Abstract
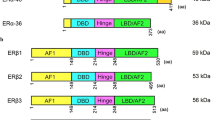
The molecular background of estrogen receptor (ER) expression is important to understand the pathophysiology of the high estrogen environment in endometriosis. However, the molecular details have not been fully understood. The objective of this study is to evaluate the molecular background of ERα and ERβ messenger RNA (mRNA) expression in endometriotic cells. The following summarizes our observations: (1) ERα mRNA expression in endometriotic cells was estimated to be approximately one-tenth of that in endometrial cells. (2) Three mRNAs, which include 3 different 5′-untranslated exons tagged to an open reading frame of wild-type ERα, were detected. (3) Expression of ERβ mRNA depends mostly on 0N promoter and includes 2 open reading frames: one for a wild-type ERβ1 and another for a splice variant ERβ2. (4) Expression of ERβ1 mRNA was approximately 40-fold higher than that in endometrial cells. (5) Expression of ERβ2 mRNA was almost at a comparable level of the ERβ1. 9 (6) ERα and ERβ mRNAs are equivalently expressed in endometriotic cells. These observations show the molecular background of ER mRNA expression in endometriotic cells and provide a clue to further understanding the estrogen-dependent pathophysiology leading to clinical application in endometriosis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Izawa M, Harada T, Taniguchi F, Ohama Y, Takenaka Y, Terakawa N. An epigenetic disorder may cause aberrant expression of aromatase gene in endometriotic stromal cells. Fertil Steril. 2008;89(suppl 5):1390–1396.
Izawa M, Taniguchi F, Uegaki T, et al. Demethylation of a non-promoter cytosine-phosphate-guanine island in the aromatase gene may cause the aberrant up-regulation in endometriotic tissues. Fertil Steril. 2011;95(1):33–39.
Olive DL, Pritts EA. Treatment of endometriosis. N Engl J Med. 2001;345(4):266–275.
Brandenberger AW, Lebovic DI, Tee MK, et al. Oestrogen receptor (ER)-alpha and ER-beta isoforms in normal endometrial and endometriosis-derived stromal cells. Mol Hum Reprod. 1999;5(7):651–655.
Fujimoto J, Hirose R, Sakaguchi H, Tamaya T. Expression of oestrogen receptor-alpha and -beta in ovarian endometriomata. Mol Hum Reprod. 1999;5(8):742–747.
Matsuzaki S, Fukaya T, Uehara S, Murakami T, Sasano H, Yajima A. Characterization of messenger RNA expression of estrogen receptor-alpha and -beta in patients with ovarian endometriosis. Fertil Steril. 2000;73(6):1219–1225.
Xue Q, Lin Z, Cheng YH, et al. Promoter methylation regulates estrogen receptor 2 in human endometrium and endometriosis. Biol Reprod. 2007;77(4):681–687.
Harris HA. The unexpected science of estrogen receptor-beta selective agonists: a new class of anti-inflammatory agents? Nucl Recept Signal. 2006;4:e012.
Xing D, Feng W, Miller AP, et al. Estrogen modulates TNF-alpha-induced inflammatory responses in rat aortic smooth muscle cells through estrogen receptor-beta activation. Am J Physiol Heart Circ Physiol. 2007;292(6):H2607–H2612.
Moore JT, McKee DD, Slentz-Kesler K, et al. Cloning and characterization of human estrogen receptor beta isoforms. Biochemical and Biophysical Research Communications. 1998;247(1):75–78.
Leung YK, Mak P, Hassan S, Ho SM. Estrogen receptor (ER)-beta isoforms: a key to understanding ER-beta signaling. PNAS. 2006;103(35):13162–13167.
Paulmurugan R, Tamrazi A, Massoud TF, Katzenellenbogen JA, Gambhir SS. In vitro and in vivo molecular imaging of estrogen receptor alpha and beta homo- and heterodimerization: exploration of new modes of receptor regulation. Mol Endocrinol. 2011;25(12):2029–2040.
Chakraborty S, Willett H, Biswas PK. Insight into estrogen receptor beta-beta and alpha-beta homo- and heterodimerization: a combined molecular dynamics and sequence analysis study. Biophys Chem. 2012;170:42–50.
Iwabe T, Harada T, Tsudo T, et al. Tumor necrosis factor-alpha promotes proliferation of endometriotic stromal cells by inducing interleukin-8 gene and protein expression. J Clin Endocrinol Metab. 2000;85(2):824–829.
Sakamoto Y, Harada T, Horie S, et al. Tumor necrosis factor-alpha-induced interleukin-8 (IL-8) expression in endometriotic stromal cells, probably through nuclear factor-kappa B activation: gonadotropin-releasing hormone agonist treatment reduced IL-8 expression. J Clin Endocrinol Metab. 2003;88(2):730–735.
Izawa M, Harada T, Deura I, Taniguchi F, Iwabe T, Terakawa N. Drug-induced apoptosis was markedly attenuated in endometriotic stromal cells. Hum Reprod. 2006;21(3):600–604.
Figtree GA, McDonald D, Watkins H, Channon KM. Truncated estrogen receptor alpha 46-kDa isoform in human endothelial cells: relationship to acute activation of nitric oxide synthase. Circulation. 2003;107(1):120–126.
Suzuki F, Akahira J, Miura I, et al. Loss of estrogen receptor b isoform expression and its correlation with aberrant DNA methylation of the 5’-untranslated region in human epithelial ovarian carcinoma. Cancer Sci. 2008;99(12):2365–2372.
Powell E, Shanle E, Brinkman A, et al. Identification of estrogen receptor dimer selective ligands reveals growth-inhibitory effects on cells that co-express ERalpha and ERbeta. PLoS One. 2012;7(2):e30993.
Lindberg MK, Moverare S, Skrtic S, et al. Estrogen receptor (ER)-beta reduces ERalpha-regulated gene transcription, supporting a ‘‘ying yang’’ relationship between ERalpha and ERbeta in mice. Mol Endocrinol. 2003;17(2):203–208.
Saji S, Hirose M, Toi M. Clinical significance of estrogen receptor beta in breast cancer. Cancer Chemother Pharmacol. 2005;56(suppl 1):21–26.
Pettersson K, Grandien K, Kuiper GG, Gustafsson JA. Mouse estrogen receptor beta forms estrogen response element-binding heterodimers with estrogen receptor alpha. Mol Endocrinol. 1997;11(10):1486–1496.
Tremblay GB, Tremblay A, Labrie F, Giguere V. Dominant activity of activation function 1 (AF-1) and differential stoichiometric requirements for AF-1 and -2 in the estrogen receptor alpha-beta heterodimeric complex. Mol Cell Biol. 1999;19(3):1919–1927.
Paterni I, Granchi C, Katzenellenbogen JA, Minutolo F. Estrogen receptors alpha (ERα) and beta (ERβ): subtype-selective ligands and clinical potential. Steroids. 2014;90:13–29.
Monsivais D, Dyson MT, Yin P, et al. ERb- and prostaglandin E2-regulated pathways integrate cell proliferation via Ras-like and estrogen-regulated growth inhibitor in endometriosis. Mol Endocrinol. 2014;28(8):1304–1315.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Izawa, M., Taniguchi, F. & Harada, T. Molecular Background of Estrogen Receptor Gene Expression in Endometriotic Cells. Reprod. Sci. 23, 871–876 (2016). https://doi.org/10.1177/1933719115623642
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719115623642