Abstract
Background
Neural progenitors produce diverse cells in a stereotyped birth order, but can specify each cell type for only a limited duration. In the Drosophila embryo, neuroblasts (neural progenitors) specify multiple, distinct neurons by sequentially expressing a series of temporal identity transcription factors with each division. Hunchback (Hb), the first of the series, specifies early-born neuronal identity. Neuroblast competence to generate early-born neurons is terminated when the hb gene relocates to the neuroblast nuclear lamina, rendering it refractory to activation in descendent neurons. Mechanisms and trans-acting factors underlying this process are poorly understood. Here we identify Corto, an enhancer of Trithorax/Polycomb (ETP) protein, as a new regulator of neuroblast competence.
Methods
We used the GAL4/UAS system to drive persistent misexpression of Hb in neuroblast 7–1 (NB7-1), a model lineage for which the early competence window has been well characterized, to examine the role of Corto in neuroblast competence. We used immuno-DNA Fluorescence in situ hybridization (DNA FISH) in whole embryos to track the position of the hb gene locus specifically in neuroblasts across developmental time, comparing corto mutants to control embryos. Finally, we used immunostaining in whole embryos to examine Corto’s role in repression of Hb and a known target gene, Abdominal B (Abd-B).
Results
We found that in corto mutants, the hb gene relocation to the neuroblast nuclear lamina is delayed and the early competence window is extended. The delay in gene relocation occurs after hb transcription is already terminated in the neuroblast and is not due to prolonged transcriptional activity. Further, we find that Corto genetically interacts with Posterior Sex Combs (Psc), a core subunit of polycomb group complex 1 (PRC1), to terminate early competence. Loss of Corto does not result in derepression of Hb or its Hox target, Abd-B, specifically in neuroblasts.
Conclusions
These results show that in neuroblasts, Corto genetically interacts with PRC1 to regulate timing of nuclear architecture reorganization and support the model that distinct mechanisms of silencing are implemented in a step-wise fashion during development to regulate cell fate gene expression in neuronal progeny.
Similar content being viewed by others
Background
In both insects and mammals, neural progenitor cells generate diverse cell types in a stereotyped birth order [1,2,3,4,5,6,7]. Throughout development, progenitors progressively lose competence to generate early-born neural cell types as they gain competence to generate the late-born cell types, thereby establishing organized tissue growth. Thus, progenitors have a limited window of time during which they are able to specify each neural cell type [8,9,10,11,12]. How competence is regulated is critical to understanding both normal development and for effectively harnessing stem cells therapeutically, but mechanisms underlying competence regulation are not well understood.
Drosophila neural progenitors, called neuroblasts, are a tractable model system to investigate mechanisms of competence regulation. In the embryonic ventral nerve cord (VNC), 30 bilaterally symmetric neuroblasts undergo repeated rounds of asymmetric divisions, producing a smaller ganglion mother cell that divides once again to generate postmitotic neural progeny. With each division, neuroblasts sequentially express a series of transcription factors, called temporal identity factors, Hunchback (Hb) ➔ Kruppel (Kr) ➔ Pou domain transcription factors 1 and 2 (collectively called Pdm) ➔ Castor (Cas), which specify the neural identity of the descendent neurons [2, 13,14,15,16,17,18]. While neuroblasts express each temporal identity factor only transiently, the neurons descended from each neuroblast division maintain active expression of the temporal identity factor, thus becoming molecularly marked by the timing of their birth.
Hb, the first of the temporal identity factor series, is a zinc-finger transcription factor that specifies early-born neural fate in multiple neuroblast lineages, similar to its mammalian homolog Ikaros, which promotes early-born identity in both the retina and the cortex [19, 20]. In neuroblasts, Hb is expressed for only the first one or two divisions and is rapidly repressed by a transient expression of its repressor, Seven-up (Svp) [21, 22]. Though neuroblasts express Hb only briefly, they remain competent to specify hb-transcribing early-born neurons for several additional divisions after hb is repressed in the neuroblast [9, 11, 12, 23]. Thus, the “early competence window” is a representation of neuroblast potential to generate early-born neurons that are marked by endogenous hb transcription, and this potential can be experimentally measured by identifying the neural progeny generated upon misexpressing Hb in the neuroblast progenitor. Previously, we showed that the early competence window is terminated when the hb gene physically relocates within the neuroblast to the nuclear lamina [11], a gene silencing hub [24, 25]. This movement to the nuclear lamina occurs much after hb is already transcriptionally silent, and renders the hb gene refractory to activation in the descendent neurons. Thus, while transcriptional repression by Svp terminates hb expression within the neuroblast, physical relocation of the hb gene to the nuclear lamina establishes a heritably silent gene state that terminates early competence (Fig. 1A). Little is known regarding the mechanisms underlying this second level of hb silencing.
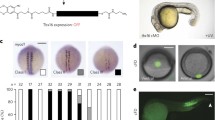
Early competence window is prolonged in corto mutants. A Left, NB7-1 lineage with neural progeny displayed by birth order, clockwise. The neuroblast is at the center and divided into time segments during which each temporal identity factor is expressed. Right, hunchback (hb) is transcriptionally repressed by transient pulse of Svp. Several divisions later, the hb gene relocates to the nuclear lamina, rendering the gene refractory to activation in the neural progeny, closing the early competence window. B NB7-1 lineage early competence assay in representative corto mutant (cortoL1) compared to sibling (heterozygous) control embryo. C Quantification of early competence window (Eve+HA+). Genotypes shown on x-axis. Each datapoint represents the average number of early-born (Eve+HA+) neurons from a single NB7-1 lineage. A minimum of three embryos were quantified per genotype. Data are represented as mean ± SD. P values represent comparisons to wild type. D Schematic summary of corto mutant competence phenotype
Here we identify an ETP protein Corto as a novel competence regulator. Loss of Corto results in a delay in hb gene relocation to the neuroblast nuclear lamina and prolongs competence to specify early-born neural identity. Furthermore, consistent with function as an ETP protein, we found that Corto genetically interacts with Psc, a core subunit of PRC1, to terminate early competence. While polycomb (PcG) chromatin factors are well known for their role in the maintenance of gene repression of its target genes during development [26,27,28], loss of Corto does not result in derepression of hb in late-stage neuroblasts. Together, our results provide insight into new regulators in nuclear architecture and the timing of neural progenitor competence transitions during development.
Materials and methods
Fly lines
Wild-type (w1118), cortoL1, corto420 (kind gift from Dr. Frédérique Peronnet, [29]), Psch27 (Bloomington, BL#5547), en-GAL4 (chrom II, [16, 30], UAS-hb [31], hbHA [11], UAS-corto [kind gift from Dr. Frédérique Peronnet, [29]]. Flies were raised on a standard cornmeal and molasses medium at 25C.
Immunostaining and antibodies
We immunostained embryos following standard protocols [32]. Briefly, we fixed embryos in a one-to-one mixture N-heptane and 4% formaldehyde diluted in PEM buffer (0.1 M Pipes, 1 mM MgS04, 2 mM EGTA) and rocked them in at room temperature for 22 min. The fixative solution was subsequently removed, and embryos were devitellinized by vigorous shaking in a 1:1 mixture of methanol:heptane. Devitellinized embryos were washed with PBS and incubated overnight at 4 °C in primary antibodies diluted in PBS-0.1% Tween 20 (PBT). After washing, embryos were incubated at room temperature in secondary antibodies for 1.5 h, and streptavidin for 20 min. The following primary antibodies were used: Mouse anti-Eve (clone 3C10, 1:50, Developmental Studies Hybridoma Bank (DSHB)), rat anti-HA (#11,867,423,001, clone 3F10, 1:500, Sigma), rabbit anti-lamin (1:2000, kind gift from Dr. Paul Fischer); rat anti-worniu (#196,362,clone 5A3AD2, 1:200, Abcam), rat anti-dpn (#195,172, clone 11D1CH11, 1:200, Abcam), mouse anti-PH3 (#14,955, 1:1000, Abcam), rabbit anti-Hb (1:200, kind gift from Dr. Chris Doe), rat anti-Zfh2 (1:200, kind gift from Dr. Chris Doe), mouse anti-AbdB (clone 1A2E9, 1:30, DSHB). Secondary antibodies against mouse, rabbit, and rat were conjugated to Alexa 488, 555, and 647 (Thermo Fisher Scientific).
Immuno-DNA FISH
DNA in situ hybridization (DNA-FISH) was performed as previously described [11, 23, 33]. Briefly, embryos were fixed in freshly-prepared 4% formaldehyde diluted in PIPES buffer (60 mM KCl, 15 mM NaCl, 0.5 mM spermidine, 0.15 mM spermine, 2 mM EDTA, 0.5 mM EGTA, 15 mM PIPES pH 7.4) and rocked for 25 min at room temperature with an equal volume of heptane. Embryos were devitellinized by vigorous shaking in a one-to-one solution of methanol:heptane. We generated fluorescent probes by PCR-amplifying approximately 10 kb of the hb genomic locus [11] and using the DNA FISH Tag kit (Thermo Fisher Scientific). After rehydration, embryos were treated with RNaseA (150 mg/ml) for 2 h at room temperature and gradually stepped into 100% pre-hybridization solution (pHM: 50% formamide; 4XSSC; 100 mM NaH2PO4, pH 7.0; 0.1% Tween 20). After one hour of pre-hybridization at 37 °C, embryos were denatured 15 min at 80 °C and incubated with denatured FISH probe diluted in pre-hybridization solution overnight at 37 °C in hybridization buffer (10% Dextran sulfate, 50% deionized formamide, 2XSSC, 0.5 mg/ml Salmon Sperm DNA). Embryos were washed at 37 °C in a series of formamide/0.3% CHAPS solutions with decreasing formamide at each wash. After the in situ hybridization protocol was completed, embryos were subsequently immunostained according to standard immunochemistry protocol (see above), mounted in Vectashield (Vectorlabs) and imaged on a confocal microscope, Zeiss LSM 700. Additional protocol details are available upon request.
Confocal imaging
We used a Zeiss 700 Axio Imager 2 laser scanning confocal for all images. DNA-FISH images were taken at a 0.4 µm step size, with pinholes that were adjusted to have equal optical section thickness in all channels.
Quantification and statistical analyses
We used standard t-tests using Prism v8. Statistical significance was classified as: ** < 0.01, *** < 0.001, **** < 0.0001. Embryos stage 14–16 were used in competence window tests. For graphs in which n’s represent individual neuroblasts, a minimum of three embryos were quantified per genotype, and for graphs in which n’s represent average of single embryos, at least twelve lineages were averaged per embryo. For DNA FISH, n’s represent each embryo, and number of FISH signals quantified are shown on the graph. Graphs show each data point used for statistical analysis, and bars indicate mean ± standard deviation.
Results
Loss of Corto extends the early competence window
Neuroblasts undergo two temporally-separated, distinct types of hb gene repression. In the first, hb is transcriptionally repressed by Svp after one-to-two divisions. In the second, several hours and divisions later, the already transcriptionally repressed hb gene physically relocates to the neuroblast nuclear lamina. This relocation causes heritable gene silencing and renders the hb gene refractory to activation in the descendent neurons, thereby closing the early competence window (Fig. 1A) [11, 23]. Thus, while neuroblasts typically generate one to two early-born neurons, they are capable of producing more until nuclear architecture reorganization terminates competence. We recently identified the PcG factors as critical players in the second step, in terminating neuroblast early competence. PcG proteins are chromatin factors best known for their function to regulate gene repression through the cooperative activity of two multimeric complexes, PRC1 and PRC2 [26, 28, 34,35,36,37,38,39]. We found that Psc is required for timely hb gene relocation to the neuroblast nuclear lamina and closing the early competence window [23].
Another class of proteins, the ETP proteins, has been shown to interact with the Trithorax and PcG complexes and regulate target genes. Corto, one such ETP protein, has been shown to genetically interact with PcG factors in Hox gene repression [29, 40, 41] and colocalizes with the majority of Psc-bound polytene chromosome sites [42]. Thus, we hypothesized that Corto may also function in regulating neuroblast competence. To test this, we focused on the NB7-1, one of the thirty neuroblast lineages of the embryonic VNC, for which we have markers to identify the progeny as well as have detailed knowledge of the temporal progression of its lineage and competence [9, 11, 12, 16, 23]. Briefly, we took advantage of the GAL4/UAS system [43] and used the engrailed GAL4 (en-GAL4) driver to drive strong, continuous Hb expression in NB7-1 throughout its entire lineage (en > hb) and determined the number of early-born neurons generated, a quantitative measure of the length of the early competence window. To distinguish between the endogenously activated Hb protein in response to early-born identity specification and the Hb protein misexpressed from the UAS-hb construct, we implemented an endogenously encoded bacterial artificial chromosome (BAC) transgene that includes the hb genomic locus and all of its regulatory enhancers. This BAC has been modified to include a hemagglutinin epitope tag (HA) fused to Hb, and our previous work has shown that this BAC insertion is sufficient to rescue a hb null animal and shows the same expression pattern as the native hb gene [11]. Thus, we are able to use HA as a proxy for endogenous Hb expression. We co-stained Hb and Even skipped (Eve), a marker for the U motoneuron progeny of the NB7-1 lineage, and quantified the number of early-born neurons (Eve+HA+) produced in response to continuous Hb misexpression. We note that in the competence assay, the degree of neuroblast response to Hb misexpression is variable even among homologous neuroblast lineages within an individual embryo. Thus, the length of the early competence window associated with a particular genotype is represented as an average of all lineages quantified. Compared to 5.1 ± 1.0 Eve+HA+ neurons in wild type NB7-1, we found 8.0 ± 2.3 early-born neurons in cortoL1 mutants (p < 0.0001). To control for any possible background mutations, we also examined transheterozygous animals of two independent loss of function alleles, corto L1 and corto420 [44] and observed consistent results, with 7.6 ± 2.7 early-born neurons generated (p < 0.0001) (Fig. 1B-D), indicating a prolonged early competence window. Thus, Corto plays an essential role in terminating early competence at mid-embryogenesis.
hb relocation to neuroblast nuclear lamina is delayed in corto mutant neuroblasts
We previously showed that the early competence window closes when the hb gene relocates to the neuroblast nuclear lamina at mid-embryogenesis [11, 23]. Thus, given the extension in early competence in corto mutants, we next examined hb gene positioning in corto mutant neuroblasts using in vivo immuno-DNA Fluorescence in situ Hybridization (FISH). Embryos were hybridized with a fluorescent DNA probe generated against approximately 10 kb at the hb gene locus and subsequently immunostained with Lamin Dm0, a B-type lamin intermediary filament that labels the nuclear envelope, and Worniu, a neuroblast-specific transcription factor. In corto mutant neuroblasts, we found that the proportion of hb gene loci at the nuclear lamina, which we defined as FISH signals that pixel-overlapped with lamin signals, was significantly reduced at mid-embryogenesis (stage 12: 56.9 ± 10.1% at lamina in wild type versus 33.7 ± 6.7% in corto−/−, P = 0.0004; stage 13/14: 56.4 + 6.6% at lamina in wild type versus 43.3 ± 2.8% in corto−/−, P = 0.0006) (Fig. 2A,B). We observed a reduction in hb localization to the nuclear lamina in corto mutant neuroblasts even when we included FISH signals that were near, but not touching the lamina (≤ 0.4µm from lamin) (Fig. 2B-ii). Interestingly, the difference between wild type and corto mutants diminished over time, and by late stage 15, there was no discernible difference in hb gene-lamina association between the genotypes (stage 15: 57.3 ± 3.8% at lamin in wild type versus 57.0 ± 3.3% in corto −/− mutants, P = not significant) (Fig. 2B). Thus, the data show that in corto mutants hb gene relocation to the neuroblast nuclear lamina is delayed. It is worth noting that we do not observe an all-or-nothing association of the hb gene with the nuclear lamina, and even at stage 11, during the early competence window, ~ 25% of the hb gene loci are localized at the lamina. In fact, reports from others show that even genomic regions outside of lamina-associated domains are located at the periphery ~ 30% of the time [45,46,47], underscoring the highly dynamic and non-static nature of the genome [48]. Importantly, we observe a two-fold increase in hb gene association with the neuroblast nuclear lamina at mid-embryogenesis in wild type embryos that has functional consequences on neuroblast competence, and this relocation is delayed in corto mutants.
hb gene relocation to neuroblast nuclear lamina is delayed in corto mutants. A Single z-plane through nucleus of representative neuroblast from intact stage 12 and 15, wild type or corto mutant (cortoL1) embryos, selected at the location of the brightest part of a hb DNA FISH signal (green spot). Immunostaining of lamin (magenta), and Worniu (pan-neuroblast, blue) shows position of hb loci within the three-dimensional space of neuroblast nuclei. Higher magnification of hb loci within neuroblast nucleus shown to the right of each FISH image; worniu is not shown for clarity. B Fraction of hb loci at the nuclear lamina (pixel-overlapping with lamin signal) quantified from wild type and corto mutant embryo neuroblasts across development. Each data point in line graph is the average fraction of hb loci at the lamina among multiple embryos (number of hb loci and embryos quantified shown below each stage). NS, not significant. Bar graphs to the right show mean fraction of hb loci either i) at lamin or ii) within 0.4µm from lamin. Each data point in bar graphs represents a single embryo. All data are represented as mean ± SD
To confirm that the delay in hb gene relocation in corto mutants is not due to a more general delay in neuroblast divisions, we co-stained embryos with Deadpan (Dpn), a pan-neuroblast marker, and phospho-histone 3 (PH3), a marker for dividing cells. There was no difference in numbers of dividing neuroblasts at either stage 12 or 14. Embryonic neuroblasts gradually divide more slowly as neurogenesis progresses, and this dynamic was unchanged in corto mutants, suggesting that neuroblast proliferation is progressing normally (Figure S1).
Normal Hb temporal dynamics in corto mutant neuroblasts
While genes localized at the nuclear lamina are typically in a repressed or silenced state, actively transcribed genes are often localized interiorly [49]. If the delay in hb gene relocation to the neuroblast nuclear lamina in Corto mutant embryos is due to prolonged hb transcription, we would expect a concomitant increase in the number of early-born neurons produced. By immunostaining for Eve, we did observe on occasion ectopic early-born neurons, but the occurrence was rare (2 Eve+Hb+ neurons in wild type compared to an average 2.1 in cortoL1/420 mutants) (Figure S2). Drosophila embryo development is highly stereotyped, allowing us to use morphological characteristics to compare stage-matched animals of different genotypes, and we found no qualitative differences in the timing of Hb repression in neuroblasts between wild type and corto mutants (Fig. 3A). By late stage 12, Hb is expressed by only a few neuroblasts in the thoracic segments (Fig. 3A, arrowheads) and is not detectable in any neuroblasts at stage 13, but rather is expressed in the neuronal layer, deeper in the embryo (Fig. 3B). Upon quantifying the proportion of Hb+ neuroblasts between stage 11 and 12, when Hb is rapidly being repressed across the neuroblast population, we measured no difference between wild type and corto mutants, indicating Hb was downregulated in a timely manner (Fig. 3C). Further arguing against a strong role for Corto as a hb transcriptional repressor, we found that Corto overexpression in neuroblasts had no effect on the production of early-born neurons (Figure S3), in contrast to hb’s known transcriptional repressor, Svp, which reduces the number of early-born neurons upon overexpression [21]. Thus, the impaired hb gene-lamina relocation phenotype in corto mutant neuroblasts we observed at stages 12–14 (7-11 h after egg lay, AEL), is well after hb is already transcriptionally downregulated, and the relocation phenotype is not due to prolonged hb transcription (prior to 7 h AEL) that keeps it in the nuclear interior.
Temporal dynamics of Hb repression in neuroblasts is not affected in corto mutants. A Ventral view of VNC wild type or corto mutant (cortoL1) embryos at late stage 12, when the last few neuroblasts in the thoracic segments still express Hb (cyan), but the rest, including all abdominal neuroblasts, are no longer Hb+. Neuroblasts are marked by pan-neuroblast marker, Dpn (red). B At stage 13, Hb is no longer detectable in any neuroblasts (top panels) but can be detected in neurons, deeper in the VNC (bottom panels). Hb shown alone in grayscale in A and B. C Quantification of fraction of neuroblasts expressing Hb at stage 11 and stage 12 comparing wild type and corto mutant embryos, showing similar decrease in Hb neuroblast expression over time. D Schematic diagram summarizing results
Psc and Corto genetically interact to restrict the early competence window
Corto has been reported to interact with PcG chromatin factors to maintain gene repression [29, 40]. In particular, Corto shares many binding sites on polytene chromosomes with Psc [40, 42], which plays a central role in PRC1 silencing activity [27]. We asked whether Corto and Psc genetically interact to terminate competence by testing the early competence window in transheterozygous mutant embryos. We examined embryos stages 14–16, the tail end of the NB7-1 lineage, and found that while neither Psc heterozygotes or corto heterozygotes alone have a competence phenotype (P value not significant), Psch27/+;cortoL1/+ transheterozygous animals have prolonged early competence (Fig. 4A) (Psch27/+;cortoL1/+ 6.4 ± 1.5 vs. wild type 5.4 + 1.1, P = 0.0004). Given the variability in response to Hb misexpression even among homologous neuroblasts within the same embryo, we additionally displayed the data as a scatterplot, showing the number of early-born neurons relative to Eve+ neurons, representing the total cells in the lineage upon Hb misexpression (Fig. 4A-ii). Psch27/+;cortoL1/+ transheterozygous neuroblasts generated, overall, a higher fraction of early-born neurons than wild type neuroblasts with similar numbers of Eve+ cells, indicating increased competence among neuroblasts with comparable levels of Hb misexpression. We note that for the stages analyzed, the number of Eve+ cells is higher in older embryos, as expected (stage 14: 18.7 ± 2.9 Eve+ cells at stages 14, n = 68 lineages from 5 embryos, vs stage15/16: 23.0 ± 3.6, n = 136 lineages from 8 embryos, p < 0.0001), whereas the number of Eve+HA+ (early-born) neurons did not correlate with stage (stage 14: 5.9 ± 1.5 Eve+HA+ neurons vs stage 15/16: 5.6 ± 1.4, not significant). This confirms that all embryos sampled had completed production of early-born neurons. Together, we conclude that Psc and Corto genetically interact to close the competence window to specify early-born neurons.
Corto and Psc genetically interact to close the early competence window. A i), NB7-1 lineage early competence assay comparing wild type, cortoL1/+, Psch27/+, or corto L1/+;Psc27/+ transheterozygous embryos. Bars represent mean ± SD. ii), Scatterplot of wild type and corto L1/+;Psc27/+ data, showing the number of Eve+HA+ (early-born) neurons relative to the total number of Eve neurons within an individual NB7-1 lineage. Each data point represents competence data of a single NB7-1 lineage quantified from three to four embryos per genotype. B Schematic circular diagram depicting results. C Early competence window test with and without co-overexpression of Corto. Example NB7-1 lineage shown for Hb and Corto co-misexpressed by en-Gal4 driver, stained for HbHA (cyan) and Eve (red). Quantification shown to the right. Each data point represents the average of at least twelve neuroblast lineages from a single embryo. Bars represent mean ± SD
In contrast, overexpression of Corto did not have any effect on competence. While wild type animals had an average of 5.5 ± 0.3 Eve+HA+ neurons upon Hb misexpression (68 hemisegments quantified from n = 4 embryos), co-overexpression of Corto did not yield a statistically different result, 6.1 ± 0.3 Eve+HA+ neurons (86 hemisegments quantified from n = 5 embryos) (Fig. 4C). If Corto acts in neuroblasts primarily through its interactions with the PcG complex, perhaps the stoichiometry or activity level of one or more PcG complex subunits is rate-limiting.
Corto does not act to maintain repression of Hb or the Hox gene, Abdominal B, in neuroblasts
PcG factors have been well studied for their roles in maintenance of target gene repression. As an enhancer of PcG, Corto has been reported to play a similar role in the repression of Abd-B, a known PcG Hox target gene, and loss of Corto results in its derepression [41]. Our recent study [23] showed that the hb intron region is a strong PcG target site, and loss of PRC1 impairs hb gene-lamina relocation. Interestingly, however, loss of PcG does not result in Hb derepression in neuroblasts even in late stage embryos. Moreover, even the most severe loss of function mutants for PRC1 (Psc-Su(z)2P3C) and PRC2 (maternal and zygotic mutants of extra sex combs, escmat/zyg), did not show any neuroblast derepression of Hb in late-stage embryos [23], raising the possibility that PcG factors may not function in transcriptional repression in this cell type. We thus examined Hb expression in late-stage corto mutant embryos to determine whether Hb becomes derepressed. Similar to the PRC1 mutants, we also did not find Hb derepressed in neuroblasts of late stage corto mutant embryos (Fig. 5A). While we did observe Abd-B derepression in corto mutants, consistent with previous observations [41], surprisingly, the derepression was limited to the epithelia, and we did not find Abd-B derepressed in neuroblasts (Fig. 5B-C). In summary (Fig. 6), our results show that Corto is required for the timely relocation of the hb gene to the neuroblast nuclear lamina and genetically interacts with PRC1 to terminate early competence. However, it does not play a role in maintaining hb gene repression, and in a departure from its known function, it does not play a role in repression of its known target Abd-B, within neuroblasts.
Hb and Abd-B are not derepressed in neuroblasts in late stage corto mutant (corto420) embryos. A VNC of control (corto+/-) or corto mutants at stage 15, stained with Worniu (pan-neuroblast marker, cyan) and Hb (red). Dashed box region shown as separate Worniu and Hb panels to the right. B Control and corto mutant whole embryos at stage 15 are shown as side and ventral views, stained with Abd-B. Dashed white lines at the top show areas of derepressed Abd-B. Yellow dashed line demarcates VNC region. B’ Higher magnification of regions labeled i) and ii) in ventral view of corto mutant in B. Yellow vertical line show VNC region. Area within yellow box in ii) are shown in higher magnification to the right
Summary model. In both wild type and corto mutant neuroblasts, hb transcriptional dynamics are largely normal (endogenous hb depicted in blue). Much after hb repression, hb gene relocation to the neuroblast nuclear lamina terminates the early competence window. Loss of Corto, which genetically interacts with PcG, delays this relocation and extends early competence (misexpressed Hb depicted in green)
Discussion
Here we identify Corto as a regulator of neuroblast early competence in the Drosophila embryo. Loss of Corto results in a delay in hb gene relocation to the nuclear lamina in neuroblasts and an extension in the early competence window. While the low numbers of extra early-born neurons found in a subset of neuroblast lineages in corto mutant embryos could indicate an occasional extension of neuroblast Hb expression, we did not observe any measurable changes to Hb transcriptional dynamics in corto mutants across the neuroblast population. In contrast, the delay in hb gene relocation was robust across neuroblasts and occurred substantially after the hb gene was already repressed and thus not a result of prolonged transcriptional activity.
In the fly embryo, neuroblasts transiently express a series of temporal identity factors which confer specific traits to the neurons descended from each division. Hb, the first of the temporal identity factors, specifies early-born identity, which includes endogenous activation of the hb gene within the postmitotic neuron. Misexpression of Hb directly in the postmitotic neuron cannot induce hb transcription [11, 12]. The molecular identity of the neuron, including its ability to activate and sustain hb transcription endogenously, must be established within the neuroblast progenitor and subsequently inherited. Thus, the neuroblast “primes” the future transcriptional program that is activated by the neural progeny. Critically, the neuroblast’s potential to generate an hb-transcribing early-born neuron ends not when hb is transcriptionally repressed within the neuroblast, but rather several divisions later when the hb gene relocates to the nuclear lamina. Upon relocation, hb becomes refractory to activation within the descendent neuron, thus closing the early competence window. Corto is required for this heritable silencing through hb gene relocation to the neuroblast nuclear lamina. However, relocation in corto mutants is only delayed, not completely blocked, as the hb gene gradually increases association with the lamina, perhaps due to compensation by PcG factors with which it interacts. Consistent with this notion, overexpression of Corto alone did not shorten the early competence window.
We have focused on hb as a model gene to study mechanism of neuroblast competence regulation, as it is the only universally expressed gene among an otherwise diverse population of early-born neurons, but it is not clear what role, if any, Hb itself plays in the postmitotic, early-born neurons of the VNC. RNAi-mediated knockdown of Hb in NB7-1 neural progeny does not alter neuronal morphology or affect larval locomotor velocity [50]. We speculate that hb is only one component of a broad transcriptional program in postmitotic neurons established by the nuclear architecture of the neuroblast upon specification of early-born identity, and hb transcription may reflect its epigenetic state, rather than a requirement for function in these neurons. In the postembryonic brain, however, Hb in postmitotic neurons has been shown to play an important role in sexually dimorphic interneuron neurite morphology [51], and a recent study established a functional role for Hb in larval Pair1 neuron in synapse number, connectivity, and behavior [52]. It would be important for future studies to determine gene-lamina interactions in neuroblasts on a genome-wide scale and relate these lamina-associated genes to neuroblast competence, gene expression and neuronal function. In this context, identifying trans-acting factors of nuclear architecture and understanding the mechanisms of their function is a critical area requiring further study. Here we have found Corto, which impacts hb gene relocation without affecting hb transcriptional dynamics.
Though Corto has been shown to participate in PcG-mediated silencing [40, 41], we did not observe any Hb derepression in neuroblasts in later embryonic stages, consistent with our recent observations for PcG [23]. While PcG factors bind the hb gene locus and are required for hb gene relocation in neuroblasts, they do not function in regulation of hb transcriptional repression, a departure from their well-known roles in maintenance of target gene repression. Further supporting the lack of a repressive role in neuroblasts, overexpression of Corto did not reduce the number of early-born neurons, which is in contrast to what has been reported for Svp, hb’s established transcriptional repressor [21, 22]. Additionally, PcG and Corto have been shown to be required for repression of Abd-B, the PcG Hox target gene, and similar to observation by others, we found Abd-B to be derepressed in corto mutants. Interestingly, however, while we found Abd-B derepression in the epithelia, we did not observe this in neuroblasts. Perhaps this suggests that PcG/Corto have a unique role in neuroblasts by facilitating changes to their nuclear architecture, a function independent of their better known roles in transcriptional repression. Further studies are necessary to investigate a more general role of PcG/Corto in neuroblasts comparing genome architecture and transcriptional regulation, and understand how such activity underlie neuroblast competence to determine neuronal identity and function.
Conclusions
Increasing evidence points to an important role for three dimensional organization of genome architecture in regulating transcriptional and competence states of progenitors during animal development [53, 54]. We have identified Corto, an ETP class chromatin factor, as a new regulator of neuroblast competence. Corto is required for the timely relocation of the hb gene to the neuroblast nuclear lamina and genetically interacts with Psc to close the early competence window. In neuroblasts, loss of Corto does not impact hb transcriptional dynamics nor does it cause derepression of Abd-B, a departure from its known role in Abd-B repression. By identifying new chromatin regulators of competence, our results provide further mechanistic insights into how progenitor competence is regulated in vivo. Together, the results show that multiple, distinct repressive mechanisms sequentially operate in a step-wise fashion as neuroblasts age over time to terminate neuroblast competence to specify early-born neural identity.
Availability of data and materials
No datasets or new fly stocks were generated. Fly stocks used in this manuscript are available from public stock centers or will be provided upon request to corresponding author.
References
Brody T, Odenwald WF. Cellular diversity in the developing nervous system: a temporal view from Drosophila. Development. 2002;129(16):3763–70.
Kohwi M, Doe CQ. Temporal fate specification and neural progenitor competence during development. Nat Rev Neurosci. 2013;14(12):823–38.
Livesey FJ, Cepko CL. Vertebrate neural cell-fate determination: lessons from the retina. Nat Rev Neurosci. 2001;2(2):109–18.
McConnell SK. Fates of visual cortical neurons in the ferret after isochronic and heterochronic transplantation. J Neurosci. 1988;8(3):945–74.
Okano H, Temple S. Cell types to order: temporal specification of CNS stem cells. Curr Opin Neurobiol. 2009;19(2):112–9.
Rossi AM, Fernandes VM, Desplan C. Timing temporal transitions during brain development. Curr Opin Neurobiol. 2017;42:84–92.
Thor S. Nervous System Development: Temporal Patterning of Large Neural Lineages. Curr Biol. 2017;27(10):R392–4.
Cepko CL, et al. Cell fate determination in the vertebrate retina. Proc Natl Acad Sci U S A. 1996;93(2):589–95.
Cleary MD, Doe CQ. Regulation of neuroblast competence: multiple temporal identity factors specify distinct neuronal fates within a single early competence window. Genes Dev. 2006;20(4):429–34.
Hirabayashi Y, et al. Polycomb limits the neurogenic competence of neural precursor cells to promote astrogenic fate transition. Neuron. 2009;63(5):600–13.
Kohwi M, et al. Developmentally regulated subnuclear genome reorganization restricts neural progenitor competence in Drosophila. Cell. 2013;152(1–2):97–108.
Pearson BJ, Doe CQ. Regulation of neuroblast competence in Drosophila. Nature. 2003;425(6958):624–8.
Baumgardt M, et al. Neuronal subtype specification within a lineage by opposing temporal feed-forward loops. Cell. 2009;139(5):969–82.
Brody T, Odenwald WF. Programmed transformations in neuroblast gene expression during Drosophila CNS lineage development. Dev Biol. 2000;226(1):34–44.
Grosskortenhaus R, et al. Regulation of temporal identity transitions in Drosophila neuroblasts. Dev Cell. 2005;8(2):193–202.
Isshiki T, et al. Drosophila neuroblasts sequentially express transcription factors which specify the temporal identity of their neuronal progeny. Cell. 2001;106(4):511–21.
Grosskortenhaus R, Robinson KJ, Doe CQ. Pdm and Castor specify late-born motor neuron identity in the NB7-1 lineage. Genes Dev. 2006;20(18):2618–27.
Bahrampour S, et al. Neural Lineage Progression Controlled by a Temporal Proliferation Program. Dev Cell. 2017;43(3):332-348e4.
Alsio JM, et al. Ikaros promotes early-born neuronal fates in the cerebral cortex. Proc Natl Acad Sci USA. 2013;110(8):E716–25.
Elliott J, et al. Ikaros confers early temporal competence to mouse retinal progenitor cells. Neuron. 2008;60(1):26–39.
Kanai MI, Okabe M, Hiromi Y. seven-up Controls switching of transcription factors that specify temporal identities of Drosophila neuroblasts. Dev Cell. 2005;8(2):203–13.
Mettler U, Vogler G, Urban J. Timing of identity: spatiotemporal regulation of hunchback in neuroblast lineages of Drosophila by Seven-up and Prospero. Development. 2006;133(3):429–37.
Lucas T, et al. Discrete cis-acting element regulates developmentally timed gene-lamina relocation and neural progenitor competence in vivo. Dev Cell. 2021;56(18):2649-2663.e6.
Shevelyov YY, Nurminsky DI. The Nuclear Lamina as a Gene-silencing Hub. Curr Issues Mol Biol. 2011;14(1):27–38.
van Steensel B, Belmont AS. Lamina-associated domains: links with chromosome architecture, heterochromatin, and gene repression. Cell. 2017;169(5):780–91.
Grossniklaus U, Paro R. Transcriptional silencing by polycomb-group proteins. Cold Spring Harb Perspect Biol. 2014;6(11):a019331.
King IF, et al. Analysis of a polycomb group protein defines regions that link repressive activity on nucleosomal templates to in vivo function. Mol Cell Biol. 2005;25(15):6578–91.
Schwartz YB, et al. Genome-wide analysis of polycomb targets in Drosophila melanogaster. Nat Genet. 2006;38(6):700–5.
Lopez A, et al. corto genetically interacts with Pc-G and trx-G genes and maintains the anterior boundary of ultrabithorax expression in Drosophila larvae. Mol Genet Genomics. 2001;266(4):572–83.
Harrison DA, et al. Activation of a Drosophila Janus kinase (JAK) causes hematopoietic neoplasia and developmental defects. EMBO J. 1995;14(12):2857–65.
Wimmer EA, et al. Bicoid-independent formation of thoracic segments in Drosophila. Science. 2000;287(5462):2476–9.
Rothwell W.F.a.S., W., Fluorescent analysis of Drosophila embryos Drosophila Protocols. 2000, Cold Spring Harbor: Cold Spring Harbor Press.
Bantignies F, et al. Polycomb-dependent regulatory contacts between distant Hox loci in Drosophila. Cell. 2011;144(2):214–26.
Negre N, et al. Chromosomal distribution of PcG proteins during Drosophila development. PLoS biology. 2006;4(6):e170.
Schuettengruber B, Cavalli G. Recruitment of polycomb group complexes and their role in the dynamic regulation of cell fate choice. Development. 2009;136(21):3531–42.
Simon J, Chiang A, Bender W. Ten different Polycomb group genes are required for spatial control of the abdA and AbdB homeotic products. Development. 1992;114(2):493–505.
Struhl G. A gene product required for correct initiation of segmental determination in Drosophila. Nature. 1981;293(5827):36–41.
Tolhuis B, et al. Genome-wide profiling of PRC1 and PRC2 Polycomb chromatin binding in Drosophila melanogaster. Nat Genet. 2006;38(6):694–9.
Beuchle D, Struhl G, Muller J. Polycomb group proteins and heritable silencing of Drosophila Hox genes. Development. 2001;128(6):993–1004.
Salvaing J, et al. The Drosophila Corto protein interacts with Polycomb-group proteins and the GAGA factor. Nucleic Acids Res. 2003;31(11):2873–82.
Salvaing J, et al. The enhancer of trithorax and polycomb corto interacts with cyclin G in Drosophila. PLoS One. 2008;3(2):e1658.
Kodjabachian L, et al. Mutations in ccf, a novel Drosophila gene encoding a chromosomal factor, affect progression through mitosis and interact with Pc-G mutations. EMBO J. 1998;17(4):1063–75.
Brand AH, Perrimon N. Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development. 1993;118(2):401–15.
Mouchel-Vielh E, et al. Involvement of the MP1 scaffold protein in ERK signaling regulation during Drosophila wing development. Genes Cells. 2008;13(11):1099–111.
Guelen L, et al. Domain organization of human chromosomes revealed by mapping of nuclear lamina interactions. Nature. 2008;453(7197):948–51.
Harr JC, et al. Directed targeting of chromatin to the nuclear lamina is mediated by chromatin state and A-type lamins. J Cell Biol. 2015;208(1):33–52.
Zullo JM, et al. DNA sequence-dependent compartmentalization and silencing of chromatin at the nuclear lamina. Cell. 2012;149(7):1474–87.
Kind J, et al. Genome-wide maps of nuclear lamina interactions in single human cells. Cell. 2015;163(1):134–47.
Meister P, et al. The spatial dynamics of tissue-specific promoters during C. elegans development. Genes Dev. 2010;24(8):766–82.
Hirono K, et al. The Hunchback temporal transcription factor establishes, but is not required to maintain, early-born neuronal identity. Neural Dev. 2017;12(1):1.
Goto J, et al. Sexually dimorphic shaping of interneuron dendrites involves the hunchback transcription factor. J Neurosci. 2011;31(14):5454–9.
Lee KM, Linskens AM, Doe CQ. Hunchback activates Bicoid in post-mitotic Pair1 neurons to regulate synapse number. bioRxiv: the preprint server for biology. Cold Spring Harbor: Cold Spring Harbor Laboratory; 2021.
Alexander JM, Lomvardas S. Nuclear architecture as an epigenetic regulator of neural development and function. Neuroscience. 2014;264:39–50.
Lucas T, Kohwi M. From insects to mammals: regulation of genome architecture in neural development. Curr Opin Neurobiol. 2019;59:146–56.
Acknowledgements
We thank Dr. Frédérique Peronnet (French National Centre for Scientific Research) for the Corto fly stocks. We thank Dr. Paul Fisher (Stonybrook University) for the lamin antibody. We thank Dr. Gary Struhl (Columbia University) for helpful discussion on the science. We thank Dr. Natalia Molotkova for technical assistance. We thank Drs. Tanguy Lucas and Rui Galvão for critical reading of the manuscript. We thank the DSHB for antibodies and the Bloomington stock center (NIH P40OD018537) for fly stocks.
Funding
This work was supported by the NICHD (R00HD072035 and HD092381), the Rita Allen Foundation, and the Whitehall Foundation.
Author information
Authors and Affiliations
Contributions
T.L.H. and M.K. designed the research. T.L.H., S.P., D.T., and MK executed the experiments, collected and analyzed the data. T.L.H. and M.K. wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable, as no vertebrate or human subjects were used in this manuscript.
Consent for publication
All authors approved this manuscript.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Hafer, T.L., Patra, S., Tagami, D. et al. Enhancer of trithorax/polycomb, Corto, regulates timing of hunchback gene relocation and competence in Drosophila neuroblasts. Neural Dev 17, 3 (2022). https://doi.org/10.1186/s13064-022-00159-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13064-022-00159-3