Abstract
Purpose
To verify if the bark of Passiflora edulis prevents obesity-related disorders and oxidative stress in db/db mice.
Methods
Obese male db/db mice (n = 14 animals) were randomly divided into two groups to receive standard chow and water (obese, n = 7 (OB)) or standard chow with bark of Passiflora edulis (BPe) (obese + BPe, n = 7 (OB + BPe)) for 16 weeks. The evaluated parameters in animals included food and caloric intake, body weight, total body fat and fat deposits, serum glucose, triglycerides, and total cholesterol. Malondialdehyde (MDA) and antioxidant capacity were evaluated in serum and organs (adipose tissue, kidney, liver, and heart). All groups were compared by Student t test, with p < 0.05.
Results
The results showed the benefits from BPe by preventing abdominal fat deposition and by reducing the total cholesterol. Moreover, the compound increased the antioxidant capacity from the organs analyzed and reduced the MDA levels in the liver.
Conclusion
It is possible to conclude that the consumption of the BPe prevents obesity-related disorders and oxidative stress in db/db mice.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The prevalence of obesity and chronic diseases, such as hypertension, hyperglycemia, and dyslipidemia, has increased around the world. The literature reports that fat accumulation, especially abdominal obesity, is related to the development of obesity-related disorders [1,2,3,4,5,6]. Several mechanisms try to explain the association between obesity and diseases, among them, higher adiposity, which would induce a redox system imbalance, characterized by the excess of oxygen reactive species (ROS) [7]. This condition leads to oxidation of different molecules such as lipids, carbohydrates, proteins, and DNA, which are involved in the development of different disorders [8, 9].
Antioxidants are substances able to protect the organism against diseases by avoiding molecule oxidation [10, 11]. They are obtained from both endogenous and exogenous sources; however, the latter is considered the most important since the nutrients from the diet are indispensable for the endogenous antioxidant synthesis [12]. Based on this, the early introduction of fruits and vegetables in the diet, which are rich in antioxidants and bioactive compounds, could be an effective strategy to prevent or delay the obesity-related disorders [13,14,15].
Passiflora edulis, popularly known as “passion fruit” or “maracujá,” is a fruit that contains several antioxidants in the pulp, leaves, seeds, and bark, such as phenolics, carotenoids, vitamin C, and polyamines [16]. In a previous study published by our group [17], it was demonstrated that the treatment of obese db/db mice with the BPe reduced the body fat and improved metabolic and antioxidant parameters. However, there is a lack of studies regarding the preventive effect of the BPe. So, the aim of this study was to verify if the BPe prevents obesity-related disorders and oxidative stress in db/db mice.
Materials and methods
Animals and experimental protocol
In this study, db/db mice were used, which are animals genetically deficient for the leptin receptor, and considered by the literature an established model for obesity and type 2 diabetes [18]. All the animals were acquired from Universidade de São Paulo—USP, Brazil. After weaning (21 days of age), male db/db mice (n = 14 animals) were randomly divided into two groups to receive standard chow (Presence for rats and mice, Presence Nutrição Animal, Brazil) and water (obese, n = 7 animals (OB)) or standard chow with BPe ((obese + BPe, n = 7 animals (OB + BPe)) during 16 weeks.
For all the animals, chow and water were offered ad libitum. Feed and water consumption were measured daily, and body weight was measured weekly. Two animals per cage were kept in an environment with controlled temperature (24 ± 2 °C), humidity (55 ± 5%), and light-dark cycle (12–12 h). The study protocol was approved by the Ethics Committee on Animal Experimentation of the Botucatu Medical School, Universidade Estadual Paulista-UNESP, (1104/2017) in São Paulo, Brazil, and followed the recommendations of the Guide for the Care and Use of Experimental Animals [19]. At the end of the experiment, the animals were anesthetized with ketamine and xylazine and then euthanized by cardiac puncture, and the organs and blood were collected for analysis.
Preparation of chow with bark of Passiflora edulis
The Passiflora edulis fruits were obtained from a rural producer in Presidente Prudente city, São Paulo, Brazil, at the ripening stage and submitted to selection, washed, cut into small pieces, and oven-dried at 60 °C for 48 h. After this process, the dried barks were milled (Logen Scientific, Diadema São Paulo, Brazil) and added to grounded commercial chow (Presence, Paulínea, São Paulo, Brazil), to reach the proportion of 7 g of Passiflora edulis bark/kg of chow (correspondent to 1.5 g/kg of body weight per day) [20]. Following this, the mixture was pelleted again for consumption.
Nutritional parameters
In order to characterize the nutritional profile, the initial body weight (IBW), final body weight (FBW), total body fat (TF—sum of fat deposits: epididymal, retroperitoneal, visceral, and subcutaneous), adiposity index (AI—total body fat/final body weight × 100), and feed intake (g/day) were considered.
Metabolic parameters
After 8-h fasting, serum glucose, triglycerides, and cholesterol levels (kits from BioClin®, Belo Horizonte, MG, Brazil) were determined by an automatic enzymatic analyzer system (Chemistry Analyzer BS-200, Mindray Medical International Limited, Shenzhen, China).
Preparation of tissues for oxidative stress analysis
Increased reactive oxygen species (ROS) are able to oxidize biomolecules and to affect the antioxidant capacity, so it was analyzed in this study: MDA levels, an important lipid peroxidation biomarker, and the antioxidant capacity in the serum and tissues (adipose, hepatic, cardiac, and renal). Tissues (100 mg) were homogenized in 1 mL of cold phosphate saline buffer (PBS), pH = 7.4, and centrifuged (800×g, 4 °C, 10 min). The supernatant was used in the following analyses:
Malondialdehyde
One hundred microliters of the homogenate was used for malondialdehyde (MDA) analysis. Briefly, 700 μL of 1% orthophosphoric acid and 200 μL of thiobarbituric acid (42 mM) were added to the samples. Then, the mixture was boiled for 60 min in a water bath, and afterward, it was immediately cooled on ice. A total of 200 μL was transferred to a 2-mL tube containing 200-μL sodium hydroxide/methanol (1:12 v/v). The sample was vortex-mixed for 10 s and centrifuged for 3 min at 13,000g. The supernatant (200 μL) was transferred to a 300-μL glass vial and 50 μL was injected onto the column. The HPLC was a Shimadzu LC-10AD system (Kyoto, Japan) equipped with a C18 Luna column (5 μm, 150 × 4.60 mm, Phenomenex Inc., Torrance, CA, USA), a Shimadzu RF-535 fluorescence detector (excitation 525 nm, emission 551 nm), and 0.5 mL/min flow of phosphate buffer (KH2PO4 1 mM, pH 6.8) [10]. The MDA was quantified by the determination of the peak area in the chromatograms relative to a standard curve of known concentrations. A calibration curve was obtained by tetra-ethoxypropane (TEP) solutions [21].
Antioxidant capacity
The hydrophilic antioxidant capacity was determined fluorometrically as described by Beretta et al. [22] using a microplate reader (VICTOR X2, Perkin Elmer-Boston, MA, USA). The antioxidant capacity was quantified by the comparison between the areas under the curve relative to the oxidation kinetics of the phosphatidylcholine (PC) suspension, used as a reference for the biological matrix. The compound 2,2′-Azobis (2-aminopropano)-dihydrochloride (AAPH) was used as the peroxyl radical initiator. The results represent the percentage of the inhibition of 4,4-difluoro-5-(4-phenyl-1,3-butadienyl)-4-bora-3a,4a-diaza-s-indacene-3-undecanoic acid (BODIPY, 581/591) in serum relative to that occurring in the control sample of BODIPY 581/591 in PC liposome. All analyses were performed in triplicate, and the results represent the percentual of protection.
Statistical analysis
Data were analyzed by the Student t test or the Mann–Whitney and the results are presented as means ± standard deviation (SD) or medians (interquartile range). Statistical analyses were performed using Sigma Stat for Windows version 3.5. (Systat Software, Inc., San Jose, CA, USA), and a p value of 0.05 was considered as statistically significant.
Results
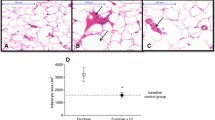
The nutritional parameters are presented in Table 1 and it is possible to verify that BPe was able to prevent the increase in both visceral and retroperitoneal adipose tissues. Moreover, the treated group (obese + BPe) showed lower total body fat and adiposity index than the obese group.
Figure 1 presents the serum biochemical parameters. It is possible to verify the positive effect of the BPe preventing the increase in the total cholesterol in the treated group. No effect was observed on glucose and triglycerides levels.
The antioxidant capacity in serum, liver, kidney, left ventricle, and adipose tissue was analyzed an is presented in Fig. 2. The BPe was able to increase the antioxidant capacity in serum, kidney, liver, and adipose tissue. Regarding the effect on the left ventricle, no change was observed.
Figure 3 presents the MDA levels in the kidney, liver, left ventricle, and adipose tissue. It is possible to verify a lower level of this marker in the liver. There was no effect on MDA levels in the kidney, left ventricle, and adipose tissue.
Discussion
The principal findings of this study are that BPe treatment conferred protection against obesity, reduced levels of total cholesterol, increased antioxidant defense, and reduced MDA in the liver. These are important findings since the BPe was effective to modulate the genome-wide expression profiling in a classic obesity experimental model [23].
The expansion of the adipose tissue, especially the visceral fat, is indicated as the central dogma regarding the physiopathology of obesity disorders [24]. Our results showed a positive effect from the BPe by preventing visceral and total fat accumulation in the treatment group. Studies show that natural bioactive compounds can prevent obesity development by the modulation of several pathways [25], among them the stimulation of the peroxisome proliferator–activated receptor gamma (PPAR-γ) synthesis. The increased levels of PPAR-γ induced by some bioactive compounds would be responsible for the adipose tissue browning, making it more metabolic and decreasing its lipid deposition [26]. Once the bark of Passiflora edulis is rich in carotenoids and flavonoids [17], the compound used in this study may be able to modulate some of these mechanisms involved in fat accumulation.
According to the literature, the reduction in body fat is responsible for significant metabolic benefits, as the prevention of insulin resistance, glucose intolerance, type 2 diabetes, and dyslipidemia [27]. In this study, the treated group presented only lower levels of total cholesterol compared with the non-treated group. This finding can be attributed to the pectin, a soluble fiber presented in the BPe, able to form gels and prevents cholesterol absorption [28]. No effect was observed in glucose level, which is an expected result once db/db mice are a genetic rodent model for type 2 diabetes [23]. The absence of effect on triglycerides levels can be related to insulin resistance. Increased secretion of triglyceride-enriched VLDL (very-low-density protein) is the commonest cause of elevated plasma triglycerides in insulin resistance and diabetes conditions [29].
The imbalance of the redox system is considered one of the main causes of obesity-related disorder development [30]. In the present study, the BPe increased both systemic and tissues antioxidant capacity, which can be attributed to the antioxidant activity from the BPe, already described in our previous study [17]. This property is an important finding that demonstrates the effectiveness to prevent diseases and modulates antioxidant response by the bioactive compounds present in discarded parts from foods.
One consequence of the oxidative stress is the lipid peroxidation, a free radical–mediated chain of reactions that result in an oxidative deterioration of polyunsaturated lipids, which are components of biological membranes and common targets of reactive species [31]. Lipid peroxidation results in MDA production, the most frequently used biomarker of the oxidative stress in many health problems such as cancer, chronic obstructive pulmonary disease, and cardiovascular diseases [32]. Our results showed that the BPe prevented lipid peroxidation (MDA production) only in the liver. There are several potential mechanisms that explain the increased lipid peroxidation in hepatic tissue. Higher levels of triglycerides are deposited inside the hepatocytes, which favors the occurrence of oxidative stress and the progression of steatosis to steatohepatitis and fibrosis. Reactive oxygen species and lipid peroxidation products impair the respiratory chain in hepatocytes, either directly or indirectly, exposing the mitochondrial genome to oxidative damage. These features, in turn, lead to the generation of more ROS, and a vicious cycle may ensue. All groups present higher levels of TG, and the BPe was able to prevent the hepatic lipid peroxidation [33].
The absence of effect on MDA in the other organs can be explained by the duration of the experiment. The occurrence of free radical production and consequently lipid peroxidation in the heart is associated with the elevated myocardial work and mechanical overload. In kidneys, the lipid peroxidation is responsible for the accumulation of adipose tissue around the kidneys of obese rats [33]. In adipose tissue, oxidative stress occurs especially in hypertrophied cells [34]. Probably the obese group did not develop a level of obesity which is able to lead to oxidative stress in all organs. However, it demonstrates that the liver is the primary organ affected by the reactive species in obesity condition and the BPe prevents the lipid peroxidation.
Conclusion
In summary, the results showed that the BPe is a good source of fiber and phytochemicals and have a wide variety of health benefits as a protection against obesity. In addition, it is noteworthy that BPe reduced the total cholesterol and increased the antioxidant capacity of treated animals. Based on these results, it is possible to conclude that the BPe consumption prevents obesity-related disorders and oxidative stress in db/db mice.
References
O’Neill S, O’Driscoll L. Metabolic syndrome: a closer look at the growing epidemic and its associated pathologies. Obes Rev. 2015;16:1–12. https://doi.org/10.1111/obr.12229.
Raimundo M, Lopes JA. Metabolic syndrome, chronic kidney disease , and cardiovascular disease : a dynamic and life-threatening triad. Cardiol Res Pract. 2011;2011:1–16. https://doi.org/10.4061/2011/747861.
Eva K, Panagiota P, Gregory K, George C. Metabolic syndrome: definitions and controversies. BMC Med. 2011;9:48.
Rasool S, Geetha T, Broderick TL, Babu JR, Lambert E. High fat with high sucrose diet leads to obesity and induces myodegeneration. 2018;9:1–10. https://doi.org/10.3389/fphys.2018.01054.
Warburton DER, Nicol CW, Bredin SSD. Health benefits of physical activity: the evidence. CMAJ. 2006;174(6):801–9. https://doi.org/10.1503/cmaj.051351.
Kopelman PG. Obesity as a medical problem. Nature. 2000;404:635–43.
Dludla P, Nkambule B, Jack B, Mkandla Z, Mutize T, Silvestri S, et al. Inflammation and oxidative stress in an obese state and the protective effects of gallic acid. Nutrients. 2018;11:23. https://doi.org/10.3390/nu11010023.
Hwang SW, Lee YM, Aldini G, Yeum KJ. Targeting reactive carbonyl species with natural sequestering agents. Molecules. 2016;21:1–13. https://doi.org/10.3390/molecules21030280.
Aldini G, Carini M, Colombo R, Rossi R, Milzani A. Protein carbonylation, cellular dysfunction, and disease progression. J Cell Mol Med. 2006;10:389–406.
Pierine DT, Navarro MEL, Minatel IO, Luvizotto RAM, Nascimento AF, Ferreira ALA, et al. Lycopene supplementation reduces TNF-α via RAGE in the kidney of obese rats. Nutr Diabetes. 2014;4:e142. https://doi.org/10.1038/nutd.2014.39.
Yeum K, Beretta G, Krinsky NI, et al. Synergistic interactions of antioxidant nutrients in a biological model system. Nutrition. 2009;25:839–46. https://doi.org/10.1016/j.nut.2009.01.011.
Kurutas EB. The importance of antioxidants which play the role in cellular response against oxidative/nitrosative stress: current state. Nutr J. 2016;15:1–22. https://doi.org/10.1186/s12937-016-0186-5.
Pérez-burillo S, Oliveras MJ, Quesada J, Ru JA, Pastoriza S. Relationship between composition and bioactivity of persimmon and kiwifruit. 2018;105:461–72. https://doi.org/10.1016/j.foodres.2017.11.022.
De Iglesia R, Loria-kohen V, Zulet MA, Martinez JA, Reglero G, De MAR. Dietary strategies implicated in the prevention and treatment of metabolic syndrome. 2016:1–21. https://doi.org/10.3390/ijms17111877.
Bahadoran Z, Mirmiran P, Azizi F. Potential efficacy of broccoli sprouts as a unique supplement for management of type 2 diabetes and its complications. J Med Food. 2013;16:1–8. https://doi.org/10.1089/jmf.2012.2559.
Noélia F, Vieira GP, Diniz B, Guedes F, Izabel M, Lima-neto ABM, et al. Antioxidant activity and physicochemical analysis of passion fruit (Passiflora glandulosa Cav.) pulp native to Cariri region. Acta Sci Biol Sci. 2017;39:417–22. https://doi.org/10.4025/actascibiolsci.v39i4.34045.
Panelli MF, Pierine DT, Borges L. Bark of Passiflora edulis treatment stimulates antioxidant capacity, and reduces dyslipidemia and body fat in db / db mice. Antioxidants. 2018;7:1–11. https://doi.org/10.3390/antiox7090120.
Wang B, Chandrasekera PC, Pippin JJ. Leptin- and leptin receptor-deficient rodent models: relevance for human type 2 diabetes. Curr Diabetes Rev. 2014;10:131–45. https://doi.org/10.2174/1573399810666140508121012.
Olfert ED, Cross BM, McWilliam AA. Guide to the care and use of experimental animals, vol. 1. Ottawa: CCAC; 1993.
Lima ES, Schwertz MC, Sobreira CRC, Borras MRL. Efeito hipoglicemiante da farinha do fruto de maracujá-do-mato (Passiflora nitida Kunth) em ratos normais e diabéticos. Rev Bras Plantas Med. 2012;14:383–8. https://doi.org/10.1590/S1516-05722012000200020.
Khoschsorur GA, Winklhofer-Roob BM, Rabl H, Auer T, Peng Z, Schaur RJ. Evaluation of a sensitive HPLC method for the determination of malondialdehyde, and application of the method to different biological materials. Chromatographia. 2000;52:181–4. https://doi.org/10.1007/BF02490453.
Beretta G, Aldini G, Facino RM, Russell RM, Krinsky NI, Yeum KJ. Total antioxidant performance: a validated fluorescence assay for the measurement of plasma oxidizability. Anal Biochem. 2006;354:290–8. https://doi.org/10.1016/j.ab.2006.04.001.
Belke DD, Severson DL. Diabetes in mice with monogenic obesity: the db/db mouse and its use in the study of cardiac consequences. Anim Model Diabetes Res. 2012:47–57. https://doi.org/10.1007/978-1-62703-068-7_4.
Aroor AR, DeMarco VG. Oxidative stress and obesity: the chicken or the egg? Diabetes. 2014;63:2216–8. https://doi.org/10.2337/db14-0424.
Nabavi SF, Russo GL, Daglia M, Nabavi SM. Role of quercetin as an alternative for obesity treatment: you are what you eat! Food Chem. 2015;179:305–10. https://doi.org/10.1016/j.foodchem.2015.02.006.
Pascual-Serrano A-A, Suárez-García BF, Suárez ALBC. Grape seed proanthocyanidin supplementation reduces adipocyte size and increases adipocyte number in obese rats. Int J Obes. 2017;41:1–10. https://doi.org/10.1038/ijo.2017.90.
Sumner AE, Sumner AE. The relationship of body fat to metabolic disease: influence of sex and ethnicity. Gend Med. 2008;5:361–71. https://doi.org/10.1016/S1550-8579(08)00108-3.
Jesch ED, Carr TP. Food ingredients that inhibit cholesterol absorption. Prev Nutr Food Sci. 2017;22:67–80. https://doi.org/10.3746/pnf.2017.22.2.67.
Malmström R, Packard CJ, Caslake M, Bedford D, Stewart P, Yki-Järvinen H, et al. Defective regulation of triglyceride metabolism by insulin in the liver in NIDDM. Diabetologia. 1997;40:454–62.
Francisqueti FV, Chiaverini LCT, Dos Santos KC, Minatel IO, Ronchi CB, Ferron AJT, et al. The role of oxidative stress on the pathophysiology of metabolic syndrome. Rev Assoc Med Bras. 2017;63:85–91. https://doi.org/10.1590/1806-9282.63.01.85.
Ayala A, Muñoz MF, Argüelles S. Lipid peroxidation: production, metabolism, and signaling mechanisms of malondialdehyde and 4-hydroxy-2-nonenal. Oxidative Med Cell Longev. 2014;2014:1–31. https://doi.org/10.1155/2014/360438.
Ansarin K, Khoubnasabjafari M, Jouyban A. Reliability of malondialdehyde as a biomarker of oxidative stress in psychological disorders 2015;5:123–127. doi:https://doi.org/10.15171/bi.2015.20.
Noeman SA, Hamooda HE, Baalash AA. Biochemical study of oxidative stress markers in the liver, kidney and heart of high fat diet induced obesity in rats. Diabetol Metab Syndr. 2011;3:1–8. https://doi.org/10.1186/1758-5996-3-17.
Manna P, Jain SK. Obesity, oxidative stress, adipose tissue dysfunction, and the associated health risks: causes and therapeutic strategies. Metab Syndr Relat Disord. 2015;13:423–44. https://doi.org/10.1089/met.2015.0095.
Funding
Fundação de Amparo à Pesquisa do Estado de São Paulo—FAPESP (2015/09292-0).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study protocol was approved by the Ethics Committee on Animal Experimentation of the Botucatu Medical School, Universidade Estadual Paulista-UNESP, (1104/2017) in São Paulo, Brazil, and followed the recommendations of the Guide for the Care and Use of Experimental Animals [19].
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Panelli, M.F., Garcia, J.L., de Souza, S.L.B. et al. Preventive effect of the bark of Passiflora edulis on obesity-related disorders and oxidative stress in db/db mice. Nutrire 44, 2 (2019). https://doi.org/10.1186/s41110-019-0101-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s41110-019-0101-x