Abstract
Background
Low-grade appendiceal mucinous neoplasm (LAMN) is the most common primary lesion of pseudomyxoma peritonei, a disease whose standard treatment is cytoreduction and hyperthermic intraperitoneal chemotherapy. The optimal management of LAMN is not well defined. This study prospectively assessed a clinical surveillance strategy for LAMN with or without limited peritoneal spread.
Methods
During 2003–2017, the study prospectively enrolled 41 patients treated by macroscopically complete surgery for LAMN with or without limited peritoneal spread (pelvis and right lower quadrant). Follow-up assessment included thoracic-abdomino-pelvic computed tomography scan and serum tumor markers scheduled after surgery, then every 6 months for 5 years, and yearly thereafter. All specimens were reviewed by a dedicated pathologist.
Results
Appendectomy and five right colectomies were performed for 36 patients. Nine patients also underwent macroscopically complete cytoreduction of mucinous peritoneal disease, and four patients had hysterectomy plus bilateral salphingo-oophorectomy. Appendiceal rupture was evaluable in 38 of the 41 patients, being present in 21 patients (51.2%). Mucin, cells, or both outside the appendix were observed in 24 patients (58.5%). The median follow-up period was 58 months (range 9.3–162 months). The 5-year recurrence-free survival rate was 95.1%. Only two patients experienced peritoneal recurrences (4.9%), respectively 18 and 22 months after appendectomy. Their primary lesions were LAMNs with and without appendix wall rupture or extra-appendiceal mucin, respectively. No death occurred.
Conclusion
These findings strongly suggest that radically resected LAMN, even with limited peritoneal spread, carries a low recurrence risk. Furthermore, appendix wall perforation and the presence of mucin, cells, or both outside the appendix were not associated with a higher risk of metachronous peritoneal dissemination. In this setting, clinical and radiologic surveillance is a viable choice.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Mucinous neoplasms comprise of 85% of all epithelial appendiceal neoplasms. The most common presentation is that of an appendiceal mucocele, which constitutes 0.2–0.3% of all appendectomies.1 Mucinous tumors of the appendix usually arise within a mucocele and may be histologically classified as low or high grade according to the degree of cytologic atypia and invasiveness.2,3,4
A distinctive feature of appendiceal mucinous neoplasms is their proclivity to spread to the peritoneum, giving rise to pseudomyxoma peritonei (PMP), a clinical entity characterized by progressive accumulation of mucinous ascites and peritoneal mucinous implants.5 Long-term survival for PMP patients treated with serial debulking, palliative chemotherapy, or both is only 20–25% for high-grade lesions and 18–45% for low-grade lesions.6,7,8,9 A treatment approach involving cytoreductive surgery (CRS) and hyperthermic intraperitoneal chemotherapy (HIPEC) has reportedly resulted in survival improvements over historical and contemporary control treatments and currently is accepted as standard treatment.10,11,12,13,14
Clinical guidelines are available for the management of appendiceal lesions unexpectedly encountered during surgery for appendicitis or found incidentally either after pathologic examination of appendectomy specimens or on radiologic cross-sectional studies.15,16 Histopathologic features and extent of peritoneal disease determine further treatments and prognosis. Referral to specialized peritoneal malignancy management centers is recommended for patients with peritoneal dissemination. Conversely, appendectomy generally is regarded as adequate treatment for low-grade appendiceal mucinous neoplasms (LAMNs) confined to the appendix, and right colectomy for tumors with high-grade histology, nodal involvement, or both.12,15,16,17
Due to the perceived risk for the development of widespread PMP even after apparently radical surgery and the improved survival outcomes after CRS-HIPEC,10,11,12,13,14,17 a few centers have developed more aggressive treatment approaches, such as prophylactic HIPEC or staged abdominal exploration for early diagnosis and treatment.18,19 However, the potential benefit of these additional proceduresmust be balanced against their economic costs and operative morbidity. Furthermore, the magnitude of the risk for peritoneal dissemination associated with LAMN still is poorly defined, even in cases with appendiceal rupture or minimal extra-appendiceal dissemination, which generally are considered pathologic features related to higher risk.4,20,21,22,23
From 2003, patients referred to our institution after macroscopically complete surgical resection for LAMN with or without minimal extra-appendiceal spread have been prospectively selected to undergo close clinical observation. The current report describes the outcome of our strategy with the aim of providing information to rationalize the clinical management of these patients.
Patients and Methods
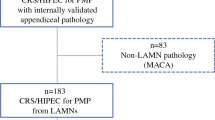
All the patients included in this study were treated according to a protocol (Fig. 1) approved by the institutional ethics committee according to the principles of the Helsinki declaration.
Between January 2003 and December 2016, 48 consecutive patients with appendiceal mucinous neoplasm were referred to our institution. All but one patient were treated elsewhere for their primary appendiceal tumors and then referred to us for a second opinion and further management. This one patient underwent computed tomography (CT) enema scan for colorectal neoplasm screening, which showed a well-circumscribed low-attenuation mass contiguous with the cecum. He underwent laparoscopic exploration, with no evidence of peritoneal disease, and appendectomy. At the pathologic examination, a LAMN was diagnosed. Such a referral pattern is presumably consistent with our institution as a comprehensive cancer center with no emergency department. During the study period, 164 patients underwent CRS-HIPEC at our institution for low-grade PMP originating from LAMN, according to criteria reported elsewhere.24
Baseline Evaluation
Baseline evaluation consisted of a thorough clinical history and physical examination. Operative reports, including recorded videos when available, were reviewed to assess disease extent and quality of surgery. All patients underwent a thoracic-abdominal-pelvic CT scan and serum tumor marker measurement (CEA, CA19.9, CA125, CA15.3). Additional diagnostic procedures such as colonoscopy, magnetic resonance imaging, or positron-emission tomography were performed at the discretion of surgeons of the Peritoneal Malignancy Program.
The histopathologic slides of all the patients were reviewed by an expert pathologist. The appendectomy specimens of 21 patients were entirely submitted to pathologic examination. Additional paraffin blocks were requested from referring hospitals for nine patients. Overall, a mean of ten appendix slides (range 5–22) and seven peritoneum slides (range 2–18) were reviewed. In this study, LAMN was pathologically defined according to the original description by Misdraji et al.,4 subsequently adopted by the World Health Organization (WHO)25 and Peritoneal Surface Oncology Group (PSOGI) consensus classification.26,27
Patients were included in the surveillance protocol if they met the following criteria: diagnosis of LAMN confirmed in our pathology department, peritoneal mucinous disease absent or confined to the right lower quadrant and/or the pelvis [peritoneal cancer index (PCI) ≤ 3],11 complete resection at the time of primary resection, negative CT scan and serum tumor markers, and willingness to undergo regular follow-up assessment. Patients older than 70 years with poor clinical conditions (WHO score > 2) or severe comorbidities contraindicating major surgery, such as CRS-HIPEC, were offered clinical surveillance, but they were not formally included in the current study.
Long-Term Follow-up Evaluation
For each patient, the final decision to start follow-up evaluation was made at multidisciplinary meetings involving both surgical and medical oncologists, pathologists, and radiologists with expertise in peritoneal surface oncology. Follow-up visits were conducted by surgeons of the Peritoneal Malignancy Program. Physical examination, CT scan, and serum marker measurements were performed 3 months after baseline evaluation, and then every 6 months during the first 5 years, and yearly thereafter.
Disease progression was defined as the detection of any CT scan abnormality compatible with mucinous tumor, with or without elevation of serum tumor markers. Patients with documented or suspected disease relapse were discussed at multidisciplinary team meetings and managed according to the following guidelines. The treatment of choice was CRS-HIPEC unless contraindicated. Palliative surgery, systemic chemotherapy, or supportive care, as indicated, was considered for cases unsuitable for CRS-HIPEC. Aggressive diagnostic management by imaging-guided percutaneous needle biopsy or laparoscopic exploration was considered for patients with non-conclusive radiologic findings. In cases showing isolated elevation of serum tumor markers at two consecutive follow-up visits, with no progressive disease documented on imaging or clinical symptoms, laparoscopic exploration was offered.
Statistical Analysis
Continuous variables were described as median and range, and categorical variables were described as frequencies and percentages. Recurrence-free survival (RFS) was determined from the date of appendectomy to the date of first recurrence. Survival curves were calculated using the Kaplan–Meier method. All statistical analyses were conducted using SPSS version 18.0.0 (IBM Corporation, Armonk, NY, USA).
Results
The study excluded seven patients due to pathologic review of appendectomy slides showing mucinous carcinoma (n = 2), persistence of disease at the baseline CT scan (n = 3), and refusal to undergo regular follow-up visits (n = 2). The clinical-pathologic characteristics of the 41 patients included in the study are summarized in Table 1. The median age was 43.3 years, and 23 patients (53%) were females. The baseline tumor markers were normal in all the patients.
At the time of primary surgery, appendectomy was the most common procedure performed. Besides appendectomy, nine patients (21.9%) had macroscopically complete removal of pelvic mucinous peritoneal implants, mucinous ascites, or both, and four patients had hysterectomy with bilateral salphingo-ophorectomy (9.7%). Five patients (12.2%) underwent right colectomy. One patient underwent appendectomy concurrently with cesarean section. After baseline assessment, all patients were deemed to have had macroscopically complete surgery.
After pathologic evaluation of slides in our pathology department, all appendiceal lesions were classified as LAMN. The appendectomy resection margin was clear in all patients. Appendiceal rupture with acellular mucin outside the appendix wall was demonstrated in 19 patients (46.3%), nine of whom had mucin collection in the pelvis. In three patients (7.3%), cellular mucin was found in the pelvis, on appendiceal serosa, or both. Two additional patients (4.9%) had direct infiltration of the ovary and right colon, respectively, by appendiceal primary. Acellular mucin deposits on appendiceal serosa with no clear evidence of appendix wall perforation were detected in two patients (4.9%).
Patient Outcomes
The median follow-up period was 58 months (range 9.3–162 months). Disease recurrence was demonstrated in two patients (4.9%), accounting for a 5-year RFS of 95.2% (Fig. 2; Table 2). No deaths occurred during the study period. The small numbers of events precluded further analyses of potential prognostic factors.
Both relapsed patients were offered CRS-HIPEC. The first patient had a carcinoembryonic antigen (CEA) increase (40 ng/mL) and a CT scan showing disease progression 18 months after appendectomy. His appendectomy specimen was entirely submitted to pathologic examination. Slide review confirmed the diagnosis of LAMN without rupture or extra-appendiceal mucin. The patient underwent CRS-HIPEC. The intraoperative PCI was 7, and no bowel resection was needed to achieve macroscopically complete cytoreduction. At this writing, he currently is alive with no recurrent disease after 20 months.
The second patient was a female who had CT scan showing mucin collection in the right iliac region and a serum CEA increase (28 ng/mL) 22 months after primary surgery. Her radiologic PCI was 4. Slide review had demonstrated LAMN with appendix wall rupture and mucin deposit on appendix serosa. The patient refused CRS-HIPEC and at this writing is alive with stable disease and no symptoms after 12 months (Table 3).
Discussion
The best management of unexpected LAMN still is a matter of discussion. Reasons are probably related to the low incidence and indolent behavior of LAMNs, which make it difficult to define standardized and widely accepted treatment strategies. In particular, the rate at which LAMN tumors disseminate and develop low-grade PMP has not been clearly established. This study aimed to follow patients prospectively who had undergone macroscopically complete surgery for LAMN and to assess the impact of a clinical surveillance strategy on long-term outcome.
The current series consisted of 41 patients with LAMN confined to the appendix or with low-volume peritoneal disease completely resected at the time of primary surgery. After a median follow-up period of 58 months, our approach has resulted in a 5-year recurrence-free rate of 95.2% and no patient deaths. These data do not support an aggressive approach for these patients, but rather suggest that strict clinical-radiologic surveillance is a reasonable option.
Thorough understanding of the natural history of diseases is essential to the development of treatment and follow-up guidelines. Currently, data to define the rate of LAMN progression to PMP is scarce. In a Dutch retrospective population study, PMP incidence was 20% of 547 patients with LAMN, with the majority of PMP detected within 24 months after appendectomy. On this basis, the authors recommended performing CT scan at least 5 years for incidental LAMN.28 However, they did not provide any information on the initial treatments received by these patients. A similar 26% rate of progression to PMP is reported in a series of 98 LAMNs and uncertain malignant potential lesions retrospectively identified from the appendiceal tumor databases at the MD Anderson Cancer Center.20 Our 4.9% recurrence rate compares favorably with these series, presumably due to our careful baseline assessment to define biologic aggressiveness, quality of surgery, and presence versus absence of demonstrable residual disease in patients referred to our center.
Analogously to colorectal cancer,29 more aggressive prophylactic approaches also have been proposed for LAMN patients. McDonald et al.18 considered appendiceal rupture, mucin spillage in the abdominal cavity, and evidence of mucin, neoplastic epithelium, or both in the appendiceal wall as strong risk factors for PMP development. They categorized lesions exhibiting these pathologic features as type 2 LAMN and lesions confined to the appendiceal lumen as type 1 LAMN. Type 1 lesions were managed by a watch-and-wait policy for 14 of 16 patients, and type 2 lesions by “prophylactic” CRS-HIPEC for 17 of 27 patients, with low morbidity and no disease progression in either group after a median follow-up period of 40 months.
Foster et al.19 monitored 22 patients with had incidental LAMN using tumor markers and CT scan. Laparoscopic exploration was performed, either in the event of positive imaging or after 12 months of no radiographic disease. Peritoneal disease was detected in five patients (23%). Only one case was detected on imaging and confirmed with laparoscopy, whereas four cases were detected only by laparoscopy. Four patients were treated with CRS-HIPEC and one with CRS alone. At this writing, the 17 patients with negative laparoscopy remain disease free after a median follow-up period of 50 months.
This series of Foster et al.19 shares with our series a rigorous selection and surveillance protocol. The rates of PMP detected on imaging in the study of Foster et al.19 (1/22) and in our study (2/40) were similar. We did not routinely include laparoscopy in our surveillance protocol, but we chose to explore only selected patients with inconclusive CT scan findings. The paper by Foster suggests a possible delay in detecting peritoneal recurrences by means of imaging compared with laparoscopy.19 However, no data exist to show that anticipating PMP diagnosis at a subclinical stage may translate into a survival advantage or less aggressive and morbid surgery. We considered LAMN and the related low-grade PMP as poorly aggressive, slow-growing conditions, with exceedingly uncommon or no nodal or distant metastases. As a matter of fact, the outcomes of CRS-HIPEC for patients with the development of PMP in both our series (n = 2) and the series by Foster et al.19 (n = 4) were favorable in terms of low PCI, optimal cytoreduction, low treatment-related morbidity, and survival. Due to the small number of cases, it is virtually impossible to compare the two different strategies.
The risk factors for the development of PMP after surgical resection of LAMN still are poorly known. Extra-appendiceal epithelial cells, mucin, or both are regarded as adverse prognostic variables. Misdraji et al.4 found that 27 patients with LAMN confined to the appendix were alive at 6 years without any evidence of disease recurrence, but the 10-year survival rate for LAMN patients with extra-appendiceal spread was only 45%.4 In contrast, among patients with LAMN confined to the appendix treated by appendicectomy, Arnason et al.30 found that the presence of neoplastic epithelium or acellular mucin at the proximal appendiceal margin was not associated with recurrent disease. Interestingly, Yantiss et al.31 reported that 2 (4%) of 49 patients with acellular periappendiceal mucin experienced diffuse peritoneal disease, exhibiting a relapse rate similar to that seen in our series. In contrast, 5 (33%) of 15 patients with cellular periappendiceal mucin experienced PMP.
In our series, extra-appendicolar mucin, cells, or both were demonstrated in 21 patients (51.2%), and 11 of these patients had disease beyond the right iliac region (n = 9) or involving adjacent organs (n = 2). Although such a considerable proportion of our LAMN patients presented with potential pathologic risk factors for PMP development, PMP developed in only a small minority during the study period. This seems to be consistent with the findings of Foster et al.,19 who reported extra-appendiceal mucin, cells, or both in all patients who had PMP development (n = 5) but also in 90% of all the patients included in their study, and 13 of 18 patients without detectable occult peritoneal metastasis at laparoscopy. Taken together, these data suggest that no definitive conclusion can be drawn in this clinical setting.
We recognize several limitations of our study, including the relatively small number of cases and events, which may have precluded further analyses. Given the indolent course of these low-grade neoplasms, a longer follow-up period would be required for a better assessment of the risk for peritoneal recurrence and the impact on survival. In addition, because our center is a tertiary cancer center, many of the slides from the resections come to us from outside facilities, limiting evaluation of the entire appendix and margin status.
Conclusions
Our findings clearly suggest that for patients with incidental LAMN, the risk of recurrence is low, and no prophilactic cytoreductive surgery is requested. Appendix wall perforation or extra-appendiceal mucin cells detected at pathologic examination do not seem to be a specific risk factor provided that low-volume localized PMP has been completely resected. For these patients, a strict clinical surveillance is associated with a favorable long-term outcome. Larger prospective studies with longer follow-up periods are needed to confirm these findings.
Change history
19 January 2018
In the original article Massimo Milione’s last name was spelled incorrectly. It is correct as reflected here. The original article has also been updated.
19 January 2018
In the original article Massimo Milione?s last name was spelled incorrectly. It is correct as reflected here. The original article has also been updated.
19 January 2018
In the original article Massimo Milione?s last name was spelled incorrectly. It is correct as reflected here. The original article has also been updated.
References
Malya FU, Hasbahceci A, Sertr G, et al. Appendiceal mucocele: clinical and imaging features of 14 cases. Chirurgia Bucur. 2014;109:788–93.
Connor SJ, Hanna GB, Frizelle FA. Appendiceal tumors: retrospective clinicopathologic analysis of appendiceal tumors from 7970 appendectomies. Dis Colon Rectum. 1998;41:75–80.
McCusker ME, Cote TR, Clegg LX, et al. Primary malignant neoplasms of the appendix: a population-based study from the surveillance, epidemiology, and end-results program, 1973–1998. Cancer. 2002;94:3307–12.
Misdraji J, Yantiss RK, Graeme-Cook FM, et al. Appendiceal mucinous neoplasms: a clinicopathologic analysis of 107 cases. Am J Surg Pathol. 2003;27:1089–103.
Smeenk RM, van Velthuysen ML, Verwaal VJ, Zoetmulder FA. Appendiceal neoplasms and pseudomyxoma peritonei: a population-based study. Eur J Surg Oncol. 2008;34:196–201.
Fernandez RN, Daly GM. Pseudomyxoma peritonei. Arch Surg. 1980;115:409–14.
Gough DB, Donohue JH, Schutt AJ, et al. Pseudomyxoma peritonei: long-term patient survival with an aggressive regional approach. Ann Surg. 1994;219:112–9.
Miner TJ, Shia J, Jaques DP, et al. Long-term survival following treatment of pseudomyxoma peritonei: an analysis of surgical therapy. Ann Surg. 2005;241:300–8.
Järvinen P, Järvinen HJ, Lepistö A. Survival of patients with pseudomyxoma peritonei treated by serial debulking. Colorectal Dis. 2010;12:868–72.
Chua TC, Moran BJ, Sugarbaker PH, et al. Early- and long-term outcome data of patients with pseudomyxoma peritonei from appendiceal origin treated by a strategy of cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. J Clin Oncol. 2012;30:2449–56.
Moran B, Baratti D, Yan TD, et al. Consensus statement on the locoregional treatment of appendiceal mucinous neoplasms with peritoneal dissemination (pseudomyxoma peritonei). J Surg Oncol. 2008;98:277–82.
Sugarbaker PH. New standard of care for appendiceal epithelial neoplasms and pseudomyxoma peritonei syndrome? Lancet Oncol. 2006;7:69–76.
Baratti D, Kusamura S, Nonaka D, Cabras AD, Laterza B, Deraco M. Pseudomyxoma peritonei: biological features are the dominant prognostic determinants after complete cytoreduction and hyperthermic intraperitoneal chemotherapy. Ann Surg. 2009;249:243–9.
Ansari N, Chandrakumaran K, Dayal S, Mohamed F, Cecil TD, Moran BJ. Cytoreductive surgery and hyperthermic intraperitoneal chemotherapy in 1000 patients with perforated appendiceal epithelial tumours. Eur J Surg Oncol. 2016;42:1035–41.
Murphy EMA, Farquharson SM, Moran BJ. Management of an unexpected appendiceal neoplasm. Br J Surg. 2006;93:783–92.
Barrios P, Losa F, Gonzalez-Moreno S. Rojo A, Go´mez-Portilla A, Bretcha-Boix P, et al. Recommendations in the management of epithelial appendiceal neoplasms and peritoneal dissemination from mucinous tumours (pseudomyxoma peritonei). Clin Transl Oncol. 2016;18:437–48.
Gonzalez-Moreno S, Sugarbaker PH. Right hemicolectomy does not confer a survival advantage in patients with mucinous carcinoma of the appendix and peritoneal seeding. Br J Surg. 2004;91:304–11.
McDonald JR, O’Dwyer ST, Rout S, et al. Classification of and cytoreductive surgery for low-grade appendiceal mucinous neoplasms. Br J Surg. 2012;99:987–9.
Foster JM, Sleightholm RL, Wahlmeier S, Loggie B, Sharma P, Patel A. Early identification of DPAM in at-risk low-grade appendiceal mucinous neoplasm patients: a new approach to surveillance for peritoneal metastasis. World J Surg Oncol. 2016;14:243.
Fournier K, Rafeeq S, Taggart M, et al. Low-grade appendiceal mucin mucinous neoplasm of uncertain malignant potential (LAMN-UMP): prognostic factors and implications for treatment and follow-up. Ann Surg Oncol. 2017;24:187–93.
Pai RK, Beck AH, Norton JA, Longacre TA. Appendiceal mucinous neoplasms: clinicopathologic study of 116 cases with analysis of factors predicting recurrence. Am J Surg Pathol. 2009;33:1425–39.
Young RH. Pseudomyxoma peritonei and selected other aspects of the spread of appendiceal neoplasms. Semin Diagn Pathol. 2004;21:134–50.
Panarelli NC, Yantiss RK. Mucinous neoplasms of the appendix and peritoneum. Arch Pathol Lab Med. 2011;135:1261–8.
Baratti D, Kusamura S, Milione M, et al. Pseudomyxoma peritonei of extra-appendiceal origin: a comparative study. Ann Surg Oncol. 2016;23:4222–30.
Carr NJ, Sobin LH. Adenocarcinoma of the appendix. In: Bosman F, Carneiro F, Hruban R, Theise N (ed). WHO classification of tumours of the digestive system. International Agency for Research on Cancer, Lyon, 2010, pp 122–25.
Carr NJ, Cecil TD, Mohamed F, et al. A consensus for classification and pathologic reporting of pseudomyxoma peritonei and associated appendiceal neoplasia: the results of the Peritoneal Surface Oncology Group International (PSOGI) modified Delphi process. Am J Surg Pathol. 2016;40:14–26.
Carr NJ, Bibeau F, Bradley RF, et al. The histopathological classification, diagnosis, and differential diagnosis of mucinous appendiceal neoplasms, appendiceal adenocarcinomas, and pseudomyxoma peritonei. Histopathology. 2017;71:847–58.
Smeenk RM, van Velthuysen ML, Verwaal VJ, Zoetmulder FA. Appendiceal neoplasms and pseudomyxoma peritonei: a population-based study. Eur J Surg Oncol. 2008;34:196–201.
Baratti D, Kusamura S, Iusco D, et al. Hyperthermic intraperitoneal chemotherapy (HIPEC) at the time of primary curative surgery in patients with colorectal cancer at high risk for metachronous peritoneal metastases. Ann Surg Oncol. 2017;24:167–75.
Arnason T, Kamionek M, Yang M, et al. Significance of proximal margin involvement in low-grade appendiceal mucinous neoplasms. Arch Pathol Lab Med. 2015;139:518–21.
Yantiss RK, Shia J, Klimstra DS, Hahn HP, Odze RD, Misdraji J. Prognostic significance of localized extra-appendiceal mucin deposition in appendiceal mucinous neoplasms. Am J Surg Pathol. 2009;33:248–55.
Acknowledgement
The study was partially supported by a grant of the Italian Association for Cancer Research (AIRC). Snita Sinukumar is a fellow of the European School of Peritoneal Surface Oncology (ESPSO).
Disclosure
The authors have no conflicts of interest or financial disclosures to declare.
Author information
Authors and Affiliations
Corresponding author
Additional information
The original version of this article was revised: Massimo Milione’s last name was spelled incorrectly.
Rights and permissions
About this article
Cite this article
Guaglio, M., Sinukumar, S., Kusamura, S. et al. Clinical Surveillance After Macroscopically Complete Surgery for Low-Grade Appendiceal Mucinous Neoplasms (LAMN) with or Without Limited Peritoneal Spread: Long-Term Results in a Prospective Series. Ann Surg Oncol 25, 878–884 (2018). https://doi.org/10.1245/s10434-017-6305-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-017-6305-5