regression analysis was performed to identify the predictive factors for OS. The Wilcoxon test was used to compare pre- and postoperative eGFR levels. The level of significance was set at 0.05 in all analyses. All statistical analyses were performed using SPSS version 25.0 (IBM Corp., Armonk, NY, USA).
Results
The baseline patient characteristics are summarized in Table 1. Of the 778 patients who underwent urinary diversion, 522 (67.1%) underwent ureteral stenting and 256 (32.9%) underwent PCN. The median patient age was 57 years (interquartile range [IQR] 47–65 years). The most common type of malignancy was upper gastrointestinal cancer, which affected 316 patients (40.6%). Of these 316 patients, 310 had stomach cancer. Before urinary diversion, 501 patients (64.4%) had undergone previous surgery, 595 (76.5%) had received chemotherapy, and 170 (21.9%) had been treated with radiotherapy for the primary malignancy.
Events related to malignant dissemination were found for 715 patients (91.9%), and 253 patients (32.5%) had comorbidities. The median values of preoperative eGFR, hemoglobin, and albumin were respectively 59 mL/min/1.73 m2 (IQR 32–79 mL/min/1.73 m2), 10.5 mg/dL (IQR 9.5–11.6 mg/dL), and g/dL 3.5 (IQR 3.0–4.0 g/dL). Chemotherapy was performed for 447 patients (57.5%), and radiotherapy was performed after urinary diversion for 107 patients (13.8%).
The median values of eGFR immediately and then 2 weeks after urinary diversion were respectively 64 mL/min/1.73 m2 (IQR 35–89 mL/min/1.73 m2) and 69 mL/min/1.73 m2 (IQR 52–93 mL/min/1.73 m2) (Fig. 1). Compared with the preoperative eGFR, renal function significantly improved during a 2-week period after urinary diversion (all p < 0.001).
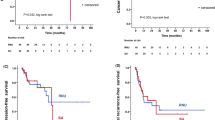
Of the 778 patients, 708 (91%) died, with a median OS of 5 months (IQR 2–12 months) after urinary diversion. The Kaplan–Meier curve for OS is demonstrated in Fig. 2. The survival rates were 75.8% at 1 month, 58.8% at 3 months, 43.7% at 6 months, and 25% at 12 months. Figure 3 demonstrates the survival curves according to chemotherapy after urinary diversion. The patients who did not receive subsequent chemotherapy had a median survival of 2 months (IQR 1–5 months). The patients who underwent chemotherapy after urinary diversion had a significant survival gain, with a median survival of 9 months (IQR 4–16 months) (p < 0.001).
The univariate analysis identified several factors associated with short survival times, including male sex (hazard ratio [HR] 1.273; p = 0.002); previous chemotherapy (HR 1.434; p < 0.001), no previous radiotherapy (HR 0.704; p < 0.001), an increasing number of events related to malignant dissemination (HR 1.343; p < 0.001), low preoperative eGFR (< 60 ml/min/1.73 m2; HR 1.360; p < 0.001), hemoglobin level lower than 10 mg/dL (HR 1.529; p < 0.001), albumin level lower than 3 g/dL (HR 2.137; p < 0.001), and no subsequent chemotherapy (HR 0.435; p < 0.001) or radiotherapy (HR 0.575; p < 0.001). These associations persisted in multivariate models after adjustment for variables (Table 2). The type of urinary diversion was not found to be associated with survival.
Discussion
The most effective method of MUO management remains unclear, leaving urologists to face a practical dilemma when dealing with patients who have MUO. The decision to treat with urinary diversion must consider the patient’s prognosis and likely QoL after urinary diversion. The preferences of the patient should be considered at all times throughout the decision-making process.
Several studies have investigated whether urinary diversion is beneficial for patients with advanced cancer. Furthermore, previous studies have examined whether the method of urinary diversion influences prognosis. Cordeiro et al.5 developed a risk model to estimate the survival of patients undergoing urinary diversion for MUO. Their study cohort included 58 patients who underwent ureteral stenting and 150 patients who underwent PCN. No differences in survival rates according to the type of urinary diversion were identified. The authors identified that an Eastern Cooperative Oncology Group (ECOG) index of 2 or higher and four or more events related to malignant dissemination were predictors of a poor prognosis after palliative urinary diversion, with a survival rate lower than 10% at 1 year postoperatively.
In deciding whether a patient will undergo urinary diversion and the type of diversion, QoL and the risk of postoperative complications should be considered. However, QoL assessment is difficult and can be unclear because the QoL and functional status of the patient may already be very poor because of the palliative setting.9 Some studies have reported that QoL is impaired after urinary diversion regardless of diversion type because of voiding symptoms, pain, and poor performance status.5,10
The impact of urinary diversion procedures on QoL remains controversial. Shekarriz et al.3 evaluated the performance status of 92 patients with MUO after urinary diversion to assess QOL indirectly and found that only 13 patients (14%) were free of pain with normal performance status after diversion and discovered no factors that predicted improved survival or performance status after the procedure. Conversely, Kanou et al.11 indirectly evaluated QoL by estimating the time that terminal patients could spend at home. Of 75 patients, 52 (69.3%) reported that urinary diversion positively influenced QoL. No differences between the diversion types were identified. Moreover, complications in patients who underwent ureteral stenting or PCN, despite the careful selection of diversion type, occurred at a rate of 41%.1
Studies have suggested that urinary diversion for MUO prevents progressive renal failure. However, renal function, which is related to the prognosis, was not restored, and the patients had poor survival even after urinary diversion.12,13 Ishioka et al.13 evaluated the outcomes of 140 patients who underwent PCN and found that the median serum creatinine levels of these patients improved from 4.33 mg/dL (range 0.54–18.57 mg/dL) to 1.1 mg/dL (range 0.4–5.5 mg/dL). However, 96% of the patients died, and the 1-year survival rate was only 12%. Yoon et al. 12 found that eGFR was significantly elevated during the 3-month period after ureteral stenting in 117 patients with a mean preoperative eGFR of 53.4 mL/min/1.73 m2. The eGFR levels remained between 53.4 and 57.8 mL/min/1.73 m2 for the following year. The 5-year OS was 22.9%, and preoperative eGFR was found to be a significant predictor of OS. Therefore, patients who require urinary diversion should undergo the procedure before renal function deteriorates to maintain renal function.
This is the largest study on urinary diversion in patients with MUO. For the 778 patients, the median OS was only 5 months (IQR 2–12 months) after urinary diversion, and the survival rate at 1 year was 25%. Chemotherapy after urinary diversion was performed for 57.5% of the patients, who had a significant survival gain of 7 months compared with the patients who had no subsequent chemotherapy. This finding is consistent with those of other studies.6,14,15
In performing chemotherapy, renal function should be preserved. In other studies, approximately 2 weeks were required for renal function to reach a nadir after urinary diversion.16,17,–18 Therefore, in this study, renal function was assessed immediately and then 2 weeks after urinary diversion. Renal function was found to improve during the 2 weeks, with a significant difference from the initial function. This meant that chemotherapy could be performed without delay.
Nevertheless, uncertainty exists regarding whether urinary diversion may prolong suffering for patients with incurable malignancy. Thus, the predictors for OS were estimated to identify patients who may not benefit from urinary diversion. This study found that poor survival was associated with male sex; previous chemotherapy without radiotherapy; increasing number of events related to malignant dissemination; low preoperative eGFR (< 60 ml/min/1.73 m2), hemoglobin (< 10 mg/dL), and albumin (< 3 g/dL) levels; and no subsequent chemotherapy. When MUO occurred in a patient who had previously undergone chemotherapy, it could be concluded that the disease had aggressively progressed despite chemotherapy, and the outcome was expected to be poor. Moreover, patients who did not receive subsequent chemotherapy had a median survival time of only 2 months. This meant that many patients died before the ureteral stent or PCN was changed for the first time. Patients with these predictors were expected to have a short life expectancy. Thus, in these cases, the option of no intervention should be discussed with the patients, their family, and clinicians.
This study has several limitations. First, all the data originated from a single institution and were retrospectively reviewed. Therefore, the results of this study may not be generalizable. Second, data concerning the functional status of patients were not presented because a large quantity of these data was not available. Third, information regarding complications was not included. However, complications associated with the diversion procedures were found to be associated with the failure of urinary diversion rather than survival, except in cases of rare severe complications, such as sepsis.2,19
In addition, because this study included both ureteral stenting and PCN, efforts were made to reduce confounding. The rates of successful ureteral stent placement and successful maintenance were significantly decreased in cases of bladder cancer, prostate cancer, and invasion of primary cancer into the bladder on cystoscopy.3,11,20,21 Thus, patients with urologic cancer, including bladder cancer and prostate cancer as well as bladder invasion of non-urologic cancer, were excluded from this study to avoid confusion in interpretation of the results. Furthermore, although the type of malignancy was a significant factor associated with OS in the univariate analysis, upper gastrointestinal cancer was associated with a short survival time in this study. However, the relationship between the type of malignancy and survival remains controversial. Several studies have found that non-gynecologic or gastrointestinal cancers were associated with short OS.10,15,22 However, despite the degree of malignancy, the level of ureteral obstruction could be diverse. Therefore, it is considered as a possible predictor of OS.6,13 As a result, the type of malignancy was excluded from the multivariate regression analysis because of its heterogeneity (27 types). Nonetheless, our results can be useful for determining whether urinary diversion is beneficial for patients with MUO.
Conclusion
In cases of ureteral obstruction with non-urologic malignancies, OS was poor despite urinary diversion. However, the patients who underwent chemotherapy after diversion had a survival gain of 7 months compared with the patients who did not receive subsequent chemotherapy. Therefore, in these patients, urinary diversion could be considered for preservation of renal function to facilitate subsequent chemotherapy. Patients who exhibit poor prognostic factors such as male sex; previous chemotherapy without radiotherapy; the presence of events related to malignant dissemination; low preoperative eGFR, hemoglobin, and albumin levels; and no subsequent treatment are expected to have a short survival time. Thus, these patients should be informed of their poor prognosis regardless of urinary diversion, and the option of no intervention should be discussed as a treatment choice.
References
Prentice J, Amer T, Tasleem A, Aboumarzouk O. Malignant ureteric obstruction decompression: how much gain for how much pain? A narrative review. J R Soc Med. 2018;111:125–35.
Kouba E, Wallen EM, Pruthi RS. Management of ureteral obstruction due to advanced malignancy: optimizing therapeutic and palliative outcomes. J Urol. 2008;180:444–50.
Shekarriz B, Shekarriz H, Upadhyay J, et al. Outcome of palliative urinary diversion in the treatment of advanced malignancies. Cancer. 1999;85:998–1003.
Chung SY, Stein RJ, Landsittel D, et al. 15-Year experience with the management of extrinsic ureteral obstruction with indwelling ureteral stents. J Urol. 2004;172:592–5.
Cordeiro MD, Coelho RF, Chade DC, et al. A prognostic model for survival after palliative urinary diversion for malignant ureteric obstruction: a prospective study of 208 patients. BJU Int. 2016;117:266–71.
Jeong IG, Han KS, Joung JY, Seo HK, Chung J. The outcome with ureteric stents for managing non-urological malignant ureteric obstruction. BJU Int. 2007;100:1288–91.
Wong LM, Cleeve LK, Milner AD, Pitman AG. Malignant ureteral obstruction: outcomes after intervention: have things changed? J Urol. 2007;178:178–83; discussion 183.
NICE Guidance–Prostate cancer: diagnosis and management: (c) NICE (2019) prostate cancer: diagnosis and management. BJU Int. 2019;124:9–26.
Sountoulides P, Pardalidis N, Sofikitis N. Endourologic management of malignant ureteral obstruction: indications, results, and quality-of-life issues. J Endourol. 2010;24:129–42.
Aravantinos E, Anagnostou T, Karatzas AD, Papakonstantinou W, Samarinas M, Melekos MD. Percutaneous nephrostomy in patients with tumors of advanced stage: treatment dilemmas and impact on clinical course and quality of life. J Endourol. 2007;21:1297–302.
Kanou T, Fujiyama C, Nishimura K, Tokuda Y, Uozumi J, Masaki Z. Management of extrinsic malignant ureteral obstruction with urinary diversion. Int J Urol. 2007;14:689–92.
Yoon JH, Park S, Park S, Moon KH, Cheon SH, Kwon T. Renal function is associated with prognosis in stent-change therapy for malignant ureteral obstruction. Investig Clin Urol. 2018;59:376–82.
Ishioka J, Kageyama Y, Inoue M, Higashi Y, Kihara K. Prognostic model for predicting survival after palliative urinary diversion for ureteral obstruction: analysis of 140 cases. J Urol. 2008;180:618–21; discussion 621.
Migita K, Watanabe A, Samma S, Ohyama T, Ishikawa H, Kagebayashi Y. Clinical outcome and management of ureteral obstruction secondary to gastric cancer. World J Surg. 2011;35:1035–41.
Izumi K, Mizokami A, Maeda Y, Koh E, Namiki M. Current outcome of patients with ureteral stents for the management of malignant ureteral obstruction. J Urol. 2011;185:556–61.
Nariculam J, Murphy DG, Jenner C, et al. Nephrostomy insertion for patients with bilateral ureteric obstruction caused by prostate cancer. Br J Radiol. 2009;82:571–6.
Jalbani MH, Deenari RA, Dholia KR, Oad AK, Arbani IA. Role of percutaneous nephrostomy (PCN) in malignant ureteral obstruction. J Pak Med Assoc. 2010;60:280–3.
Wilson JR, Urwin GH, Stower MJ. The role of percutaneous nephrostomy in malignant ureteric obstruction. Ann R Coll Surg Engl. 2005;87:21–4.
Allen DJ, Longhorn SE, Philp T, Smith RD, Choong S. Percutaneous urinary drainage and ureteric stenting in malignant disease. Clin Oncol R Coll Radiol. 2010;22:733–9.
Feng MI, Bellman GC, Shapiro CE. Management of ureteral obstruction secondary to pelvic malignancies. J Endourol. 1999;13:521–4.
Ganatra AM, Loughlin KR. The management of malignant ureteral obstruction treated with ureteral stents. J Urol. 2005;174:2125–8.
Alawneh A, Tuqan W, Innabi A, et al. Clinical factors associated with a short survival time after percutaneous nephrostomy for ureteric obstruction in cancer patients: an updated model. J Pain Symptom Manage. 2016;51:255–61.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
There are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Heo, J.E., Jeon, D.Y., Lee, J. et al. Clinical Outcomes After Urinary Diversion for Malignant Ureteral Obstruction Secondary to Non-urologic Cancer: An Analysis of 778 Cases. Ann Surg Oncol 28, 2367–2373 (2021). https://doi.org/10.1245/s10434-020-09423-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-020-09423-4