Abstract
Background
Skeletal muscle mishaps are the most well-known incidents in society, especially among athletes and the military population. From the various urgency, this accident needs to be cured more quickly. However, the current treatment still has some shortcomings and is less effective. In this case, Paired box 3 and Paired box 7 (Pax3/Pax7) proteins that induce stem cells could potentially be an alternative treatment for skeletal muscle injuries. This paper aimed to analyse the potential treatment of Pax3/Pax7 proteins inducing the stem cell for skeletal muscle injuries.
The main body of the abstract
We did a narrative review by gathering several scientific journals from several leading platforms like PubMed and Scopus. As common accidents, skeletal muscle disease could be due to workplace and non-workplace causes. The highest risk occurs in the athlete and military environment. The treatment of current skeletal muscle injuries is protection, rest, ice, compression, and elevation (PRICE), non-steroidal anti-inflammatory drugs (NSAIDs), and mechanical stimulation. However, it is considered less effective, especially in NSAIDs, inhibiting myogenic cell proliferation. The current finding indicates that the stem cells have markers known as Pax3/Pax7. The role of both markers in muscle injury, Pax3/Pax7, as transcription factors will induce cell division by H3K4 methylation mechanisms and chromatin modifications that stimulate gene activation.
Conclusion
Regulation by Pax3/Pax7 factors that affect stem cells and stem cell proliferation is one of the alternative treatments. This regulation can accelerate the healing of injury victims, especially injuries to the skeletal muscles. Finally, after being compared, Pax3/Pax7 induces stem cells to have the potential to be one of the skeletal muscle injury treatments.
Keywords
Pax3 and Pax7, Pax3/Pax7, Skeletal muscle, Athlete, Stem cells, Cell proliferation, Injuries.
Similar content being viewed by others
Background
Skeletal muscle comprises about 40% of our weight and multinucleated contractile muscle strands [1, 2]. Skeletal muscle functions for the body to move and undertake activities, as well as maintain vital organs in our body [2, 3]. Therefore, it is easier to get injuries, especially for groups who conduct much physical activity, such as athletes and military personnel [4,5,6,7]. In a retrospective study, 65 of 79 gymnastic athletes experienced skeletal muscle injuries [8]. In addition, in the military environment, American soldiers in Afghanistan, as many as 35% of 15,000 soldiers experienced skeletal muscle injuries [9].
As the population is most commonly affected, skeletal muscle injuries can impact the productivity and performance of athletes and military personnel in conducting their duty. Hence, the recovery stage needs to be completed immediately and effectively [5]. Current therapies for skeletal muscle injuries are protection, rest, ice, compression, and elevation (PRICE or RICE) principles, pharmacological intervention such as non-steroidal anti-inflammatory drugs (NSAIDs), surgical treatment, and mechanical stimulation [10,11,12]. PRICE can stop intramuscular bleeding and muscle injuries [7, 13], while pharmacological intervention with NSAIDs can decrease the inflammatory cell reaction to recover the injury [14]. Likewise, surgery can be implemented in specific conditions and may also be considered if the patient has persistent pain for more than 4 to 6 months [13]. Finally, mechanical incitement can expand the recovery of the seriously harmed skeletal muscle, lessening the requirement for exogenous or cell development factors [15].
Despite these existing treatments, current therapies still have several drawbacks. For example, PRICE or RICE is still not clinically proven in randomised control trials (RCTs) studies; likewise, a long treatment period can increase bleeding [7]. When using higher doses of NSAIDs, many side effects may arise, and delayed muscle regeneration can be developed [7, 14]. Another therapy option with surgical treatment should be carried out under certain circumstances, and only the cost is quite expensive. A previous study stated that after surgery, patients could feel pain and irritation [7, 11]. When treating skeletal muscle injuries with mechanical stimulation, this treatment can enlarge the scar and continue the rupture [7]. Therefore, stem cell therapy can be another option to tackle these constraints.
Stem cell is one of the areas of interest in health and contributes to treating diseases. As an alternative treatment in skeletal muscle, stem cells have functional marker proteins in cell proliferation, particularly Pax3 and Pax7 (Pax3/Pax7). In one study, this protein strongly influences stem cell division [16, 17]. Hence, it is necessary to comprehend the function of the proteins themselves to medicate the disease.
Taking together those reasons, the purpose of this review is in its application, and it should be noted that these proteins can be used as an alternative treatment for skeletal muscle injuries. We will explain the mechanism and value of this protein innovation on gene regeneration and compare it with previous treatments.
Main text
The cause of skeletal muscle injuries
Skeletal muscle fibres originate from bone and connective tissue inserted through tendons (Fig. 1) [13]. Skeletal muscle comprises two fundamental parts, myofibres and connective tissue [11, 14]. Skeletal muscle functions for the body to move and perform activities, as well as maintain vital organs in our body [2, 3]. Hence, the skeletal muscles are essential to body parts [18].
Skeletal muscle injuries are common traumas in many populations, especially in sports and the military [4, 5, 13, 14]. This type of injury usually involves various severity and events, both direct and indirect trauma [1]. In the sports population, the incidence varies from 10 to 50% [14], while in the military, another study mentioned that non-battle injury could reach 77% of the 3300 American soldiers [19]. Of the two populations, the body parts that suffered the most injuries were the low back (17%), knee (12%), shoulder (10%), and most of the time when working (65%) [5].
Several factors that cause skeletal muscle injuries are sprains, bruises, and lacerations. Furthermore, more than 90% of incidents associated with sports are bruises and sprains [13]. The causes of skeletal muscle injuries are divided into two factors, particularly workplace and non-workplace factors. Workplace factors such as climbing, lifting, and overwork are associated with biomechanical risk factors, individual predispositions, and psychosocial conditions [20], while non-workplace factors such as physical stress and even comorbidities cause skeletal muscle injuries [6, 20]. Among athletes and the military population, these causes of skeletal muscle injuries are divided into two major factors, in particular intrinsic factors, such as age, sex, anatomy, and physical activity, as well as extrinsic factors, such as the amount of training, the type of training, and the acceleration of training [5, 6]. Meanwhile, there are more causes of skeletal muscle injuries from retrospective studies summarised in Table 1.
Some factors are known to cause skeletal muscle injuries, for example, the presence of viral pathogens. Recently, the SARS-CoV-2 virus can cause serious injury to skeletal muscle by downregulating angiotensin-converting enzyme 2 (ACE-2) receptors, increasing angiotensin (Ang) II levels, and reducing Ang 1–7. The SARS-CoV-2 virus can cause muscle damage when these levels drop. Another viral mechanism leading to skeletal muscle injuries is inducing inflammation in skeletal muscle. A previous study by Lentscher et al. created an engineered Chikungunya virus (CHIK) strain that exhibits in-cell replication via the miR-206 target sequence. They found that significantly reduced inflammation and necrosis in muscles arose when CHIKV replication in skeletal muscle cells was restricted. Another study by Gavino-Leopoldino et al. created a mouse model of Zika virus (ZIKV), and its infection induces necrotic lesions, inflammation, and fibre atrophy [21,22,23,24,25].
Another cause of muscle injuries, bacterial infections, such as Clostridium perfringens bacteria, can exacerbate skeletal muscle injuries, leading to myonecrosis due to the inflammatory factor IFN-γ. Zúñiga-Pereira et al., in their study, characterised the process of muscle regeneration after myonecrosis in experiments on murine infected with C. perfringens. This bacterial pathogenicity causes reduced muscle regenerative activity [26]. Finally, chemicals such as oxime can cause acute skeletal muscle injury by necrotising and performing inflammatory mechanisms. In one study, oxime was tested on mice leading to changes in the morphology. Muscle degeneration occurred when given treatment with a dose of 0.5 LD50, and acute toxic muscle injury developed in 7 days, resulting in necrosis [27].
Current treatment for skeletal muscle injuries and their limitations
Skeletal muscle injury is the most common injury which goes through three phases: the acute inflammatory and degenerative phase, the repair phase, and the remodelling phase. Current conservative treatments include protection, rest, ice, compression, and elevation (PRICE), nonsteroidal anti-inflammatory drugs (NSAIDs), and physical therapy [7]. It turns out that NSAIDs are used to suppress the inflammatory phase in injury. Other therapies and strategies are growth factors, stem cells, scaffolds, anti-fibrotic therapy, and mechanical stimulation [10]. In several journals, it is stated that, in athletes and military populations, antioxidants are used to prevent necrosis and degeneration, growth factors to promote muscle healing, and NSAIDs to reduce inflammation [4, 28].
RICE or PRICE
RICE or PRICE or protection, rest, ice, compression, and elevation is a mechanical treatment to minimise the injury at the injury site and prevent the extension of trauma to adjacent muscle areas [11, 13]. This method is a traditional method and is not widely known, but it can still be used and is not overly threatening for injured muscles. This method begins with resting the wound for 7 days and physiotherapy, respectively [7]. Nevertheless, PRICE is still not clinically proven in RCT studies, while a long treatment period can increase bleeding [7].
NSAIDs
NSAIDs can interfere with the activity of macrophages and breaking point phagocytic, in any event, hindering the creation of development inciting factors liable for recovery after muscle injury [7]. This drug has become an option for reducing pain after an injury. However, the use of NSAIDs stays dubious for their utilisation in the treatment of muscle injury [4]. NSAIDs can lead to incomplete recovery and, when continuously used, can affect muscle regeneration and contractile function [11, 29]. Therefore, this pharmacological treatment can potentially inhibit muscle regeneration in long-term consumption [7].
Mechanical stimulation
This treatment is the simplest method for stimulating mechanics. Mechanical strength is a biological regulator as crucial as chemicals and genes and has excellent potential for the development of mechano-therapy to treat muscle damage [15]. In an in vitro study, the use of this method works in muscle growth, differentiation, and muscle function, as well as in controlled stimulation to increase myogenesis [30]. Furthermore, mechanical stimulation aids in enhancing cellular alignment and inducing the formation and growth of radial muscular fibres, mimicking the fibre orientation in a healthy setting [31,32,33,34].
Pax3/Pax7 as an alternative therapy for skeletal muscle injury
A surrogate answer to recuperating skeletal muscle wounds is cell treatment since skeletal muscle is an optimal objective for targeting cell treatment [29]. The purpose of cell therapy is to work on the structure and capacity of muscle tissue [11]. The cells used are stem cells due to their strong dividing ability [35, 36].
Stem cells are characterised by the capability to expand and can be differentiated into various cell and tissue sources [16]. Stem cells are differentiated into a few characterisations dependent on differentiation potential, one of which is unipotent cells, which are cells that can only be differentiated into one specific cell type, such as muscle cells [16]. Based on the source, adult stem cells are unipotent. Thus, the skeletal muscle stem cells in adult bodies have become satellite cells [37].
Skeletal muscle regeneration requires important cells to regenerate, for instance, satellite cells and skeletal stem cells [38]. The candidate cells are myogenic cells and non-myogenic cells [11]. The myogenic stem cell method occurs when muscle-derivate stem cells (MDSCs) are transplanted, forming myofibres grafted onto the damaged area [11, 35]. Different types of cells do not consistently have similar differentiation abilities. The use of other stem cells can also be used with non-myogenic cell methods, such as using mesenchymal stem cells (MSCs) or adipose-derived stem cells (ADSCs) [39, 40].
Satellite cell-dependent myogenesis is a complex process characterised by the muscle-specific transcription factor Pax3/Pax7 from MyoD members [36]. Pax genes, especially Pax7, are significant in muscle development [41, 42]. When skeletal muscle is injured, the intervention of Pax3/Pax7 genes will continue to increase and begin to regulate cell division to cover the injury [41, 43]. The combination of several transcription factors with Pax3/Pax7 can induce skeletal muscle stem cells by activating transcription the cell proliferation [44, 45].
The mechanism of action of Pax3/Pax7 on skeletal muscle stem cell
Cellular mechanism of Pax3/Pax7 in cell proliferation: regulation of gene expression
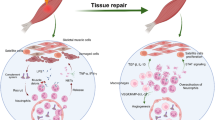
Generally, skeletal muscle regeneration is divided into three stages, in particular, an inflammatory phase caused by muscle damage; satellite cell activation due to proliferation; and the maturation phase of differentiation, the process of forming new myofibres. In the first stage, muscle cells will degenerate due to damage. Inflammation goes hand in hand with the immune response due to muscle tissue damage. Inflammation begins with the innate immune system response, induced by damage-associated molecular patterns (DAMPs) released by necrotic cells. DAMPs activate immune cells, such as mast cells, monocytes, and macrophage cells. These cells release pro-inflammatory cytokines and chemokines, causing activation of T-lymphocytes, either T helper (Th) or cytotoxic T cells, and circulating these lymphocytes to the injury site. Activated Th cells release inflammatory factors, causing vasodilation such as interferon γ (IFN-γ). Cytotoxic T cells can induce tissue damage, aggravating muscle injury [37, 46]. In the second stage, satellite cells are described by Pax7 articulation but do not express myogenic differentiation (MyoD). However, in a state of injury, this stage is done to prioritise entry into the cell cycle, begin to express MyoD and relocate to the injury site, and combine and become ancestor myogenic cells [38]. The third stage is maturation into myofibres which become the contractile units of skeletal muscle.
In general, satellite cells are the primary support in the recovery of skeletal muscle cells [38]. Pax3/Pax7 is conveyed by the satellite cells and directed during myogenic separation. The existence of Pax3/Pax7 helps myogenic factor 5 (Myf5) and has a positive–negative function in cell proliferation [47]. Pax3/Pax7, together with Myf5, controls the input when starting the cell programme. If these three genes are not present, cell differentiation will not exist. Pax3/Pax7 assume an equal part in MyoD initiation, resulting in skeletal muscle separation [41]. Finally, Pax3/Pax7 can control the occurrence of division and inhibit cell differentiation (Fig. 2) [41, 48].
The mechanism of action of Pax3/Pax7 for skeletal muscle stem cells is divided into two mechanisms; cellular and molecular mechanisms. A Cellularly, the mechanism consists of six stages: (1) muscle damage, (2) proliferation, (3) differentiation, (4) fusion, (5) formation, and (6) myofibre repair. B Molecularly, the mechanism is divided into several stages: (1) histone modification and (2) proliferation activation against the recruitment of a histone methyltransferase complex (HMT)
Molecular mechanism of Pax3/Pax7 on skeletal muscle stem cell
Transcription factors in myogenic genes
Pax3/Pax7 are the transcription factors of the specified muscle cells characterised. During the undeveloped turn of events, the transcription factors Pax3/Pax7 are viewed as significant controllers in the detail of muscle and tissue-framing cells [36]. However, in grown-up satellite cells, Pax7 is deeply overexpressed, and Pax3 is poorly expressed in the muscle area [36].
As explained above, the muscle regeneration process in stage two activates satellite cells. The transcription factor is viewed as a marker in fixed satellite cells, and Myf5 is the objective of transcription factor Pax7 once initiated. Activated satellite cells assert Pax7 and Myf5 and enter a proliferative state because Pax7 induces histone modification that stimulates transcription activation to regulate myogenic development [17, 36].
Proliferation activation against the recruitment of a histone methyltransferase complex (HMT)
In general, Pax7 specifically binds to the methylation regulatory element H3K4 in the target gene leading to the creation of the HMT core complex, for example, the strong trimethylation of H3K4 from the regulatory element, thereby activating subsequent gene expression. Another circumstance shows communication between Wdr5-Ash2L and Pax7 with the association of Pax7 with the HMT complex. Pax7 induces satellite cells by recruiting the HMT complex onto Myf5 by generating transcription activation [17]. The Pax3/Pax7 binding protein (Pax3/Pax7 BP) links this binding, so HMT-bound Pax7 is dependent on Pax3/Pax7 BP [49]. Thus, Pax7 can induce chromatin modification that stimulates the activation of target genes so that cells enter into the myogenic development programme [17].
The role of Pax3/Pax7 in hypoxia
During normoxia, Pax7 is downregulated by microRNA1 and 206 (miR1 and miR206), leading to decreased self-renewal of the satellite cells (Fig. 3A). While in hypoxia, the expression of miR1 & miR206 will be reduced, leading to the upregulation of asymmetric self-renewal division of satellite cells by Pax 7. This process mainly occurs due to the activation of the NOTCH signalling pathway (Fig. 3B). Hypoxic conditions can stimulate cell division in human primary myoblasts by increasing the expression of myogenic transcription factors Myf5 and MyoD1 [50]. Not only escalate the level of Myf-5 and MyoD1, but hypoxia also increases the level of myogenin which play a pivotal role in cell differentiation. The level rise occurs because hypoxia can induce H2K27 methylation and activate hypoxia-inducible factor 1-alpha (HIF1A) so that myogenesis factor expression is reduced [50, 51].
The role of Pax7 appears when its levels increase in hypoxia conditions. Pax7 will initiate a satellite cell asymmetric self-renewal programme and a cell proliferation [50, 52]. This mechanism aligns with the previous study, which indicated that 48 h under hypoxia, proliferation, and motility of skeletal muscle stem cell was enhanced. The number of undifferentiated cells positive for Pax 7 was increased at 72 h after hypoxia [52].
In vitro and in vivo studies of Pax3/Pax7
Previous in vitro and in vivo studies have been directed to acquire the mechanical properties of Pax3/Pax7. For example, one in vitro study using primary myoblast cells said that the prevailing job of Pax7 is because of high homeobox partiality in grown-up myogenesis. Moreover, one of the in vivo studies using mice was injected with mutant embryonic stem cells (ESCs) that lost the Pax7 gene and said that the initiation of myogenic differentiation is facilitated while Pax7 is absent. Those studies are described and summarised in Table 2.
Despite in vitro and in vivo studies previously conducted, clinical studies that mention Pax3/Pax7 testing in skeletal muscle injuries are not yet available on related websites such as https://www.cochranelibrary.com/central/about-central and https://clinicaltrials.gov/.
Conclusion
Stem cells characterised by Pax3/Pax7 proteins have great potential to become one the injury healing targets; drug substituents can be developed to target these marker proteins. This potential treatment could be seen from the ability of Pax3/Pax7 as transcription factors in stem cells to produce cell division proteins. The comparison between one study to another study indicates the mutant mice modified by the lacking Pax7 (Pax7−/−) were shown to decrease the growth and differentiation of skeletal muscle cells, leading to a lack of satellite cell function. These findings have a great potential to be developed as a new treatment; however, there are still limitations. The constraints of this study could be an opportunity for the state-of-the-art medication to treat skeletal muscle injuries. In addition, gene-edited test animals are needed to test their potency.
The comparison between current treatment (RICE or PRICE principles, NSAIDs, surgery, and mechanical stimulation) with alternative solutions with stem cells induced by Pax3/Pax7 resulted that the potential for stem cells induced by Pax3/Pax7 is very likely to be one of the treatments of skeletal muscle injuries.
Furthermore, it still needs to be studied further through in vitro and in vivo studies regarding the activity of Pax3/Pax7 that induce stem cells and the potential of these proteins for the treatment of skeletal muscle injuries. Assessment is also needed to find the best formulation for this treatment preparation. After studies in vitro and in vivo, it is hoped that this treatment will be developed into a clinical study and solve the current obstacle to treating athletes and military personnel who have a high incidence of skeletal muscle injury.
Availability of data and materials
There is no availability of data and materials.
Abbreviations
- ADSCs:
-
Adipose-derived stem cells
- ACE-2:
-
Angiotensin-converting enzyme 2
- Ang:
-
Angiotensin
- CHIK:
-
Chikungunya virus
- DAMPs:
-
Damage-associated molecular patterns
- ESCs:
-
Embryonic stem cells
- HIF1A:
-
Hypoxia-inducible factor 1-alpha
- HMT:
-
Histone methyltransferase complex
- IFN-γ:
-
Interferon γ
- LD50:
-
Lethal dose of 50
- MDSCs:
-
Muscle-derivated suppressor cells
- miR-1:
-
MicroRNA 1
- mir-206:
-
MicroRNA 206
- MSCs:
-
Mesenchymal stem cells
- MuCSs:
-
Muscle stem cells
- Myf5:
-
Myogenic factor 5
- MyoD:
-
Myogenic differentiation
- NSAIDs:
-
Nonsteroidal anti-inflammatory drugs
- Pax3:
-
Paired box 3
- Pax7:
-
Paired box 7
- Pax3/Pax7:
-
Paired box 3 and Paired box 7
- Pax3/Pax7BP:
-
Pax3/Pax7 binding protein
- PRICE:
-
Protection, rest, ice, compression, and elevation
- TCDD:
-
2,3,7,8-tetrachlorodibenzo-p-dioxin
- Wdr5:
-
WD repeat domain 5
- ZIKV:
-
Zika virus
References
Laumonier T, Menetrey J (2016) Muscle injuries and strategies for improving their repair. J Exp Orthop 3(1):1–9. https://doi.org/10.1186/s40634-016-0051-7 (Springer Berlin Heidelberg)
Trovato FM, Imbesi R, Conway N, Castrogiovanni P (2016) Morphological and functional aspects of human skeletal muscle. J Funct Morphol. 1(3):289–302. https://doi.org/10.3390/JFMK1030289 (Multidisciplinary Digital Publishing Institute)
Dave HD, Shook M, Varacallo M (2020) Anatomy. StatPearls, StatPearls Publishing, Skeletal Muscle
Gates C, Huard J (2005) Management of skeletal muscle injuries in military personnel. Operative Techniques in Sports Medicine 13:247–256. https://doi.org/10.1053/j.otsm.2006.01.012
Owens BD, Cameron KL (2016) Musculoskeletal injuries in the military. Springer, New York. https://doi.org/10.1007/978-1-4939-2984-9
Andersen KA, Grimshaw PN, Kelso RM, Bentley DJ (2016) Musculoskeletal lower limb injury risk in army populations. Sports Med Open 2:22. https://doi.org/10.1186/s40798-016-0046-z
Baoge L, Van Den Steen E, Rimbaut S, Philips N, Witvrouw E, Almqvist KF (2012) Treatment of skeletal muscle injury: a review. ISRN ISRN Orthop 2012:1–7. https://doi.org/10.5402/2012/689012
Roy TC, Knapik JJ, Ritland BM, Murphy N, Sharp MA (2012) Risk factors for musculoskeletal injuries for soldiers deployed to Afghanistan. Aviat Space Environ Med 83:1060–1066. https://doi.org/10.3357/ASEM.3341.2012
Giacomo F, Francesco F, Alessandra Z, Francesco M, Alessia M, Francesco PB et al (2021) Musculoskeletal pain in gymnasts: a retrospective analysis on a cohort of professional athletes. Int J Environ Res Public Health 18:5460. https://doi.org/10.3390/IJERPH18105460
Laumonier T, Menetrey J (2016) Muscle injuries and strategies for improving their repair. J Exp Orthop 3(1):9. https://doi.org/10.1186/S40634-016-0051-7 (SpringerOpen)
Qazi TH, Duda GN, Ort MJ, Perka C, Geissler S, Winkler T (2019) Cell therapy to improve regeneration of skeletal muscle injuries. J Cachexia Sarcopenia Muscle 10:501–516. https://doi.org/10.1002/JCSM.12416
Garg K, Corona BT, Walters TJ (2015) Therapeutic strategies for preventing skeletal muscle fibrosis after injury. Front Pharmacol 6:87. https://doi.org/10.3389/FPHAR.2015.00087
Fernandes TL, Pedrinelli A, Hernandez AJ (2011) Muscle injury – physiopathology, diagnosis, treatment and clinical presentation. Rev Bras Ortop 46:247–255. https://doi.org/10.1016/s2255-4971(15)30190-7
Järvinen TAH, Järvinen TLN, Kääriäinen M, Kalimo H, Järvinen M (2005) Muscle injuries: biology and treatment. Am J Sports Med 33:745–764. https://doi.org/10.1177/0363546505274714
Cezar CA, Roche ET, Vandenburgh HH, Duda GN, Walsh J, Mooney DJ (2016) Biologic-free mechanically induced muscle regeneration. Proc Natl Acad Sci U S A 113(6):1534–9. https://doi.org/10.1073/pnas.1517517113
Kolios G, Moodley Y (2012) Introduction to stem cells and regenerative medicine. Respiration. 85(1):3–10. https://doi.org/10.1159/000345615
Mckinnell IW, Ishibashi J, Le Grand F, Punch VGJ, Addicks GC, Greenblatt JF (2008) Pax7 activates myogenic genes by recruitment of a histone methyltransferase complex. Nat Cell Biol 10:77–84. https://doi.org/10.1038/ncb1671
Gusetu G, Pop D, Pop-Mindru D, Zdrenghea D (2015) Exercise NT-pro-BNP and global longitudinal strain in systemic lupus erythematosus patients. Eur J Prev Cardiol 22:S135. https://doi.org/10.5281/zenodo.1069104.Funding
Molloy JM, Pendergrass TL, Lee IE, Chervak MC, Hauret KG, Rhon DI (2020) Musculoskeletal injuries and United States army readiness part I: overview of injuries and their strategic impact. Mil Med 185:E1461–E1471. https://doi.org/10.1093/milmed/usaa027
Barbe MF, Barr AE (2006) Inflammation and the pathophysiology of work-related musculoskeletal disorders. Brain Behav Immun 20(5):423–429. https://doi.org/10.1016/j.bbi.2006.03.001
Lentscher AJ, McCarthy MK, May NA, Davenport BJ, Montgomery SA, Raghunathan K (2020) Chikungunya virus replication in skeletal muscle cells is required for disease development. J Clin Invest 130:1466–1478. https://doi.org/10.1172/JCI129893
Gavino-Leopoldino D, Figueiredo CM, da Silva MOL, Barcellos LG, Neris RLS, Pinto LDM (2021) Skeletal muscle is an early site of Zika Virus replication and injury, which impairs myogenesis. J Virol 95(22):e0090421. https://doi.org/10.1128/JVI.00904-21
De Giorgio MR, Di Noia S, Morciano C, Conte D (2020) The impact of SARS-CoV-2 on skeletal muscles. Acta Myol. 39(4):307. https://doi.org/10.36185/2532-1900-034
Seixas MLGA, Mitre LP, Shams S, Lanzuolo GB, Bartolomeo CS, Silva EA (2022) Unraveling Muscle Impairment Associated With COVID-19 and the Role of 3D Culture in Its Investigation. Front Nutr. 9:115. https://doi.org/10.3389/FNUT.2022.825629/BIBTEX
Sciorati C, Rigamonti E, Manfredi AA, Rovere-Querini P (2016) Cell death, clearance and immunity in the skeletal muscle. Cell Death Differ 23:927–937. https://doi.org/10.1038/cdd.2015.171
Zúñiga-Pereira AM, Santamaría C, Gutierrez JM, Alape-Girón A, Flores-Díaz M (2019) Deficient skeletal muscle regeneration after injury induced by a clostridium perfringens strain associated with gas gangrene. Infect Immun. 87(8):e00200-19. https://doi.org/10.1128/IAI.00200-19
Jaćević V, Nepovimova E, Kuča K (2019) Toxic injury to muscle tissue of rats following acute oximes exposure. Sci Rep 9:1–13. https://doi.org/10.1038/s41598-018-37837-4
Sauers SE, Smith LB, Scofield DE, Cooper A, Warr BJ (2016) Self-management of unreported musculoskeletal injuries in a U.S Army brigade. Mil Med 181:1075–1080. https://doi.org/10.7205/MILMED-D-15-00233
Judson RN, Rossi FMV (2020) Towards stem cell therapies for skeletal muscle repair. Npj Regen Med 5:1–6. https://doi.org/10.1038/s41536-020-0094-3
Rangarajan S, Madden L, Bursac N (2014) Use of flow, electrical, and mechanical stimulation to promote engineering of striated muscles. Ann Biomed Eng 47(7):1391–405. https://doi.org/10.1007/s10439-013-0966-4
Soltow QA, Zeanah EH, Lira VA, Criswell DS (2013) Cessation of cyclic stretch induces atrophy of C2C12 myotubes. Biochem Biophys Res Commun 434:316–321. https://doi.org/10.1016/J.BBRC.2013.03.048
Niklason LE, Gao J, Abbott WM, Hirschi KK, Houser S, Marini R (1999) Functional Arteries Grown in Vitro. Science 284:489–493. https://doi.org/10.1126/SCIENCE.284.5413.489
Dahl SLM, Kypson AP, Lawson JH, Blum JL, Strader JT, Li Y (2011) Readily available tissue-engineered vascular grafts. Sci Transl Med 3(68):68ra9. https://doi.org/10.1126/SCITRANSLMED.3001426
Todros S, Spadoni S, Maghin E, Piccoli M, Pavan PG (2021) A Novel Bioreactor for the Mechanical Stimulation of Clinically Relevant Scaffolds for Muscle Tissue Engineering Purposes. Processes 2021, Vol 9, Page 474, Multidisciplinary Digital Publishing Institute. 9, 474. https://doi.org/10.3390/PR9030474
Maclean S, Khan WS, Malik AA, Anand S, Snow M (2012) The potential of stem cells in the treatment of skeletal muscle injury and disease. Stem Cells Int 2012:282348. https://doi.org/10.1155/2012/282348
Segalés J, Perdiguero E, Muñoz-Cánoves P (2015) Epigenetic control of adult skeletal muscle stem cell functions. FEBS J 282:1571–1588. https://doi.org/10.1111/febs.13065
Yin H, Price F, Rudnicki MA (2013) Satellite cells and the muscle stem cell niche. Physiol Rev 93:23–67. https://doi.org/10.1152/physrev.00043.2011
Schmidt M, Schüler SC, Hüttner SS, Von Eyss B, Von Maltzahn J (2019) Adult stem cells at work: regenerating skeletal muscle. Cell Mol Life Sci 76(13):2559–2570. https://doi.org/10.1007/s00018-019-03093-6
Gorecka A, Salemi S, Haralampieva D, Moalli F, Stroka D, Candinas D (2018) Autologous transplantation of adipose-derived stem cells improves functional recovery of skeletal muscle without direct participation in new myofiber formation. Stem Cell Res Ther 9:1–12. https://doi.org/10.1186/S13287-018-0922-1
Qazi TH, Duda GN, Ort MJ, Perka C, Geissler S, Winkler T (2019) Cell therapy to improve regeneration of skeletal muscle injuries. J Cachexia Sarcopenia Muscle 10:501–516. https://doi.org/10.1002/jcsm.12416
Relaix F, Montarras D, Zaffran S, Gayraud-Morel B, Rocancourt D, Tajbakhsh S (2006) Pax3 and Pax7 have distinct and overlapping functions in adult muscle progenitor cells. J Cell Biol 172:91–102. https://doi.org/10.1083/jcb.200508044
Buckingham M, Relaix F (2015) PAX3 and PAX7 as upstream regulators of myogenesis. Semin Cell Dev Biol 44:115–125. https://doi.org/10.1016/j.semcdb.2015.09.017
Wang M, Song W, Jin C, Huang K, Yu Q, Qi J (2021) Pax3 and Pax7 exhibit distinct and overlapping functions in marking muscle satellite cells and muscle repair in a marine teleost, sebastes schlegelii. Int J Mol Sci. 22(7):3769. https://doi.org/10.3390/IJMS22073769
Sato T, Higashioka K, Sakurai H, Yamamoto T, Goshima N, Ueno M (2019) Core transcription factors promote induction of PAX3-positive skeletal muscle stem cells. Stem Cell Reports 13:352–365. https://doi.org/10.1016/J.STEMCR.2019.06.006
Buckingham M (2007) Skeletal muscle progenitor cells and the role of Pax genes. C R Biol 330:530–533. https://doi.org/10.1016/J.CRVI.2007.03.015
Wilkinson HN, Hardman MJ (2020) Wound healing: cellular mechanisms and pathological outcomes. Open Biology, The Royal Society. 10. https://doi.org/10.1098/RSOB.200223
Collins CA, Gnocchi VF, White RB, Boldrin L, Perez-Ruiz A, Relaix F (2009) Integrated functions of Pax3 and Pax7 in the regulation of proliferation, cell size and myogenic differentiation. PLoS ONE 4:e4475. https://doi.org/10.1371/journal.pone.0004475
Soleimani VD, Punch VG, Kawabe YI, Jones AE, Palidwor GA, Porter CJ (2012) Transcriptional dominance of Pax7 in adult myogenesis is due to high-affinity recognition of homeodomain motifs. Dev Cell 22:1208–1220. https://doi.org/10.1016/j.devcel.2012.03.014
Diao Y, Guo X, Li Y, Sun K, Lu L, Jiang L (2012) Pax3/7BP is a Pax7- and Pax3-binding protein that regulates the proliferation of muscle precursor cells by an epigenetic mechanism. Cell Stem Cell 11:231–241. https://doi.org/10.1016/j.stem.2012.05.022
Pircher T, Wackerhage H, Aszodi A, Kammerlander C, Böcker W, Saller MM (2021) Hypoxic signaling in skeletal muscle maintenance and regeneration: a systematic review. Front Physiol. 12:912. https://doi.org/10.3389/FPHYS.2021.684899/BIBTEX
Zhang Z, Zhang L, Zhou Y, Li L, Zhao J, Qin W (2019) Increase in HDAC9 suppresses myoblast differentiation via epigenetic regulation of autophagy in hypoxia. cell death & disease 2019 10:8. Nat Publ Group 10:1–15. https://doi.org/10.1038/s41419-019-1763-2
Elashry MI, Kinde M, Klymiuk MC, Eldaey A, Wenisch S, Arnhold S (2022) The effect of hypoxia on myogenic differentiation and multipotency of the skeletal muscle-derived stem cells in mice. Stem Cell Res Ther 13:1–17. https://doi.org/10.1186/S13287-022-02730-5/FIGURES/5
Lilja KC, Zhang N, Magli A, Gunduz V, Bowman CJ, Arpke RW (2017) Pax7 remodels the chromatin landscape in skeletal muscle stem cells. PLoS One. 12(4):e0176190. https://doi.org/10.1371/journal.pone.0176190
Florkowska A, Meszka I, Zawada M, Legutko D, Legutko D, Proszynski TJ (2020) Pax7 as molecular switch regulating early and advanced stages of myogenic mouse ESC differentiation in teratomas. Stem Cell Res Ther 11:1–18. https://doi.org/10.1186/s13287-020-01742-3
Zalc A, Rattenbach R, Auradé F, Cadot B, Relaix F (2015) Pax3 and Pax7 play essential safeguard functions against environmental stress-induced birth defects. Dev Cell 33(1):56–66. https://doi.org/10.1016/j.devcel.2015.02.006
Kuang S, Chargé SB, Seale P, Huh M, Rudnicki MA (2006) Distinct roles for Pax7 and Pax3 in adult regenerative myogenesis. J Cell Biol 172:103–113. https://doi.org/10.1083/jcb.200508001
Crist CG, Montarras D, Pallafacchina G, Rocancourt D, Cumano A, Conway SJ (2009) Muscle stem cell behavior is modified by microRNA-27 regulation of Pax3 expression. Proc Natl Acad Sci U S A. 106(32):13383–7. https://doi.org/10.1073/pnas.0900210106
Kassar-Duchossoy L, Giacone E, Gayraud-Morel B, Jory A, Gomès D, Tajbakhsh S (2005) Pax3/Pax7 mark a novel population of primitive myogenic cells during development. Genes Dev 19:1426–1431. https://doi.org/10.1101/GAD.345505
Acknowledgements
The authors are thankful to the faculty.
Funding
No funding was obtained to draft this review article.
Author information
Authors and Affiliations
Contributions
MA gathered information; MA, BWKW, and ER compiled and drafted the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Azhar, M., Wardhani, B.W.K. & Renesteen, E. The regenerative potential of Pax3/Pax7 on skeletal muscle injury. J Genet Eng Biotechnol 20, 143 (2022). https://doi.org/10.1186/s43141-022-00429-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s43141-022-00429-x