Abstract
Background
The prevalence and characteristics of actual 5-year survivors after surgical treatment of hilar cholangiocarcinoma (HC) have not been described previously.
Methods
Patients who underwent resection for HC from 2000 to 2015 were analyzed through a multi-institutional registry from 10 U.S. academic medical centers. The clinicopathologic characteristics and both the perioperative and long-term outcomes for actual 5-year survivors were compared with those for non-survivors (patients who died within 5 years after surgery). Patients alive at last encounter who had a follow-up period shorter than 5 years were excluded from the study.
Results
The study identified 257 patients with HC who underwent curative-intent resection with an actuarial 5-year survival of 19%. Of 194 patients with a follow-up period longer than 5 years, 23 (12%) were 5-year survivors. Compared with non-survivors, the 5-year survivors had a lower median pretreatment CA 19-9 level (116 vs. 34 U/L; P = 0.008) and a lower rate of lymph node involvement (42% vs. 15%; P = 0.027) and R1 margins (39% vs. 17%; P = 0.042). However, the sole presence of these factors did not preclude a 5-year survival after surgery. The frequencies of bile duct resection alone, major hepatectomy, caudate lobe resection, portal vein or hepatic artery resection, preoperative biliary sepsis, intraoperative blood transfusion, serious postoperative complications, and receipt of adjuvant chemotherapy were comparable between the two groups.
Conclusions
One in eight patients with HC reaches the 5-year survival milestone after resection. A 5-year survival can be achieved even in the presence of traditionally unfavorable clinicopathologic factors (elevated CA 19-9, nodal metastasis, and R1 margins).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Hilar cholangiocarcinoma is a challenging malignancy arising from neoplastic transformation of the ductal epithelium at the biliary confluence. In one of the earliest reports on hilar cholangiocarcinoma, Gerald Klatskin1 observed the difficulty of diagnosing these tumors and concluded that surgery was the most effective treatment method to relieve biliary obstruction and prolong life.
Unresectability rates remain high, ranging from 24 to 66%, and can be attributed to tumor involving bilateral second-order biliary radicals, main or bilateral portal vein or hepatic artery invasion, presence of hepatic lobar atrophy with contralateral second-order biliary radical or major vascular involvement, and distant metastases.2,3,4 Due to the propensity of these tumors to extend both radially and longitudinally, surgical resection can be challenging. However, in the last 20 years, adoption of a carefully planned and aggressive surgical strategy has been reported to improve resectability and survival outcomes.
Actuarial 5-year survival after resection of hilar cholangiocarcinoma ranges widely from 14 to 48%.5,6,7,8,9,10,11 Variability in these outcomes is due to differences in follow-up evaluation, inclusion of palliative resections, and combined analysis with other types of bile duct cancers. Furthermore, early experience with resection of hilar cholangiocarcinoma was complicated by high perioperative mortality rates associated with increased rates of liver failure.12,13,14,15 Moreover, all the studies reported estimated (actuarial) 5-year survival probabilities.
Actual 5-year survival has not been described previously, and factors associated with this long-term survival are not well known. Hence, in this report we describe a contemporary, multi-center experience of patients who underwent curative-intent resection for hilar cholangiocarcinoma, with the goal of identifying clinical and pathologic factors associated with survival longer than 5 years after surgery.
Methods
Study Population
The study population included patients who underwent curative-intent surgical resection for hilar cholangiocarcinoma between 1 January 2000 and 14 December 2014 at 1 of 10 academic institutions participating in the U.S. Extrahepatic Biliary Malignancy Consortium (John Hopkins Hospital, Baltimore, MD; Emory University, Atlanta, GA; Stanford University, Palo Alto, CA; University of Wisconsin, Milwaukee, WI; The Ohio State University, Columbus, OH; Washington University, St. Louis, MO; Vanderbilt University, Nashville, TN; New York University, New York, NY; University of Louisville, Louisville, KY; Wake Forest University, Winston-Salem, NC).
Patients were retrospectively identified and stratified into the following two groups based on survival: actual 5-year survivors and non-survivors (who died within 5 years after resection). Patients with less than 5 years of follow-up evaluation but alive at the last encounter were excluded from the study. Only patients who underwent curative-intent surgical resection (R0 or R1) were included in the study. Institutional Review Board approval was obtained from each of the participating institutions.
Data Collection
Standard demographics and both pathologic and treatment characteristics were retrospectively reviewed. The demographic data included age, sex, race, and comorbidity. The treatment data collected included type of preoperative biliary drainage procedures, common bile duct resection, type of surgery, intraoperative blood transfusion, and estimated blood loss (EBL). The pathology data included American Joint Commission on Cancer (AJCC) T stage, nodal status, histology, margins, tumor grade, and presence of lymphovascular or perineural invasion. The seventh edition of the AJCC staging manual was used to determine stage.16 Postoperative morbidity was graded using the modified Clavien-Dindo classification of surgical complications.17
Statistical Analysis
Continuous variables were reported as median values with interquartile ranges and compared using the Mann–Whitney U test or Student’s t test where applicable. Categorical variables were expressed as frequency with percentages and compared using Pearson Chi square or Fisher’s exact test where appropriate. The actuarial overall survival (OS) was calculated using the Kaplan–Meier method and compared with the log-rank test. Differences were considered statistically significant if the P value was lower than 0.05. All statistical analyses were performed using STATA version 13.0 (College Station, TX, USA) and SPSS version 22.0 (IBM Inc, Chicago, IL, USA).
Results
Study Population and Clinical Demographics
Of the 328 patients with hilar cholangiocarcinoma in the U.S. Extrahepatic Biliary Malignancy Consortium database, 43 underwent surgical exploration (laparoscopic or open) and were found to have unresectable locally advanced or distant disease. The study excluded 30 patients due to palliative R2 resections. Of the remaining 257 patients who underwent curative-intent resection, 194 had more than 5 years of follow-up evaluation, with 23 patients (12%) classified as 5-year survivors and 171 patients (88%) classified as non-survivors. Table 1 compares the preoperative baseline patient characteristics between the 5-year survivors and non-survivors.
The cohort predominantly consisted of males (59%), and the median age was 67 years. The patient demographics and comorbidities were comparable between the two groups. The incidence of clinical jaundice (87% vs. 83.4%; P = 0.66) and median peak serum bilirubin levels (5.5 vs. 4.9 mg/dL; P = 0.51) were similar between the 5-year survivors and non-survivors. The median preoperative serum CA19-9 level was significantly lower among the 5-year survivors than among the non-survivors (34.5 vs. 116 U/L; P = 0.0008). The type of preoperative biliary drainage procedure performed (i.e., endoscopic or percutaneous) did not differ significantly between the two groups. No statistically significant difference in Blumgart T stage was found between 5-year survivors and non-survivors. None of the 5-year survivors had preoperative systemic sepsis (i.e., ascending cholangitis) or underwent portal vein embolization before surgery. However, the prevalence of these two variables did not differ between the two groups.
Operative Data
The data on operative management are shown in Table 2. The type of resection (bile duct resection alone, hemihepatectomy, trisectionectomy, or concomitant caudate lobe resection) was comparable between the two groups. Encountering a positive margin on frozen section intraoperatively did not prevent patients from reaching the 5-year survival milestone. However, none of the 5-year survivors in this series underwent portal vein or hepatic artery resection.
Pathology
The pathologic characteristics of the actual 5-year survivors and non-survivors are compared in Table 3. The 5-year survivors had more commonly undergone R0 resection (82.6% vs. 60.8%; P = 0.042). However, 17% (n = 4) of the 5-year survivors had microscopically positive (R1) resection margins. Early T stage and absence of lymph node involvement were more common among the 5-year survivors than among the non-survivors. None of the 5-year survivors had T3 or T4 disease. Tumor size and grade were similar between the two groups. Lymphovascular invasion was less common among the 5-year survivors, and this difference approached significance (22.2% vs. 45%; P = 0.066). Among the patients who had a particularly unfavorable outcome (death within 2 years, n = 127), 7 (5.5%) had poorly differentiated N1 tumors, and 22 (17.3%) had R1N1 tumors.
Postoperative Outcomes and Adjuvant Therapy
The 90-day mortality rate was 6% for the entire study population of 257 patients with hilar cholangiocarcinoma. Among the 194 patients with a 5-year follow-up period, the rates for any complication, major complication, surgical-site infection, intraabdominal abscess, postoperative bleeding, bile leak, anastomotic leak, postoperative drainage procedure, 90-day readmission, and reoperation were comparable between the two groups (Table 4). Only half of the 5-year survivors received adjuvant chemotherapy, which was comparable with the 63% of non-survivors who received adjuvant therapy. The use of adjuvant radiation were similar between the two groups.
Actuarial Survival Analysis
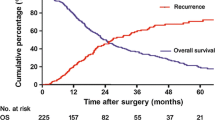
The actuarial 5-year OS was 19% (Fig. 1A). Of the 23 actual 5-year survivors, 10 were actual 7-year survivors and 4 were actual 10-year survivors. The 5-year recurrence-free survival rate was 16% (Fig. 1B). For the 257 patients who underwent curative-intent resection for hilar cholangiocarcinoma, older age, advanced T-stage, nodal involvement, poor differentiation, and elevated postoperative bilirubin were independent predictors of poor survival (Table S1).
Discussion
In this multi-institutional study of 194 patients with hilar cholangiocarcinoma who underwent curative-intent resection and had more than 5 years of follow-up evaluation, we identified 23 actual 5-year survivors (12%). It is interesting to note that the fraction of patients who actually survived 5 years (12%) differed from the actuarial 5-year survival rate of 19% noted for the entire cohort of 257 patients in the study (irrespective of follow-up length).
Kaplan–Meier survival analyses are helpful in estimating survival probabilities, but they tend to overestimate survival due to the effect of patients lost to follow-up evaluation, who are censored in the analysis. In contrast, data from the current study provide more pragmatic and tangible information on the long-term prognosis for these patients.
Not surprisingly, the actual 5-year survivors had a lower incidence of unfavorable prognostic factors (e.g., nodal metastasis and microscopically positive resection margins) than the non-survivors. However, the existence of these traditionally adverse prognostic factors did not categorically prevent patients from achieving a 5-year survival. In fact, among the 5-year survivors, a small fraction of patients was noted to have established poor prognostic factors, such as N1 disease (15%), poor differentiation (16%), R1 margins (17%), perineural invasion (70%), and lymphovascular invasion (22%). These findings underscore the notion that although such prognostic factors can be used to predict outcome, their presence cannot entirely rule out the possibility of long-term survival. That being said, among the aforementioned predictors of outcome, the only modifiable one is margin status, and surgeons should make every effort to achieve a margin-negative resection for this patient population.
Data on actual 5-year survivors after surgical resection of intrahepatic cholangiocarcinoma and pancreatic cancer are almost identical to those reported in this study for hilar cholangiocarcinoma. For example, in a multi-institutional study of 1154 patients with intrahepatic cholangiocarcinoma who underwent resection, Bagante et al.18 showed that the rate of actual 5-year survivors was 13%. Similarly, among these long-term survivors were patients with traditionally adverse clinicopathologic factors such as perineural invasion, intrahepatic metastasis, satellitosis, N1 status, tumors larger than 5 cm, and direct invasion into an adjacent organ.
Along the same lines, Ferrone et al.19 reported on a single-institution series of 618 patients with pancreatic adenocarcinoma, 12% of whom were actual 5-year survivors after resection. Among these long-term survivors, again were patients with advanced T and N stage disease as well as R1 margins.
Although studies have shown that concomitant portal vein and/or hepatic artery resection are associated with higher perioperative morbidity and mortality after resection of hilar cholangiocarcinoma, the reported long-term survival outcome of vascular resection varies widely in the literature.8,20,21,22,23 On the one hand, in a study consisting of 161 patients who underwent surgery for hilar cholangiocarcioma, Miyazaki et al.22 noted that patients who did not require vascular resection had better survival than those requiring portal vein and/or hepatic artery resection (5-year survival, 41% vs. 25% vs. 0%, respectively; P < 0.001).
On the other hand, Neuhaus et al.23 reported on a series of 50 patients with hilar cholangiocarcinoma, observing that right trisectionectomy with en bloc “no touch” portal vein resection and reconstruction was associated with better survival than conventional major hepatectomy without portal vein resection (5-year DSS, 58% vs. 29%; P = 0.021). However, most studies have shown that the needed portal vein resection is not associated with worse or improved survival. Specifically, a study from our group previously found that patients who underwent portal vein resection and patients without portal vein resection had a similar median OS (24 vs. 21 months; P = 0.818) and a similar median recurrence-free survival (17 vs. 16 months; P = 0.199).24
Although the current study showed that none of the 5-year survivors underwent portal vein or hepatic artery resection, the survivors and non-survivors did not differ significantly in the frequency of vascular resection. Perhaps with a more mature follow-up evaluation, we would have observed some 5-year survivors in the vascular reconstruction group. Concomitant vascular resections when necessary should be undertaken by highly experienced surgical teams to achieve negative margins in carefully selected patients with hilar cholangiocarcinoma.
Similarly, none of the actual 5-year survivors in this series was noted to have preoperative biliary sepsis. Although this finding can be attributed to the small sample size of the survivor cohort and the short follow-up period, it still underscores the potential adverse systemic effects of sepsis in cell immunity, which can consequently promote tumor growth.25
Our group has previously shown an association between infectious complications and tumor recurrence in extrahepatic cholangiocarcinoma.26 This association also has been demonstrated in hepatocellular carcinoma27 and intrahepatic cholangiocarcinoma.28 In addition, the presence of preoperative cholangitis has been recognized recently as a significant predictor of fatal postoperative liver failure after resection of hilar cholangiocarcinoma, especially when the future liver remnant is less than 30% of the standardized liver volume.29 This observation may provide an additional explanation for the rarity of long-term survivors after resection of hilar cholangiocarcinoma in the presence of preoperative cholangitis.
The current study had several major limitations. First, the retrospective nature of the study introduced selection biases, mainly due to the exclusion of patients who were not candidates for curative-intent resection. As such, it is debatable whether the data collected from these highly selected patients (who eventually underwent curative-intent surgery) actually reflect the true denominator for this disease. Second, variability and lack of standardization in operative and perioperative management certainly can exist among the 10 academic institutions participating in this study. Third, the total number of actual 5-year survivors still is small, and some other differences may not have been detected due to small sample size or the short follow-up period. Nevertheless, this large multi-institutional study provided generalizable results for a rare malignancy and identified factors that can be used to facilitate prognostication for patients with hilar cholangiocarcinoma for whom surgical resection is considered.
In conclusion, although hilar cholangiocarcinoma is a challenging malignancy, one in eight patients actually survive longer than 5 years after surgical resection. In this study, several clinicopathologic factors were associated with a lower likelihood of survivorship (e.g., elevated CA19-9 level, T3/4 stage, N1 disease, and R1 margins), yet long-term survival did occur among a small subset of patients who had these poor prognostic features. Therefore, the pre- or intraoperative discovery of involved regional nodes or the anticipation of a close-margin resection should not deter surgeons from pursuing curative-intent resections for this challenging malignancy. On the one hand, this striking lack of absolute correlation between stage, margins, and actual 5-year survival exemplifies the diverse biology of long-term cancer survivors. On the other hand, these data support the notion that surgical resection still is an inadequate treatment for the majority of patients with hilar cholangiocarcinoma, even for the favorable subset of those with resectable disease. An improved understanding concerning the molecular foundation of this disease is needed for identification of more effective systemic therapeutic approaches that could further enhance the outcome of surgical resection.
References
Klatskin G. Adenocarcinoma of the hepatic duct at its bifurcation within the porta hepatis: an unusual tumor with distinctive clinical and pathological features. Am J Med. 1965;38:241–56.
Burke EC, Jarnagin WR, Hochwald SN, Pisters PW, Fong Y, Blumgart LH. Hilar cholangiocarcinoma: patterns of spread, the importance of hepatic resection for curative operation, and a presurgical clinical staging system. Ann Surg. 1998;228:385–94.
Ito F, Agni R, Rettammel RJ, et al. Resection of hilar cholangiocarcinoma: concomitant liver resection decreases hepatic recurrence. Ann Surg. 2008;248:73–9.
Matsuo K, Rocha FG, Ito K, et al. The Blumgart preoperative staging system for hilar cholangiocarcinoma: analysis of resectability and outcomes in 380 patients. J Am Coll Surg. 2012;215:343–55.
Abd ElWahab M, El Nakeeb A, El Hanafy E, et al. Predictors of long-term survival after hepatic resection for hilar cholangiocarcinoma: a retrospective study of 5-year survivors. World J Gastrointest Surg. 2016;8:436–43.
Jang JY, Kim SW, Park DJ, et al. Actual long-term outcome of extrahepatic bile duct cancer after surgical resection. Ann Surg. 2005;241:77–84.
Konstadoulakis MM, Roayaie S, Gomatos IP, et al. Aggressive surgical resection for hilar cholangiocarcinoma: is it justified? Audit of a single center’s experience. Am J Surg. 2008;196:160–9.
Nagino M, Ebata T, Yokoyama Y, et al. Evolution of surgical treatment for perihilar cholangiocarcinoma: a single-center 34-year review of 574 consecutive resections. Ann Surg. 2013;258:129–40.
Silva MA, Tekin K, Aytekin F, Bramhall SR, Buckels JA, Mirza DF. Surgery for hilar cholangiocarcinoma; a 10 year experience of a tertiary referral centre in the UK. Eur J Surg Oncol. 2005;31:533–9.
Neuhaus P, Jonas S, Bechstein WO, et al. Extended resections for hilar cholangiocarcinoma. Ann Surg. 1999;230:808–18; discussion 819.
Nishio H, Nagino M, Nimura Y. Surgical management of hilar cholangiocarcinoma: the Nagoya experience. HPB Oxf. 2005;7:259–62.
Belghiti J, Ogata S. Preoperative optimization of the liver for resection in patients with hilar cholangiocarcinoma. HPB Oxf. 2005;7:252–3.
Cherqui D, Benoist S, Malassagne B, Humeres R, Rodriguez V, Fagniez PL. Major liver resection for carcinoma in jaundiced patients without preoperative biliary drainage. Arch Surg. 2000;135:302–8.
Lai EC, Chu KM, Lo CY, et al. Surgery for malignant obstructive jaundice: analysis of mortality. Surgery. 1992;112:891–6.
Soares KC, Kamel I, Cosgrove DP, Herman JM, Pawlik TM. Hilar cholangiocarcinoma: diagnosis, treatment options, and management. Hepatobiliary Surg Nutr. 2014;3:18–34.
Edge SB, American Joint Committee on Cancer, American Cancer Society. AJCC cancer staging manual. 7th ed. Springer, New York, London, 2010.
Clavien PA, Barkun J, de Oliveira ML, et al. The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg. 2009;250:187–96.
Bagante F, Spolverato G, Weiss M, et al. Defining long-term survivors following resection of intrahepatic cholangiocarcinoma. J Gastrointest Surg. 2017;21:1888–97.
Ferrone CR, Brennan MF, Gonen M, et al. Pancreatic adenocarcinoma: the actual 5-year survivors. J Gastrointest Surg. 2008;12:701–6.
Nagino M, Nimura Y, Nishio H, et al. Hepatectomy with simultaneous resection of the portal vein and hepatic artery for advanced perihilar cholangiocarcinoma: an audit of 50 consecutive cases. Ann Surg. 2010;252:115–23.
Noji T, Tsuchikawa T, Okamura K, et al. Concomitant hepatic artery resection for advanced perihilar cholangiocarcinoma: a case-control study with propensity score matching. J Hepatobiliary Pancreat Sci. 2016;23:442–8.
Miyazaki M, Kato A, Ito H, et al. Combined vascular resection in operative resection for hilar cholangiocarcinoma: Does it work or not? Surgery. 2007;141:581–8.
Neuhaus P, Thelen A, Jonas S, et al. Oncological superiority of hilar en bloc resection for the treatment of hilar cholangiocarcinoma. Ann Surg Oncol. 2012;19:1602–8.
Schimizzi GV, Jin LX, Davidson JTt, et al. Outcomes after vascular resection during curative-intent resection for hilar cholangiocarcinoma: a multi-institution study from the US extrahepatic biliary malignancy consortium. HPB Oxf. 2018;20:332–9.
Cavassani KA, Carson WFt, Moreira AP, et al. The post sepsis-induced expansion and enhanced function of regulatory T cells create an environment to potentiate tumor growth. Blood. 2010;115:4403–11.
Buettner S, Ethun CG, Poultsides G, et al. Surgical-site infection is associated with tumor recurrence in patients with extrahepatic biliary malignancies. J Gastrointest Surg. 2017;21:1813–20.
Margonis GA, Sasaki K, Andreatos N, et al. Prognostic impact of complications after resection of early stage hepatocellular carcinoma. J Surg Oncol. 2017;115:791–804.
Spolverato G, Yakoob MY, Kim Y, et al. Impact of complications on long-term survival after resection of intrahepatic cholangiocarcinoma. Cancer. 2015;121:2730–9.
Ribero D, Zimmitti G, Aloia TA, et al. Preoperative cholangitis and future liver remnant volume determine the risk of liver failure in patients undergoing resection for hilar cholangiocarcinoma. J Am Coll Surg. 2016;223:87–97.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
There are no conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tran, T.B., Ethun, C.G., Pawlik, T.M. et al. Actual 5-Year Survivors After Surgical Resection of Hilar Cholangiocarcinoma. Ann Surg Oncol 26, 611–618 (2019). https://doi.org/10.1245/s10434-018-7075-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-018-7075-4