Abstract
Background
The insulin/insulin-like signalling (IIS) pathway is common in mammals and invertebrates, and the IIS pathway is unknown in Fasciola gigantica. In the present study, the IIS pathway was reconstructed in F. gigantica. We defined the components involved in the IIS pathway and investigated the transcription profiles of these genes for all developmental stages of F. gigantica. In addition, the presence of these components in excretory and secretory products (ESPs) was predicted via signal peptide annotation.
Results
The core components of the IIS pathway were detected in F. gigantica. Among these proteins, one ligand (FgILP) and one insulin-like molecule binding protein (FgIGFBP) were analysed. Interestingly, three receptors (FgIR-1/FgIR-2/FgIR-3) were detected, and a novel receptor, FgIR-3, was screened, suggesting novel functions. Fg14-3-3ζ, Fgirs, and Fgpp2a exhibited increased transcription in 42-day-old juveniles and 70-day-old juveniles, while Fgilp, Fgigfb, Fgsgk-1, Fgakt-1, Fgir-3, Fgpten, and Fgaap-1 exhibited increased transcription in metacercariae. FgILP, FgIGFBP, FgIR-2, FgIR-3, and two transcription factors (FgHSF-1 and FgSKN-1) were predicted to be present in FgESPs, indicating their exogenous roles.
Conclusions
This study helps to elucidate the signal transduction pathway of IIS in F. gigantica, which will aid in understanding the interaction between flukes and hosts, as well as in understanding fluke developmental regulation, and will also lay a foundation for further characterisation of the IIS pathways of trematodes.
Similar content being viewed by others
Background
The insulin/insulin-like signalling (IIS) pathway is initiated by insulin and insulin-like growth factors and amplified by signalling cascades that are regulated by transcription factors, thereby regulating a range of biological processes [1]. When IIS is activated, downstream transcription factors are highly important for the communication and coordination of cells, tissues and organs [2, 3]. In mammals, IIS is involved in the regulation of lifespan, reproductive development, and metabolism [4, 5]. Research in Caenorhabditis elegans has revealed that the IIS pathway regulates lifespan, reproduction, and metabolism, suggesting that this pathway has a conserved function [6, 7]. In addition, IIS signalling plays a role in dauer development in C. elegans [8]. As insulin receptors are activated, transduced signals induce the phosphorylation of the transcription factor DAF-16 and subsequently promote the development of the worm under favourable circumstances [9, 10]. When unphosphorylated DAF-16 promotes downstream gene transcription, C. elegans can enter the dauer phase under unfavourable circumstances. In a study of Schistosoma mansoni, the upstream components of the IIS pathway were shown to potentially regulate fluke development and interaction with the host [11].
Research on the IIS pathway in Fasciola gigantica is lacking, and the IIS pathway model in F. gigantica is unknown. In-depth exploration of this pathway is helpful for exploring fluke development and fluke–host interactions, which may guide the prevention of fascioliasis. In addition, given the evolutionary conservation of the IIS pathway, studies regarding its role in F. gigantica may help to elucidate its function and regulation in other organisms and promote the treatment of metabolic diseases, such as diabetes and tumour [12, 13]. Fortunately, transcriptomic and genomic studies of F. gigantica have laid a solid foundation for further exploration. In this study, we explored the IIS pathway, mainly by (i) identifying insulin signal transduction components, (ii) examining their transcriptional profiles at different stages in the F. gigantica lifespan, and (iii) predicting their presence in excretory-secretory products (ESPs).
Methods
Identification of insulin signalling pathway components
Since the IIS pathway in C. elegans has been fully annotated (https://parasite.wormbase.org/index.html) and insulin pathway components have been partially identified in Schistosoma japonicum, the homologous components of the F. gigantica insulin signalling pathway were screened based on the corresponding components. BLAST searches were performed using known insulin pathway components to screen homologues of F. gigantica in nonredundant protein sequences, as well as the F. gigantica genome (PRJNA339660) and transcriptome (NCBI BioProject accession no. PRJNA230515) databases. Then, all screened components were mapped using BLAST (E-value cut-off: 10− 5) to verify that their regions were homologous to known insulin signalling components. All of the screened proteins were characterised by their primary amino acid sequence, and their structural and/or functional domains were inferred using bioinformatics tools (i.e., SignaIP5.0, TMHMM2.0, SWIMMMODEL, Expaxy, InterproScan and SMART) [14,15,16].
In addition, phylogenetic analyses were performed to analyse 3 receptors in the insulin signalling pathway of F. gigantica. Based on sequences from nematodes (C. elegans DAF-2), invertebrates (Drosophila melanogaster IR-1/IR-2), platyhelminths (S. mansoni IR-1/IR-2, S. japonicum IR-1/IR-2, Fasciola hepatica IR-1/IR-2/IR-3, Fasciolopsis buski IR-1/IR-2/IR-3, Echinococcus multilocularis IR-1/IR-2), and chordates (Homo sapiens IR-1/IR-2, Mus musculus IR-1/IR-2), the evolutionary relationships of FgIR-1/FgIR-2/FgIR-3 were explored. Phylogenetic analyses of the aligned sequence data were conducted using the neighbour‒joining (NJ), maximum parsimony (MP), and maximum likelihood (ML) methods and the Jones–Taylor–Thornton (JTT) model in MEGA 11.0. The confidence intervals were evaluated using a bootstrap procedure with 1000 pseudoreplicates. A 50% cut-off value was used for the consensus tree [17].
Analysis of differential transcription
Transcriptomic data from different developmental stages of F. gigantica, including the egg, miracidiae, rediae, cercariae, and metacercariae stages, as well as 42-day old juveniles, 70-day old juveniles, and adults, were previously generated by RNA-seq, using 3 samples from each stage and age group [18] and made publicly available (NCBI BioProject accession no.PRJNA350370). The sequences were aligned to the available F. gigantica genome sequence (accession number GWHAZTT00000000). The transcription of the genes were calculated by the fragments per kilobase per million reads (FPKM) method [19], which was used to compare the differences in gene transcription levels across different developmental stages. The false discovery rate (FDR) is a statistical method that was used to correct for p values. Genes with an adjusted p value < 0.05 according to DESeq were considered differentially transcribed genes [20]. The transcription data were analysed, clustered, and visualised using an online program (https://cloud.oebiotech.com/task/detail/heatmap/).
Prediction of insulin signalling pathway components in ESPs
While classic secretory proteins were predicted by the SignalP 5.0, TargetP and TMHMM programs [21,22,23], noncanonical secretory proteins were predicted by the SecretomeP-2.0 program [24].
Results
Identification and characterisation of insulin signalling pathway components
The upstream components of the IIS pathway in F. gigantica were screened based on the S. japonicum insulin-like peptide and receptor components. One insulin-like peptide homologue (FgILP), one insulin-like molecular binding protein (FgIGFBP), and 3 insulin receptors (FgIR-1, FgIR-2 and FgIR-3) were identified (Table 1).
Fourteen homologous components downstream of the pathway in C. elegans were screened in F. gigantica, including 11 signal transduction proteins (FgIRS, FgAAP, FgPI3K, FgPDK-1, FgAKT-1, FgAKT-2, FgSGK-1, FgPTEN, FgPP2A, Fg14-3-3ζ, and FgDDL-1) and 3 transcription factors (FgFOXO, FgSKN-1 and FgHSF-1) (Table 2).
In the present study, one insulin ligand, FgILP, which encodes 137 aa and has 35.88% similarity to SjILP, was identified. InterScan analysis revealed that FgILP contains IGF family domains, which include the insulin B chain and insulin A chain. Like other insulin-like peptides, FgILP has a conserved cysteine motif, namely, the ‘CCXXXXCXXXXXXXC’ sequence (Additional file 1, Fig. S1).
FgIGFBP encodes 435 aa and has 27.70% similarity to HsIGFBP. FgIGFBP contains an insulin-like growth factor binding protein region located at aa 59 to 116, and the IB domain is located at the C-terminus (Additional file 1, Fig. S1).
Three receptors, FgIR-1, FgIR-2 and FgIR-3, were analysed, and each of them contained protein kinase binding regions and ATP binding sites. FgIR-1 shares 58.37% similarity with SmIR-1, and FgIR-2 shares 50.51% similarity with SmIR-2. FgIR-3 shares 41.49% similarity with FgIR-1 and shows 48.03% similarity with FgIR-2. However, a BLAST search of FgIR-3 revealed no homologues in trematodes (Additional file 1, Fig. S1).
Eleven intracytoplasmic components, including FgIRS, FgAAP, FgPI3K, FgPDK-1, FgAKT-1, FgAKT-2, FgSGK-1, FgPTEN, FgPP2A, Fg14-3-3ζ and FgDDL were investigated. FgAAP encodes 1171 aa, is 30.93% similar to CeAAP-1, and contains a protein kinase C domain and src homology 2 domain (SH2 domain). FgPI3K encodes aa 1304, which includes a p85-binding domain and a Ras-binding domain. In addition, the catalytic domain of class I phosphoinositide 3-kinase is located at the C-terminus. FgPDK-1 shares 31.71% similarity with CePDK-1 and contains a serine/threonine kinase catalytic site and a PH domain (Additional file 1, Fig. S2).
As protein kinases, FgAKT-1 and FgAKT-2 encode 957 and 278 aa, respectively, each of which contains a protein kinase domain. FgSGK-1, which encodes a sequence of 350 aa, contains a protein kinase domain and a S–TK–X domain. FgPTEN encodes 646 aa and contains the C2 domain of the PTEN tumour-suppressor protein (PTPc domain). FgPP2A shares 60.57% similarity with CePPTR-1, which consists of a protein phosphatase 2 A regulatory B subunit. As a homologue of CeFTT-2 and CePAR-5, Fg14-3-3ζ, which encodes 252 aa, shares 62.66% similarity with CePAR-5 and 64.98% similarity with CeFTT-2, and consists of 14-3-3 domains. FgDDL-1, which encodes 91 aa, shares 40.32% similarity with CeDDL-2 and contains a WASH complex subunit homologue domain. Three transcription factors, FgFOXO, FgSKN, and FgHSF were also identified. FgFOXO encodes 987 aa and consists of conserved FH-FOXO motifs. FgSKN-1 shares 39.13% similarity with CeSKN-1 and consists of the basic leucine zipper (bZIP) domain of bZIP transcription factors. FgHSF-1, which encodes 258 aa and shows 30.33% similarity to CeHSF-1, contains a heat shock transcription factor domain (Additional file 1, Fig. S2).
The genes were cloned using primers (Additional file 2, Table S1) as shown in the gel (Additional file 3, Fig. S1) and sequenced. The actual length of sequences were consistent with the transcriptome data.
Phylogenetic analysis
Phylogenetic trees were constructed to explore the relationships of the 3 receptors (Fig. 1). The topologies of the NJ, MP and ML trees were concordant (Additional file 1, Fig. S3). FgIR-1 was grouped with SmIR-1 with 91% bootstrap support. FgIR-2 was grouped with SmIR-2 and EmIR-2, while FgIR-3 and FhIR-3 constituted an independent cluster.
Phylogenetic relationships of insulin receptors in F. gigantica and homologues of 11 other species. A neighbour‒joining tree was constructed, and bootstrap values were shown below the branches. The sequences used and their GenBank accession numbers were as follows: F. hepatica (IR-1, THD26449; IR-2, THD19358; IR-3, THD24184); F. gigantica (IR-1, TPP62725; IR-2, TPP64585; IR-3, TPP63095. ); F. buski (IR-1, KAA0195064; IR-2, KAA0201248); S. mansoni (IR-1, AAN39120; IR-2, AAV65745); S. japonicum (IR-1, ACT20714; IR-2, ACT20715); C. elegans (DAF-2, AAC47715); E. multilocularis (IR-1, CAD30260; IR-2, CDS42114); H. sapiens (IR-1, XP-047288401; IR-2, NP-001073285); Danio rerio (IR-1, NP-00111670; IR-2, NP-001136144; D. melanogaster (IR-1, NP-00113809); M. musculus (IR-1, NP-035962; IR-2, NP-034698).
The IIS pathway in F. gigantica
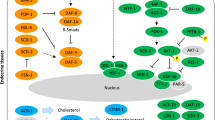
Using the available information, we constructed an insulin-like signalling pathway for F. gigantica (Fig. 2). FgILP can bind to and activate the receptor FgIR1/FgIR2/FgIR3, while FgIGFBP can aggregate FgILP to reduce its free concentration, thereby regulating insulin signalling. FgIR activation results in the recruitment of FgPI3K. Then, the downstream kinases FgPDK-1, FgAKT-1, and FgAKT-2 are activated, resulting in the phosphorylation of the transcription factor FgFOXO. Fg14-3-3-ζ regulates its interaction with FgFOXO to regulate FgFOXO localisation. FgSKN-1 is phosphorylated by FgSGK-1, inhibiting its subcellular translocation to the nucleus. In addition to participating in a series of phosphorylation cascades, FgTLP can also bind to and activate the receptor and subsequently phosphorylate FgDDL-1 directly, in turn regulating the transcription of FgHSF-1.
In the process of signal transduction, FgPTEN acts as an activator to promote the transformation of PIP3 to PIP2, inducing the phosphorylation of FgPDK-1 and promoting the posttranscriptional translation of transcription factors. As an antagonist, FgPP2A can bind to FgAKT, inhibiting its phosphorylation and subsequent signal transduction. Numerous downstream target genes are involved in various processes, such as stress reactions, longevity, reproduction and the activation of other pathways.
Schematic of the F. gigantica IIS pathway. After binding to the receptor, FgILP recruit the insulin receptor substrate IRS, leading to PI3K phosphorylation and promoting the conversion of PIP3 to PIP2. PIP3 signalling activates downstream kinase cascades and promotes FgFOXO and FgSKN-1 transcription factor translocation (solid line). The transcription factor FgHSF-1 is activated by FgDDL-1 and undergoes nuclear translocation (the dashed line indicates this). The core components of the insulin pathway are represented by green boxes; phosphorylation is represented by red circles, and the blue boxes represents the transcription factors. The insulin pathway regulates the transcription of various genes and subsequently regulates key processes, including stress reactions, resistance, longevity, ageing, reproduction, development, metabolism and other pathways, such as the TGF-β signalling pathway
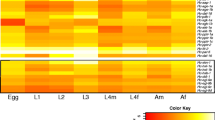
Transcript abundance based on RNA-seq analysis
Among the 19 genes, group A genes (5 genes, including Fg14-3-3-ζ, Fgir-2, Fgirs, Fgpp2a, and Fgddl-1) were highly transcribed in 42-day-old juveniles and 70-day-old juveniles (Fig. 3). Group B (6 genes, including Fgskn-1, Fgpdk-1, Fgir-1, Fgfoxo, Fghfs-1, and Fgpi3k) exhibited increased transcription in miracidiae, while group C (3 genes, including Fgaap, Fgsgk-1, and Fgilp) exhibited increased transcription in cercariae and metacercariae. Group D (4 genes, including Fgakt-1, Fgakt-2, Fgir-3, and Fgpten) exhibited increased transcription in metacercariae.
Apart from Fgir-2 and Fgpdk-1, 17 genes involved in the insulin signalling pathway were significantly differentially transcribed across the eight life stages of F. gigantica (Additional file 4, Table S2).
The presence of F. gigantica insulin signalling pathway components in ESPs
Among the 19 components of the IIS, 8 were predicted to be present in ESPs. While FgILP, FgIGFBP, and FgPTEN were predicted to be classic secretory proteins, FgIR-2, FgIR-3, FgDDL-1, FgHSF-1, and FgSKN-1 were predicted to be noncanonical secretory proteins.
Discussion
In this study, we constructed the IIS pathway, analysed the transcription profiles of the components, and predicted their presence in ESPs. Since the components of the IIS pathway have been poorly studied in trematodes, the functions of F. gigantica-derived components were speculated, and further studies are needed to verify their specific roles.
Multicellular communication is a key factor in the survival of parasitic helminths. The hormone host–worm cross-communication hypothesis suggests that the endocrine and paracrine hormone systems of mammals (even invertebrates) can affect the physiology, development, and survival of metazoan parasites by stimulating the evolutionarily conserved signalling system [25]. According to previous studies, the insulin signalling pathway is widely involved in the reproductive development of parasitic helminths and host–parasite interactions [26]. As an endocrine-related pathway, the insulin signalling pathway of F. gigantica may have intrinsic and extrinsic functions, and exploring this pathway will deepen the understanding of fluke development, as well as the interaction between flukes and hosts, to guide the prevention of fascioliasis and the treatment of metabolic diseases.
In S. japonicum, one ligand, SjILP, was detected. Immunohistochemistry has shown that SjILPs are abundantly expressed in the basement membranes and ovaries of adult worms [27]. Du et al. confirmed that SjILP can bind to ADP, which is the main agonist of platelet recruitment, indicating that SjILP functions in glucose metabolism [28]. In Taenia solium, two ILP sequences were cloned, and TsILP was found to be localised to the ovary, possibly related to reproduction [29]. Here, in F. gigantica, one ligand homologue, FgILP, was screened, and FgILP may also be localised to reproductive organs, participating in the reproductive development and glucose metabolism of flukes. Mammalian IGFBP can bind to ILP and regulate its bioavailability. In addition, IGFBP has multiple proteolytic enzyme binding sites and can be broken down into multiple fragments during metabolism, suggesting that its role is independent of the ILP molecular network [30, 31]. Similarly, FgIGFBP may also bind to FgILP and be involved in a role independent of the FgILP molecular network.
Two insulin receptors were characterised in both S. japonicum and E. multilocularis, and the two receptors differs in terms of structural motifs and tissue localisation. In terms of structure, SmIR-1 and SmIR-2 show 25.6% similarity, and the LBD and FN3 domains are greatly altered, indicating that they can be activated in different ways by binding to various ligands. SmIR-1 is localised to the muscle, intestinal epithelia, and basement membrane teguments of adult females and is also associated with the glucose transporters SGTP1 and SGTP4, suggesting that it participates in glucose uptake [32]. The structural characteristics of EmIR-1 and EmIR-2 are similar to those of SmIR-1 and SmIR-2. RT‒PCR analysis revealed that EmIR-1 is transcribed in both protoscoleces and echinococcosis, mainly in glycogen storage cells, indicating that EmIR-1 may be related to glucose metabolism [33]. The phylogenetic tree showed that FgIR-1 clustered on the same branch as SjIR-1 and EmIR-1. Therefore, FgIR-1 may be involved in glucose metabolism.
In addition to its intrinsic role, insulin-like signalling is involved in parasite–host communication. In S. japonicum, SjIR-2 can specifically bind to host insulin; further studies have suggested that S. japonicum can use host insulin for its growth and development and regulate cell proliferation and differentiation through the same pathway as host cells [34]. You et al. reported that genes related to the S. japonicum insulin signalling pathway, such as SjIR-2, were upregulated in the presence of human insulin [35]. After co-culturing schistosomes with host insulin and schistosomes for 24 h, microarray analysis showed that the transcription of two genes in the MAPK pathway was up-regulated, indicating that host insulin can act on the trematode insulin pathway and participate in the information exchange between trematodes and hosts in combination with the MAPK signaling pathway [36]. In mouse infection models of E. multilocularis, the addition of host insulin can promote the phosphorylation of EmIR-2 in vesicles, significantly altering the phosphorylation profile of PI3K/Akt signalling pathway components in vesicles and leading to the activation of downstream signalling pathways [37]. Although the E. multilocularis genome encodes ILP, yeast two-hybrid experiments revealed that EmIR-1 does not interact with EmILP but rather that the parasite-derived receptor EmIR-1 strongly binds to host proinsulin [38]. As F. gigantica is a multicellular organism, its parasitism may be regulated by signalling networks. FgIR-2 is predicted to be expressed in FgESP, and it may play a similar role as SjIR-2 and EmIR-2 in parasite–host interactions; these proteins may activate and amplify the downstream signals of the parasite by binding to the insulin of the host and promoting reproductive development. FgIR-3 was clustered on a separate branch independent of IR-1/IR-2 homologues, and its homologues were not found in other flukes, including E. multilocularis, S. japonicum, and S. mansoni. FgIR-3 may have evolved in Fasciola to adapt to parasitism, and further study of FgIR-3 is necessary to determine its role in the development and parasitism of F. gigantica.
FgIRS, FgPI3K, FgAPP, FgAKT-1, FgAKT-2, Fg14-3-3-ζ, and FgDDL-1 were also screened as cytoplasmic signal transduction components. In S. mansoni, an AKT isoform has been characterised, and blocking SmAKT reduces male–female cohesion and decreases the oviposition rate [39]. FgAKT-1 and FgAKT-2 may also participate in the reproductive process.
Two regulatory molecules involved in the insulin signalling pathway were screened in F. gigantica. The homologues of CeDAF-18 and FgPTEN were analysed. In C. elegans, DAF-18 controls dauer development and lifespan by inhibiting the phosphorylation of PIP3, and DAF-18 also acts as a negative regulator of DAF-2 and AGE-1 [40, 41]. Similarly, FgPTEN may act as an antagonist to promote the longevity of F. gigantica. Moreover, PPTR-1 acts as an inhibitor of the IIS pathway in C. elegans, and its homologues in humans and Drosophila can restrain the activation of AKT [42]. As a homologue of CePPTR-1, FgPP2A may also inhibit insulin signalling transduction by blocking AKT phosphorylation.
In F. gigantica, three transcription factors, FgFOXO, FgSKN-1, and FgHSF-1, were identified. Forkhead box O (FOXO) proteins constitute a subfamily of transcription factors that are conserved across invertebrates and mammals, and studies have consistently shown that FOXO proteins are important determinants of ageing and longevity. In C. elegans, FOXO is closely related to the stress response and cell cycle [43, 44]. In Drosophila, the upregulation of dFOXO in the bodies of adults is sufficient to promote longevity and antioxidative stress [45]. Here, FgFOXO may also play roles in the ageing and longevity in F. gigantica. SKN-1 mediates the transcription of genes involved in detoxification and stress responses [46] and can also promote protein homeostasis by regulating proteasome production [47], which helps to extend lifespan [48]. FgSKN-1 may also regulate the detoxification and stress response of F. gigantica. HSF-1 is a conserved regulator of heat-shock-induced gene transcription and regulates the overall stress response and stress resistance. In C. elegans, HSF-1 deficiency accelerates tissue ageing and shortens the lifespan [49]. In addition, DDL-1/2 can negatively regulate the activity of HSF-1 and regulate the stress response [50]. Therefore, it can be speculated that FgSKN-1 may regulate the stress response and stress resistance of F. gigantica.
In C. elegans, the insulin signalling pathway is thought to play a role in dauer development. Further studies have demonstrated the similar function of this pathway in V clade parasitic nematodes; that is, the pathway promotes development from the iL3 stage, analogous to the dauer stage of C. elegans, to the parasitic stage [51]. In the present study, the B, C, and D group genes shown in Fig. 3 exhibited increased transcription in metacercariae. As metacercariae are a stage of flukes awaiting host development that are resistant to the external environment, which is analogous to the dauer stage of C. elegans, the insulin signalling pathway may promote the transition of metacercariae to the parasitic stage. However, this hypothesis needs further exploration.
Conclusion
The insulin signalling pathway of F. gigantica was reconstructed, and the transcription profiles of these genes at different developmental stages were studied. In addition, the presence of these components in ESPs was explored, which provides a reference for future research on fluke development and F. gigantica–host interactions and promotes the exploration of other signalling pathways involved in this neglected tropical pathogen.
Data availability
All data analysed during this study are included in this published article. Zhang XX, Cong W, Elsheikha HM, Liu GH, Ma JG, Huang WY, Zhao Q, Zhu XQ. De novo transcriptome sequencing and analysis of the juvenile and adult stages of Fasciola gigantica. Infect Genet Evol. 2017.
References
Guo S. Insulin signaling, resistance, and the metabolic syndrome: insights from mouse models into disease mechanisms. J Endocrinol. 2014;8(2):T1–23.
Kenyon CJ. The genetics of ageing. Nature. 2010;464:504–12.
Li Y, Lu Y, Lin SH, Li N, Han Y, Huang Q, Zhao Y, Xie F, Guo Y, Deng B, Tsun A, Du J, Li D, Sun J, Shi G, Zheng F, Su X, Duan S, Zheng SG, Wang G, Tong X, Li B. Insulin signaling establishes a developmental trajectory of adipose regulatory T cells. Nat Immunol. 2021;22(9):1175–85.
Posner BI. Insulin signalling: the Inside Story. Can J Diabetes. 2017;41(1):108–13.
Fontana L, Partridge L, Longo VD. Extending healthy life span-from yeast to humans. Science. 2010;328:321–6.
Lee Y, An S, Artan M et al. Genes and Pathways That Influence Longevity in Caenorhabditis elegans; in Aging Mechanisms, Mori N and Mook-Jung I, editors.2015; 123–169.
Morris JZ, Tissenbaum HA, Ruvkun G. A phosphatidylinositol-3-OH kinase family member regulating longevity and diapause in Caenorhabditis elegans. Nature. 1996;382:536–9.
Kimura KD, Tissenbaum HA, Liu Y, Ruvkun G. daf-2, an insulin receptor-like gene that regulates longevity and diappause in Caenorhabditis elegans. Science. 1991;277:942–6.
Thomas JH, Birnby DA, Vowels JJ. Evidence for parallel processing of sensory information controlling dauer formation in Caenorhabditis elegans. Genetics. 1993;134(4):1105–17.
Vowels JJ, Thomas JH. Genetic analysis of chemosensory control of dauer formation in Caenorhabditis elegans. Genetics. 1992;130(1):105–23.
Wang S, Luo X, Zhang S, Yin C, Dou Y, Cai X. Identification of putative insulin-like peptides and components of insulin signaling pathways in parasitic platyhelminths by the use of genome-wide screening. FEBS J. 2014;281(3):877–93.
Camaya I, Donnelly S, O’Brien B. Targeting the PI3K/Akt signaling pathway in pancreatic β-cells to enhance their survival and function: an emerging therapeutic strategy for type 1 diabetes. J Diabetes. 2022;14(4):247–60.
Luckhart S, Riehle MA. The insulin signaling cascade from nematodes to mammals: insights into innate immunity of Anopheles mosquitoes to malaria parasite infection. Dev Comp Immunol. 2007;31(7):647–56.
Nielsen H, Brunak S, von Heijne G. Machine learning approaches for the prediction of signal peptides and other protein sorting signals. Protein Eng. 1999;12(1):3–9.
Hallgren J, Tsirigos KD, Damgaard Pedersen M, Juan J, Armenteros A, Marcatili P, Nielsen H, Krogh A, Winther O. DeepTMHMM predicts alpha and beta transmembrane proteins using deep neural networks. BioRxiv. 2022;04:08.
Naga D, Muster W, Musvasva E, Ecker GF. Off-targetp ML: an open source machine learning framework for off-target panel safety assessment of small molecules. J Cheminform. 2022;7(1):27.
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S. MEGA6: Molecular Evolutionary Genetics Analysis version 6.0. Mol Biol Evol. 2013;30(12):2725–9.
Zhang XX, Cong W, Elsheikha HM, Liu GH, Ma JG, Huang WY, Zhao Q, Zhu XQ. De novo transcriptome sequencing and analysis of the juvenile and adult stages of Fasciola gigantica. Infect Genet Evol. 2017;51:33–40.
Trapnell C, Hendrickson DG, Sauvageau M, Goff L, Rinn JL, Pachter L. Differential analysis of gene regulation at transcript resolution with RNA-seq. Nat Biotechnol. 2013;31(1):46–53.
Anders S, Huber W. Differential expression analysis for sequence count data. Genome Biol. 2010;11(10):R106.
Petersen TN, Brunak S, von Heijne G, Nielsen H. SignalP 4.0:discriminating signal peptides from transmembrane regions. Nat Methods. 2011;29(10):785–6.
Emanuelsson O, Nielsen H, Brunak S, von Heijne G. Predicting subcellular localization of proteins based on their N-terminal amino acid sequence. J Mol Biol. 2000;21(4):1005–16.
Krogh A, Winther O. DeepTMHMM predicts alpha and beta transmembrane proteins using deep neural networks. BioRxiv. 2022;04:08.
Bendtsen JD, Jensen LJ, Blom N, Von Heijne G, Brunak S. Feature-based prediction of non-classical and leaderless protein secretion. Protein Eng Des Sel. 2004;17(4):349–56.
Brehm K. Echinococcus multilocularis as an experimental model in stem cell research and molecular host-parasite interaction. Parasitology. 2010;137:537–55.
Jeong DE, Artan M, Seo K, Lee SJ. (2012) Regulation of lifespan by chemosensory and thermosensory systems: findings in invertebrates and their implications in mammalian aging. Front Genet. 2012 3, 218 199.
Du X, McManus DP, Cai P, Hu W, You H. Identification and functional characterisation of a Schistosoma japonicum insulin-like peptide. Parasit Vectors. 2017;14(1):181.
Du X, Jones MK, Nawaratna SSK, Ranasinghe S, Xiong C, Cai P, McManus DP, You H. Gene expression in Developmental stages of Schistosoma Japonicum provides further insight into the importance of the Schistosome insulin-like peptide. Int J Mol Sci. 2019;28(7):1565.
Wei YL, Guo AJ, Zhang SH, Liu GX, Hou JL, Luo XN. Identification of Taenia solium insulin receptor TsIR-1316 and expression of its ligand binding domain. Zhongguo Ji Sheng Chong Xue Yu Ji Sheng Chong Bing Za Zhi. 2016;34(2):93–8.
Li T, Forbes ME, Fuller GN, Li J, Yang X, Zhang W. IGFBP2: integrative hub of developmental and oncogenic signaling network. Oncogene. 2020;39(11):2243–57.
Bach LA, Headey SJ, Norton RS. IGF-binding proteins–the pieces are falling into place. Trends Endocrinol Metab. 2005;16(5):228–34.
Khayath N, Vicogne J, Ahier A, BenYounes A, Konrad C, Trolet J, Viscogliosi E, Brehm K, Dissous C. Diversification of the insulin receptor family in the helminth parasite Schistosoma mansoni. FEBS J. 2007;274(3):659–76.
Tsai IJ, Zarowiecki M, Holroyd N, Garciarrubio A, Sanchez-Flores A, Brooks KL, Tracey A, Bobes RJ, Fragoso G, Sciutto E, Aslett M, Beasley H, Bennett HM, Cai J, Camicia F, Clark R, Cucher M, De Silva N, Day TA, Deplazes P, Estrada K, Fernández C, Holland PW, Hou J, Hu S, Huckvale T, Hung SS, Kamenetzky L, Keane JA, Kiss F et al. The genomes of four tapeworm species reveal adaptations to parasitism. Nature.2013;496:57–63.
You H, Zhang W, Jones MK, Gobert GN, Mulvenna J, Rees G, Spanevello M, Blair D, Duke M, Brehm K, McManus DP. Cloning and characterisation of Schistosoma japonicum insulin receptors. PLoS ONE. 2010;24(3):e9868.
Wang L, Yang Z, Li Y, Yu F, Brindley PJ, McManus DP, Wei D, Han Z, Feng Z, Li Y, Hu W. Reconstruction and in silico analysis of the MAPK signaling pathways in the human blood fluke, Schistosoma Japonicum. FEBS Lett. 2006;580:3677–86.
You H, Zhang W, Moertel L, McManus DP, Gobert GN. Transcriptional profiles of adult male and female Schistosoma japonicum in response to insulin reveal increased expression of genes involved in growth and development. Int J Parasitol. 2009;39(14):1551–9.
Hemer S, Konrad C, Spiliotis M, Koziol U, Schaack D, Förster S, Gelmedin V, Stadelmann B, Dandekar T, Hemphill A, Brehm K. Host insulin stimulates Echinococcus Multilocularis insulin signalling pathways and larval development. BMC Biol. 2014;27:12:5.
Brehm K, Spiliotis M. The influence of host hormones and cytokines on Echinococcus multilocularis signalling and development. Parasite. 2008;15(3):286–90.
Morel M, Vanderstraete M, Cailliau K, Lescuyer A, Lancelot J, Dissous C. Compound libraryscreening identified Akt/PKB kinase pathway inhibitors as potential key molecules for thedevelopment of new chemotherapeutics against schistosomiasis. Iat J Parasitol Drugs DrugResist. 2014;4:256–66.
Ogg S, Ruvkun G. The C. Elegans PTEN homolog, DAF-18, acts in the insulin receptor-like metabolic signaling pathway. Mol Cell. 1998;2(6):887–93.
Liu J, Chin-Sang ID. C. Elegans as a model to study PTEN’s regulation and function. Methods. 2015;77 /78:180–90.
Rodríguez-Escudero I, Oliver MD, Andrés-Pons A, et al. A comprehensive functional analysis of PTEN mutations: implications in tumor- and autism-related syndromes. Hum Mol Genet. 2011;20(21):4132–42.
Martins R, Lithgow GJ, Link W. Long live FOXO: unraveling the role of FOXO proteins in aging and longevity. Aging Cell. 2016;15(2):196–207.
Libina N, Berman JR, Kenyon C. Tissue-specific activities of C. Elegans DAF-16 in the regulation of lifespan. Cell. 2003;115:489–502.
Hwangbo DS, Gershman B, Tu MP, Palmer M, Tatar M. Drosophila dFOXO controls lifespan and regulates insulin signalling in brain and fat body. Nature.2004;429, 562–566.
An JH, Vranas K, Lucke M, et al. Regulation of the Caenorhabditis elegans oxidative stress defense protein SKN-1 by glycogen synthase kinase-3. Proc Natl Acad Sci USA. 2005;102:16275–80.
Wang J, Robida-Stubbs S, Tullet JM, Rual JF, Vidal M, Blackwell TK. RNAi screening implicates a SKN-1-dependent transcriptional response in stress resistance and longevity deriving from translation inhibition. PLoS Genet. 2010;5(8):e1001048.
Ewald CY, Landis JN, Porter Abate J, Murphy CT, Blackwell TK. Dauer-independent insulin/IGF-1- signalling implicates collagen remodelling in longevity. Nature. 2015;519:97–101.
Brunquell J, Snyder A, Cheng F, Westerheide SD. HSF-1 is a regulator of miRNA expression in Caenorhabditis elegans. PLoS ONE. 2017;24(8):e0183445.
Chiang WC, Ching TT, Lee HC, et al. HSF-1 Regulators DDL-1 /2 link insulin-like signaling to heat-shock responses and modulation of. Longev Cell. 2012;148(1/2):322–34.
Altintas O, Park S, Lee SJ. The role of insulin/IGF-1 signaling in the longevity of model invertebrates, C. Elegans and D. Melanogaster. BMB Rep. 2016;49(2):81–92.
Acknowledgements
The authors thank the anonymous reviewers for their insightful suggestions and comments.
Funding
This work was supported by the Guangxi Natural Science Foundation (grant no. 2022GXNSFBA035583), which was awarded to Wenda Di.
Author information
Authors and Affiliations
Contributions
DW: Validation, Investigation, Writing Original Draft. YY, YY, LL, SF and LW: writing, review, editing, and investigation. TL, XL, WZ: Resources, Visualisation. WD: Conceptualisation, writing, editing, supervision, project administration, funding acquisition. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Wu, D., Yang, Y., Yang, Y. et al. An insulin-like signalling pathway model for Fasciola gigantica. BMC Vet Res 20, 252 (2024). https://doi.org/10.1186/s12917-024-04107-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12917-024-04107-7