Abstract
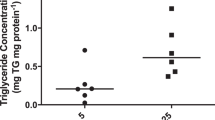
Placental metabolism is an important mechanism for the regulation of fetal growth and long-term health of the newborns. In this study, we investigated the effects of maternal metabolic environment on human placental fatty acid and glucose metabolism. We used placental explants from uncomplicated pregnancies or pregnancies complicated with gestational diabetes mellitus (GDM), undergoing vaginal delivery (VD) or cesarean section (CS). Fatty acid oxidation (FAO) and glucose uptake (2-DOG) were similar in both modes of delivery in normal and GDM pregnancies. However, placental explants from GDM exhibited 40% to 50% reduced FAO capacity compared to control placentas in women undergoing VD or CS. In contrast, 2-DOG uptake was 2- to 3-fold higher in placental explants from GDM compared to control placentas in women undergoing VD or CS, respectively. In conclusion, ex vivo placental fuel selection is influenced by maternal GDM, but placental metabolic characteristics are not altered by the mode of delivery.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Barbour LA. Unresolved controversies in gestational diabetes: implications on maternal and infant health. Curr Opin Endocrinol Diabetes Obes. 2014;21(4):264–270.
Catalano PM, Hauguel-De Mouzon S. Is it time to revisit the Pedersen hypothesis in the face of the obesity epidemic? Am J Obstet Gynecol. 2011; 204(6):479–487.
Burton GJ, Sebire NJ, Myatt L, et al. Optimising sample collection for placental research. Placenta. 2014;35(1):9–22.
Mendez-Figueroa H, Chien EK, Ji H, Nesbitt NL, Bharathi SS, Goetzman E. Effects of labor on placental fatty acid beta oxidation. J Matern Fetal Neonatal Med. 2012;26(2):150–154.
Visiedo F, Bugatto F, Sanchez V, Cozar-Castellano I, Bartha JL, Perdomo G. High glucose levels reduce fatty acid oxidation and increase triglyceride accumulation in human placenta. Am J Physiol Endocrinol Metab. 2013; 305(2):E205–E212.
Brar HS, Platt LD, DeVore GR, Horenstein J, Medearis AL. Qualitative assessment of maternal uterine and fetal umbilical artery blood flow and resistance in laboring patients by Doppler velocimetry. Am J Obstet Gynecol. 1988;158(4):952–956.
Fleischer A, Anyaegbunam AA, Schulman H, Farmakides G, Randolph G. Uterine and umbilical artery velocimetry during normal labor. Am J Obstet Gynecol. 1987;157(1):40–43.
Bartha JL, Visiedo F, Fernandez-Deudero A, Bugatto F, Perdomo G. Decreased mitochondrial fatty acid oxidation in placentas from women with preeclampsia. Placenta. 2012;33(2): 132–134.
Perdomo G, Martinez-Brocca MA, Bhatt BA, Brown NF, O’Doherty RM, Garcia-Ocana A. Hepatocyte growth factor is a novel stimulator of glucose uptake and metabolism in skeletal muscle cells. J Biol Chem. 2008;283(20):13700–13706.
Yung HW, Colleoni F, Atkinson D, et al. Influence of speed of sample processing on placental energetics and signalling pathways: implications for tissue collection. Placenta. 2014;35(2):103–108.
Di Santo S, Malek A, Sager R, Andres AC, Schneider H. Trophoblast viability in perfused term placental tissue and explant cultures limited to 7–24 hours. Placenta. 2003; 24(8–9):882–94.
Baumann MU, Schneider H, Malek A, et al. Regulation of human trophoblast GLUT1 glucose transporter by insulin-like growth factor I (IGF-I). PLoS One. 2014; 9(8):e106037.
Ramsay TG, Karousis J, White ME, Wol verton CK. Fatty acid metabolism by the porcine placenta. J Anim Sci. 1991;69(9):3645–3654.
Jansson T, Wennergren M, Powell TL. Placental glucose transport and GLUT 1 expression in insulin-dependent diabetes. Am J Obstet Gynecol. 1999;180(1 pt 1):163–168.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Visiedo, F., Bugatto, F., Quintero-Prado, R. et al. Glucose and Fatty Acid Metabolism in Placental Explants From Pregnancies Complicated With Gestational Diabetes Mellitus. Reprod. Sci. 22, 798–801 (2015). https://doi.org/10.1177/1933719114561558
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719114561558