Abstract
Background
c-Met relies on CD44v6 for its activation and signaling in several cancer cell lines. However, the correlation of c-Met and CD44v6 expression and its biological significance in esophageal squamous cell carcinoma (ESCC) remains unknown.
Methods
Expression of c-Met and CD44v6 was examined by immunohistochemistry (IHC) in 147 ESCC specimens. We analyzed the impact of c-Met and CD44v6 expression on clinicopathological parameters, including chemoresistance or prognosis in ESCC.
Results
High expression of c-Met and CD44v6 in cancerous lesions was identified in 49.7% and 50.3% of all patients, respectively. The c-Met-high group comprised more advanced pT and pM stages than the c-Met-low group. In addition, more patients in the c-Met-high group received neoadjuvant chemotherapy (NACT) than the c-Met-low group (64.4% vs. 43.2%, P = 0.010). On the other hand, the CD44v6-high group was associated with more advanced pT/pN stages and a poorer clinical response to NACT (response rate 53.5% vs. 77.8%, P = 0.025) than the CD44v6-low group. Double-positive immunostaining of c-Met and CD44v6 was identified in 28.6% of all cases, and multivariate analysis of overall survival (OS) identified them (hazard ratio 1.79, 95% confidence interval 1.03–3.04, P = 0.038) as independent prognostic factors in addition to pN and pM stage.
Conclusions
c-Met/CD44v6 were associated with tumor progression or chemoresistance. Double-positive expression of c-Met and CD44v6 negatively impacted patient prognosis in ESCC, implying that c-Met and CD44v6 are candidates for targeted therapy in ESCC.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Esophageal cancer has a poor prognosis and is the sixth most common cause of cancer-related death.1 Although multidisciplinary treatments for ESCC, including chemotherapy, chemoradiotherapy (CRT), and surgery, have been developed, their contributions to patient outcomes are still insufficient.2 Because nonresponders to chemotherapy or CRT for ESCC have a poor prognosis, elucidation of the resistance mechanism and the development of novel molecular-targeted drugs are urgently needed.3
c-Met is expressed in both normal and malignant cells as an epithelial/endothelial cell surface transmembrane receptor tyrosine kinase with specificity for hepatocyte growth factor (HGF), also known as scatter factor.4 Abnormal c-Met activation in cancer correlates with various biological reactions, including cell proliferation, dispersion, mobility, invasion, apoptosis, and angiogenesis.5,6 In various cancers, overexpression of c-Met has been reported to be associated with malignant features of cancer, cancer stem cells, and chemoresistance or radioresistance.7,8,9,10,11,12,13,14,15 Accordingly, novel treatments targeting c-Met have been already introduced for several types of cancer, including gastric cancer, non-small cell lung cancer, and hepatocellular carcinoma.16,17,18 However, the clinical use of c-Met inhibitor for ESCC has not yet been reported.
CD44 was identified as a stem-cell marker in various cancers. The extracellular domain of CD44v6 forms a ternary complex with c-Met and HGF, which is necessary for c-Met activation.6,19,20,21,22 CD44v6 also has been reported to be related to stem cell-ness in breast cancer and colorectal cancer.23,24 In pancreatic cancer, co-expression of c-Met and CD44v6 is associated with the potency of lymph node and lung metastasis; neither c-Met nor CD44v6 knockdown pancreatic cancer cells have metastatic potency in xenograft mice.22 The downstream c-Met/HGF axis depends on the presence of CD44v6, and these studies emphasize the importance of assessing CD44v6 in addition to c-Met.21,22
A few recent studies have shown that c-Met or CD44v6 expression is a significant prognostic factor in ESCC.25,26,27,28,29 However, the relationship between c-Met and CD44v6 expression in clinical samples of ESCC and their association with malignant potential or chemoresistance remain to be determined. The present study was designed to evaluate the expression of both c-Met and CD44v6 in resected ESCC specimens and clarify their biological significance, including chemoresistance or prognosis, in ESCC patients.
Patients and Methods
Patients
The present study included a total of 147 thoracic ESCC patients who underwent radical esophagectomy with standard lymphadenectomy between 2004 and 2012 at our hospital. We excluded patients who underwent preoperative chemoradiation therapy, were diagnosed as pathological complete response (grade 3) to preoperative chemotherapy, or had a history of other cancer. No patients underwent targeted therapy, including receptor tyrosine kinase inhibitors. The data for all patients were included in a prospectively collected database based primarily on the surgical and perioperative laboratory data. Pathological staging was based on the seventh edition of The Union for International Cancer Control (UICC) TNM Classification.30 The present study was approved by the Human Ethics Review Committee of Osaka University School of Medicine and signed consent was obtained from each participant.
Surgical Procedure and Treatment of Esophageal Cancer
Our standard surgical procedures were a subtotal esophagectomy with mediastinal lymphadenectomy via right thoracotomy, upper abdominal lymphadenectomy, reconstruction with a gastric tube via the posterior mediastinum, and anastomosis in the cervical incision.31,32,33,34 Preoperative chemotherapy consisted of cisplatin, 5-FU, and either Adriamycin (ACF) or docetaxel (DCF), followed by curative esophagectomy, as described previously.31,32,33,35 Our indication for preoperative chemotherapy based on the TNM classification was cT1-3N1-3 as an absolute indication and either cT2-3N0 or cT4Nany without massive infiltration to the bronchus or aorta as a relative indication.31 Patients who underwent preoperative chemotherapy had an Eastern Cooperative Oncology Group performance status of 0–1 and normal bone marrow, kidney, and liver function.
Evaluation of Clinical and Pathological Responses to Chemotherapy
The clinical response to neoadjuvant chemotherapy (NACT) was determined by computed tomography, fluorodeoxyglucose positron emission tomography, and upper gastrointestinal endoscopy.36 The surgical specimens were classified into five categories based on the degree of histopathological tumor regression. The proportion of viable residual tumor cells in the sample was assessed as follows: grade 3 (no viable residual tumor cells), grade 2 (less than one-third residual tumor cells), grade 1b (one-third to two-thirds residual tumor cells), grade 1a (more than two-thirds residual tumor cells), grade 0 (no significant response to preoperative therapy).32,35,37
Immunohistochemistry
Paraffin-embedded tumor tissue samples were cut into 4-µm sections and mounted on adhesive glass slides. Slides were deparaffinized in xylene and hydrated with alcohol and then were pretreated in a microwave oven to enhance antigen accessibility to the antibody. The primary antibodies included rabbit monoclonal anti-total c-Met (clone SP44, Spring Bioscience, Pleasanton, CA; 1: 400 dilution) and mouse monoclonal anti-CD44v6 (clone VFF-18, Abcam, Cambridge, UK; 1: 200 dilution). A negative control was prepared by omitting the primary antibody. Antibody binding was visualized using a Vectastain Elite ABC kit (Vector Laboratories, Burlingame, CA). The slides were incubated in 3,3’-diaminobenzidine tetrahydrochloride (DAB) with 0.05% hydrogen peroxide for 80 s (c-Met) and 40 s (CD44v6). Finally, the slides were counterstained with 0.1% hematoxylin for 30 s. Normal human placenta tissue was used as a positive control for c-Met, and normal human esophageal tissue from the same patients was used as a positive control for CD44v6.
Evaluation of Immunohistochemical Staining
Both c-Met and CD44v6 expression were evaluated in five randomly selected images of representative areas at a magnification of 100 with a high-power field. Staining intensities were evaluated according to the following classification: 0, no staining; 1+ , weaker than the basal layer; 2+, the same intensity as the basal layer; 3+ , stronger than the basal layer. Staining proportions were determined as the percentage of positive cells with each intensity. Finally, we calculated an H-score by summing the values obtained when multiplying the intensities and proportions at each intensity.25,38 The cases scoring higher than the cutoff value, according to the median of each H-score, were c-Met and CD44v6 positive, and the others were c-Met and CD44v6 negative. All specimens were examined independently by two observers (T.H. and T.M.) blinded to the patient’s clinical findings, and one pathologist (N.M.) confirmed their diagnosis.
Statistical Analysis
The relationship between c-Met and CD44v6 expression and the clinicopathological factors was analyzed using the Chi-squared test for categorical variables and Student’s t test for continuous variables. The relationship between c-Met and CD44v6 H-score was estimated using Pearson’s rank correlation coefficient. Overall survival (OS) was defined as the time elapsed from the date of surgery to the date of death from any cause. The survival rate was estimated using the Kaplan–Meier method, and survival was compared using the log-rank test for two comparisons and Bonferroni’s method for multiple comparisons. A multivariate Cox proportional hazard regression model was used to analyze the independent prognostic factors. P values < 0.05 were considered to indicate significance. Statistical analyses were performed using JMP Pro® 13 software (SAS Institute Inc., Cary, NC).
Results
c-Met and CD44v6 Expression Status in Normal Epithelium and Cancer
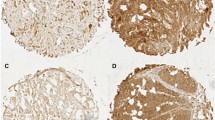
The representative immunohistochemistry (IHC) results for c-Met and CD44v6 in ESCC, normal esophagus, and positive control are shown in Fig. 1. For ESCC, c-Met staining was localized primarily in the cytoplasm and membrane, whereas CD44v6 staining was observed primarily in the membrane. Both c-Met and CD44v6 stained homogeneously in tumor areas in the majority of cases (the proportion of homogeneous c-Met and CD44v6 staining; 87.8% and 87.8%, respectively). The median H-score based on IHC results for c-Met was 90 (range 20–280), and the proportion of c-Met-high was 49.7% (73/147) when using the median H-score as a cutoff. Similarly, the median H-score for CD44v6 IHC was 110 (range 30–270) and the proportion of CD44v6-high 50.3% (74/147). The grading of immunostained sections was almost identical with the two observers, with interobserver variation < 10%.
Representative immunohistochemistry results. (a) c-Met staining in the normal esophagus and (b) normal placenta (positive control). (c) 0, no staining; (d) 1+ , weaker than the basal layer, (e) 2+ , staining with the same intensity as the basal layer, and (f) 3+ , stronger than the basal layer. (g) CD44v6 staining in the normal esophagus. (h) 0, no staining, (i) 1+ , weaker than the basal layer, (j) 2+ , staining with the same intensity as the basal layer, and (k) 3+ , stronger than the basal layer. Scale bar: 100 μm
Concordance of c-Met and CD44v6 Expression
The relationship between c-Met and CD44v6 expression in resected ESCC specimens is shown in Fig. 2. The rate of co-expression (double positive for c-Met and CD44v6) was identified in 28.6% (42/147) of all cases, whereas iso-expression (high expression of either c-Met or CD44v6) and double-negative (low expression of both c-Met and CD44v6) expression was observed in 42.8% (63/147) and 28.6% (42/147) of all cases (Fig. 2a). A weak but significant correlation was observed between c-Met and CD44v6 expression (r = 0.24, P = 0.0033; Fig. 2b).
Correlation Between c-Met/CD44v6 Expression and Clinicopathological Parameters
The relationships between clinicopathological parameters and expression status of c-Met and CD44v6 were analyzed as shown in Table 1. The c-Met-high group (n = 73) had a significantly higher proportion of advanced pT stage (54.8% vs. 31.1%, P = 0.0037) and pM stage than the c-Met-low group (n = 74; 11.0% vs. 2.7%, P = 0.047). Furthermore, significantly more patients in the c-Met-high group received preoperative chemotherapy compared with the c-Met-low group (64.4% vs. 43.2%, P = 0.010). However, the expression of c-Met was associated with neither a clinical nor pathological response to NACT.
On the other hand, protein accumulation of CD44v6 significantly correlated with advanced stage, including advanced pT stage (52.7% vs. 32.9%, P = 0.015) and pN stage (70.3% vs. 52.1%, P = 0.023). In addition, high expression of CD44v6 was significantly associated with a poor clinical response to NACT in 79 cases (responders 53.5% vs. 77.8%, P = 0.025). Neither chemotherapy regimens significantly correlated with the expression of two molecules (ACF (%): c-Met high vs. low 76.7% vs. 86.1%, P = 0.29; CD44v6 high vs. low 78.7% vs. 84.4%, P = 0.53).
Survival Analysis
The c-Met-high group had significantly worse OS than the c-Met-low group in all 147 cases (5-year OS 39.0% vs. 72.0%, P < 0.0001; Fig. 3a). When divided into two subgroups according to the presence of preoperative treatment, this trend was observed in both nonpreoperative therapy (non-NACT group; n = 68, 5-year OS 43.4% vs. 67.7%, P = 0.0046) and the NACT group (n = 79, 5-year OS 38.0% vs. 77.0%, P = 0.0003) as shown in Supplemental Figs. S1a, d. Patients with high expression of CD44v6 tended to have worse OS than those with low CD44v6 expression in all cases but with no significant difference (5-year OS 49.0% vs. 61.7%, P = 0.21; Fig. 3b). A similar trend was observed in patients who underwent NACT (n = 79, 5-year OS 45.8% vs. 63.1%, P = 0.15) but comparable OS was obtained in the non-NACT group (n = 68, 5-year OS 52.6% vs. 60.4%, P = 0.82; Supplemental Figs. S1b, e).
When categorized into three subgroups based on the pattern of c-Met and CD44v6 immunostaining, the 5-year OS of patients with co-expression, iso-expression, and double-negative c-Met and CD44v6 expression were 36.8%, 52.9%, and 77.3%, respectively (Fig. 3c). Notably, patients with c-Met and CD44v6 co-expression had significantly worse OS than those with double-negative expression (P = 0.0002), whereas there was no significance compared with those with iso-expression (P = 0.37). This trend was more remarkable in the NACT group (Supplemental Figs. S1c, f). The 5-year OS of patients with co-expression, iso-expression, and double-negative c-Met and CD44v6 expression was 29.3%, 59.9%, and 80.4%, respectively. However, the OS of the three groups was comparable in the non-NACT group (56.2%, 42.2%, and 75.1%, respectively).
We further performed survival analysis according to c-Met/CD44v6 expression in pT and pN subgroups, respectively (Supplemental Figs S2a-d). In the pT1-2 group, patients co-expressing c-Met and CD44v6 had significantly worse OS than those with double-negative expression (5-year OS: 33.3% vs. 77.9%, P = 0.0014). Similarly, survival of patients co-expressing c-Met and CD44v6 tended to be worse as compared to those with iso-expression, however, with no statistical significance (5-year OS: 33.3% vs. 62.7%, P = 0.24). Although a similar trend was observed in the pT3-4 group, survival difference was not statistically significant among the three groups (5-year OS: double-negative vs. iso-expression vs. co-expression 75.0% vs. 43.7% vs. 39.7%, P = 0.32). On the other hand, in the pN1-3 group, patients co-expressing c-Met and CD44v6 showed significantly worse OS than those with double-negative expression (5-year OS: 18.9% vs. 83.3%, P = 0.0002). A similar trend was observed when comparing to those with iso-expression (5-year OS: 18.9% vs. 40.3%, P = 0.070). However, there was no significant difference of survival among the three groups in the pN0 group (5-year OS: double negative vs. iso-expression vs. co-expression 72.6% vs. 73.9% vs. 81.8%, P = 0.18).
Univariate analysis of OS identified pT, pN, pM stage, and co-expression of c-Met and CD44v6 as significant in all cases (P = 0.018, 0.0003, 0.0043, and 0.0050, respectively; Table 2). Multivariate analysis further identified c-Met and CD44v6 co-expression (hazard ratio [HR] 1.79, 95% confidence interval [CI] 1.03–3.04, P = 0.038) as independent prognostic factors, along with pN (HR 2.28, 95% CI 1.25–4.38, P = 0.0063) and pM stage (HR 2.46, 95% CI 1.05–5.08, P = 0.039; Table 2).
Discussion
In the present study, high expression of c-Met was significantly associated with advanced pT, pM, and the presence of preoperative chemotherapy in ESCC patients, whereas CD44v6-high expression correlated with advanced pT, pN, and poor clinical response to NACT. Notably, double-positive expression of c-Met and CD44v6 was identified in 28.6% of all cases and was shown to be an independent prognostic factor in ESCC, implying that both c-Met and CD44v6 could be therapeutic targets in ESCC.
c-Met expression has been reported to be elevated in several types of cancer, including lung, breast, ovary, kidney, colon, thyroid, liver, and gastric carcinoma. This upregulation of c-Met is considered to be the result of transcriptional activation, hypoxia-induced overexpression, or amplification.39 In fact, a DNA amplification rate of 9% has been reported for c-Met in both ESCC and esophageal adenocarcinoma.40,41 Downstream signaling of c-Met is well-known to prompt various biological activities in cancer cells, including cell motility, invasion, and metastasis, via the MAPK, P13 K/Akt, and ATAT pathways.10 On the other hand, expression of CD44v6, generated by alternative splicing of CD44, is reported to correlate with tumor progression, metastasis, and chemoresistance in prostate, colorectal, pancreatic, and esophageal cancer.42,43 Furthermore, clinical trials and in vivo studies of targeted therapy for c-Met or CD44v6 have been performed in several types of cancer, including hepatocellular, gastroesophageal, and pancreatic carcinoma.17,22,44 Because HGF-induced c-Met phosphorylation depends on CD44v6 for its activation or signaling, we considered it to be important to evaluate the expression status of both c-Met and CD44v6 to assess the biological features of resected ESCC specimens.22
The expression of c-Met and CD44v6 in ESCC, as determined by IHC, has been reported to be 43.3–69.2% and 22.0–77.6%, respectively, which is consistent with the present results. In addition, high expression of c-Met and CD44v6 is associated with advanced tumors and poor survival.25,26,27,28,29,38,43,45,46 In the present study, c-Met-high expression was significantly associated with advanced pT, pM stage, and unfavorable prognosis. High CD44v6 expression, on the other hand, correlated with advanced pT, pN stage, and poor clinical response to NACT, but it did not negatively influence the prognosis of ESCC patients. Furthermore, we evaluated the expression of both c-Met and CD44v6 in resected ESCC specimens for the first time, identifying 28.6% of all patients as double-positive, which was confirmed using an independent prognostic factor.
In the current study, the c-Met-high group had a significantly higher proportion of patients with NACT than the c-Met-low group, and CD44v6-high expression was significantly related to a poorer clinical response to NACT. This finding is consistent with previous reports, because these molecules are considered to be a cancer stem-cell marker associated with tumor initiation, propagation, and resistance to conventional chemotherapy and radiation, as previously reported in several types of cancer.10,11,23,24 Considering the prognostic implication of double-positive c-Met and CD44v6 expression as an independent prognostic factor, these results imply that a novel therapy targeting both c-Met and CD44v6 may be more effective than one targeting each of them in ESCC.
We acknowledge some limitations to this study. First, we did not analyze the more precise status of “co-expression” of c-Met and CD44v6 by comparing the localization of immunostaining in serial sections of resected specimen. Second, the phosphorylation status of c-Met or expression of HGF, a ligand of c-Met, was not evaluated. Finally, although we assessed the relationship between chemoresistance and the expression of these two molecules by IHC in resected specimens from patients who underwent NACT, the more detailed relationship between chemoresistance and the expression of two molecules could be clarified by a comparison with IHC results in pretherapeutic biopsy samples.
Conclusions
This report is the first to clarify the correlation between c-Met and CD44v6 expression as determined by IHC and the impact of this expression on clinicopathological parameters, including prognosis, in ESCC. Although the present results should be validated in the future, they may help us to understand further the mechanism of chemoresistance in ESCC. In addition, these two molecules could be therapeutic targets in ESCC.
References
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65(2):87–108.
Ando N, Kato H, Igaki H, et al. A randomized trial comparing postoperative adjuvant chemotherapy with cisplatin and 5-fluorouracil versus preoperative chemotherapy for localized advanced squamous cell carcinoma of the thoracic esophagus (JCOG9907). Ann Surg Oncol. 2012;19(1):68–74.
Makino T, Doki Y, Miyata H, et al. Use of (18)F-fluorodeoxyglucose-positron emission tomography to evaluate responses to neo-adjuvant chemotherapy for primary tumor and lymph node metastasis in esophageal squamous cell carcinoma. Surgery. 2008;144(5):793–802.
Bahrami A, Shahidsales S, Khazaei M, et al. C-Met as a potential target for the treatment of gastrointestinal cancer: Current status and future perspectives. J Cell Physiol. 2017;232(10):2657–73.
Boccaccio C, Comoglio PM. Invasive growth: a MET-driven genetic programme for cancer and stem cells. Nat Rev Cancer. 2006;6(8):637–45.
Trusolino L, Bertotti A, Comoglio PM. MET signalling: principles and functions in development, organ regeneration and cancer. Nat Rev Mol Cell Biol. 2010;11(12):834–48.
Tomihara H, Yamada D, Eguchi H, et al. MicroRNA-181b-5p, ETS1, and the c-Met pathway exacerbate the prognosis of pancreatic ductal adenocarcinoma after radiation therapy. Cancer Sci. 2017;108(3):398–407.
Kim JH, Kim BJ, Kim HS. Clinicopathological impacts of high c-Met expression in head and neck squamous cell carcinoma: a meta-analysis and review. Oncotarget. 2017;8(68):113120–8.
Wilson GD, Thibodeau BJ, Fortier LE, et al. Cancer stem cell signaling during repopulation in head and neck cancer. Stem Cells Int. 2016;2016:1894782.
Li C, Wu JJ, Hynes M, et al. c-Met is a marker of pancreatic cancer stem cells and therapeutic target. Gastroenterology. 2011;141(6):2218–27 e2215.
Sun S, Wang Z. Head neck squamous cell carcinoma c-Met(+) cells display cancer stem cell properties and are responsible for cisplatin-resistance and metastasis. Int J Cancer. 2011;129(10):2337–48.
Bicaku E, Xiong Y, Marchion DC, et al. In vitro analysis of ovarian cancer response to cisplatin, carboplatin, and paclitaxel identifies common pathways that are also associated with overall patient survival. Br J Cancer. 2012;106(12):1967–75.
Aebersold DM, Kollar A, Beer KT, Laissue J, Greiner RH, Djonov V. Involvement of the hepatocyte growth factor/scatter factor receptor c-met and of Bcl-xL in the resistance of oropharyngeal cancer to ionizing radiation. Int J Cancer. 2001;96(1):41–54.
De Bacco F, Luraghi P, Medico E, et al. Induction of MET by ionizing radiation and its role in radioresistance and invasive growth of cancer. J Natl Cancer Inst. 2011;103(8):645–61.
Chen GZ, Dai WS, Zhu HC, et al. Foretinib enhances the radiosensitivity in esophageal squamous cell carcinoma by inhibiting phosphorylation of c-Met. J Cancer. 2017;8(6):983–92.
Scagliotti GV, Shuster D, Orlov S, et al. Tivantinib in combination with erlotinib versus erlotinib alone for EGFR-Mutant NSCLC: an exploratory analysis of the Phase 3 MARQUEE Study. J Thorac Oncol. 2018;13(6):849–54.
Pant S, Patel M, Kurkjian C, et al. A Phase II Study of the c-Met inhibitor tivantinib in combination with FOLFOX for the treatment of patients with previously untreated metastatic adenocarcinoma of the distal esophagus, gastroesophageal junction, or stomach. Cancer Invest. 2017;35(7):463–72.
Rimassa L, Abbadessa G, Personeni N, et al. Tumor and circulating biomarkers in patients with second-line hepatocellular carcinoma from the randomized phase II study with tivantinib. Oncotarget. 2016;7(45):72622–33.
Lai AZ, Abella JV, Park M. Crosstalk in Met receptor oncogenesis. Trends Cell Biol. 2009;19(10):542–51.
Tremmel M, Matzke A, Albrecht I, et al. A CD44v6 peptide reveals a role of CD44 in VEGFR-2 signaling and angiogenesis. Blood. 2009;114(25):5236–44.
Orian-Rousseau V, Chen L, Sleeman JP, Herrlich P, Ponta H. CD44 is required for two consecutive steps in HGF/c-Met signaling. Genes Dev. 2002;16(23):3074–86.
Matzke-Ogi A, Jannasch K, Shatirishvili M, et al. Inhibition of tumor growth and metastasis in pancreatic cancer models by interference with CD44v6 signaling. Gastroenterology. 2016;150(2):513–25 e510.
Snyder EL, Bailey D, Shipitsin M, Polyak K, Loda M. Identification of CD44v6(+)/CD24- breast carcinoma cells in primary human tumors by quantum dot-conjugated antibodies. Lab Invest. 2009;89(8):857–66.
Todaro M, Gaggianesi M, Catalano V, et al. CD44v6 is a marker of constitutive and reprogrammed cancer stem cells driving colon cancer metastasis. Cell Stem Cell. 2014;14(3):342–56.
Ozawa Y, Nakamura Y, Fujishima F, et al. c-Met in esophageal squamous cell carcinoma: an independent prognostic factor and potential therapeutic target. BMC Cancer. 2015;15:451.
Zhang T, Li H, Zhang Y, Wang P, Bian H, Chen ZN. Expression of proteins associated with epithelial-mesenchymal transition in esophageal squamous cell carcinoma. Oncol Lett. 2018;15(3):3042–8.
Yang H, Liu J, Yu H, et al. Expression and association of CD44v6 with prognosis in T2-3N0M0 esophageal squamous cell carcinoma. J Thorac Dis. 2014;6(2):91–8.
Ren JL, Wu HF, Wang WJ, et al. C-Met as a potential novel prognostic marker in squamous cell carcinoma and adenocarcinoma of esophagus: evidence from a meta-analysis. Panminerva Med. 2017;59(1):97–106.
Xu Y, Peng Z, Li Z, et al. Expression and clinical significance of c-Met in advanced esophageal squamous cell carcinoma. BMC Cancer. 2015;15:6.
Sobin LH, Gospodarowicz MK, Wittekind C. TNM classification of malignant tumors. 7th edn. 2010, Oxford.
Yamashita K, Makino T, Yamasaki M, et al. Comparison of short-term outcomes between 2- and 3-field lymph node dissection for esophageal cancer. Dis Esophagus. 2017;30(11):1–8.
Miyata H, Yamasaki M, Miyazaki Y, et al. Clinical importance of supraclavicular lymph node metastasis after neoadjuvant chemotherapy for esophageal squamous cell carcinoma. Ann Surg. 2015;262(2):280–5.
Makino T, Yamasaki M, Miyata H, et al. Solitary Lymph node recurrence of esophageal squamous cell carcinoma: surgical failure or systemic disease? Ann Surg Oncol. 2016;23(6):2087–93.
Colvin H, Mizushima T, Eguchi H, Takiguchi S, Doki Y, Mori M. Gastroenterological surgery in Japan: the past, the present and the future. Ann Gastroenterol Surg. 2017;1(1):5–10.
Miyata H, Yamasaki M, Takahashi T, et al. Relevance of [18F]fluorodeoxyglucose positron emission tomography-positive lymph nodes after neoadjuvant chemotherapy for squamous cell oesophageal cancer. Br J Surg. 2013;100(11):1490–7.
Makino T, Yamasaki M, Tanaka K, et al. Metabolic tumor volume change predicts long-term survival and histological response to preoperative chemotherapy in locally advanced esophageal cancer. Ann Surg. 2018. https://doi.org/10.1097/SLA.0000000000002808
Diseases JSfE. Guidelines for the clinical and pathologic studies on carcinoma of the esophagus. 11th edn. 2015, Tokyo, Japan.
Xu YP, Lin G, Sun XJ, et al. C-Met as a molecular marker for esophageal squamous cell carcinoma and its association with clinical outcome. J Cancer. 2016;7(5):587–94.
Sierra JR, Tsao MS. c-MET as a potential therapeutic target and biomarker in cancer. Ther Adv Med Oncol. 2011;3(1 Suppl):S21–35.
Dulak AM, Stojanov P, Peng S, et al. Exome and whole-genome sequencing of esophageal adenocarcinoma identifies recurrent driver events and mutational complexity. Nat Genet. 2013;45(5):478–86.
Bandla S, Pennathur A, Luketich JD, et al. Comparative genomics of esophageal adenocarcinoma and squamous cell carcinoma. Ann Thorac Surg. 2012;93(4):1101–6.
Chen C, Zhao S, Karnad A, Freeman JW. The biology and role of CD44 in cancer progression: therapeutic implications. J Hematol Oncol. 2018;11(1):64.
Hu B, Luo W, Hu RT, Zhou Y, Qin SY, Jiang HX. Meta-analysis of prognostic and clinical significance of CD44v6 in esophageal cancer. Medicine (Baltimore). 2015;94(31):e1238.
Rimassa L, Assenat E, Peck-Radosavljevic M, et al. Tivantinib for second-line treatment of MET-high, advanced hepatocellular carcinoma (METIV-HCC): a final analysis of a phase 3, randomised, placebo-controlled study. Lancet Oncol. 2018;19(5):682–93.
Wang H, Jiang D, Song Q, et al. Prognostic impact and potential interaction of EGFR and c-Met in the progression of esophageal squamous cell carcinoma. Tumour Biol. 2016;37(7):9771–9.
Zheng B, Ni CH, Chen H, et al. New evidence guiding extent of lymphadenectomy for esophagogastric junction tumor: Application of Ber-Ep4 Joint with CD44v6 staining on the detection of lower mediastinal lymph node micrometastasis and survival analysis. Medicine (Baltimore). 2017;96(14):e6533.
Author information
Authors and Affiliations
Contributions
TH, TM, MY, MM, and YD: conception, design of the study; TH, TM, MY, KT, and YD: surgery and acquisition of clinical data; TH: immunohistochemical staining; TH, TM, and NM: evaluation of immunohistochemical staining; TH, TM, YM, TT, YK, and KN: analysis of data; TH: drafting the article; TM: critical revision of the article; MM and YD: final approval of the article.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Fig. S1
Kaplan-Meier survival analysis of overall survival among patients who (a) did not receive preoperative therapy classified by c-Met expression, (b) did not receive preoperative therapy classified by CD44v6 expression, (c) did not receive preoperative therapy classified by c-Met and CD44v6 expression, (d) received preoperative chemotherapy classified by c-Met expression, (e) received preoperative chemotherapy classified by CD44v6 expression, and (f) received preoperative chemotherapy classified by c-Met and CD44v6 expression (TIFF 193 kb)
Supplemental Fig. S2
Kaplan-Meier survival analysis of overall survival according to c-Met/CD44v6 expression in (a) pT1-2 group, (b) pN0 group, (c) pT3-4 group, (d) pN1-3 group (TIFF 154 kb)
Rights and permissions
About this article
Cite this article
Hara, T., Makino, T., Yamasaki, M. et al. Effect of c-Met and CD44v6 Expression in Resistance to Chemotherapy in Esophageal Squamous Cell Carcinoma. Ann Surg Oncol 26, 899–906 (2019). https://doi.org/10.1245/s10434-018-07126-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-018-07126-5