Abstract
Background
The purpose of this study is to address concerns that expanded access may negatively impact the ultimate regulatory action and product labeling for new drugs.
Methods
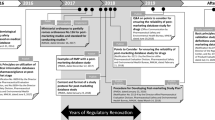
We performed queries of FDA’s Center for Drug Evaluation and Research (CDER) document tracking system to determine the effect of expanded access on FDA’s regulatory decision making from 2010 through 2016. We also examined product labeling to determine whether safety events occurring under expanded access had an adverse effect on the approved product labeling.
Results
There were 321 regulatory decisions made by FDA, with 28% of the drugs having prior expanded access. The approval rate for drugs with expanded access (84%) was higher than those that did not (76%). None of the negative regulatory marketing decisions were based on the adverse experiences reported under expanded access. The vast majority of deaths and serious adverse events that occurred under expanded access were not interpreted by FDA to be due to the investigational drug and did not affect product labeling. There was only 1 instance, a drug-drug interaction, for which safety events occurring during expanded access alone lead to potentially adverse product labeling.
Conclusions
There was no instance in which expanded access lead to a negative regulatory decision regarding a drug application, and there was only 1 instance that safety events under expanded access had a potentially negative effect on product labeling. Concern that expanded access will have a negative impact on drug development and review is not based on the evidence and is unwarranted.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Jarow JP, Lemery S, Bugin K, Khozin S, Moscicki R. Expanded access of investigational drugs: the experience of the Center of Drug Evaluation and Research over a 10-year period. Therapeutic Innovation & Regulatory Science. 2016;50:705–709.
Jarow JP, Lemery S, Bugin K, Lowy N. Ten-year experience for the Center for Drug Evaluation and Research, Part 2: FDA’s role in ensuring patient safety. Therapeutic Innovation & Regulatory Science. 2017;51:246–249.
Jacob JA. Questions of safety and fairness raised as right-to-try movement gains steam. JAMA. 2015;314:758–760.
Senior JR. Evolution of the Food and Drug Administration approach to liver safety assessment for new drugs: current status and challenges. Drug Saf. 2014;37(suppl 1):S9–S17.
Jarow JP, Casak S, Chuk M, Ehrlich LA, Khozin S. The majority of expedited investigational new drug safety reports are uninformative. Clin Cancer Res. 2016;22:2111–21113.
Investigational new drug safety reporting requirements for human drug and biological products and safety reporting requirements for bioavailability and bioequivalence studies in humans. Fed Regist. 2010;75:188.
Caplan AL, Ray A. The ethical challenges of compassionate use. JAMA. 2016;315:979–980.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jarow, J.P., Moscicki, R. Impact of Expanded Access on FDA Regulatory Action and Product Labeling. Ther Innov Regul Sci 51, 787–789 (2017). https://doi.org/10.1177/2168479017707800
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1177/2168479017707800