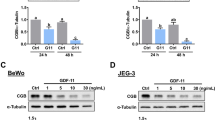
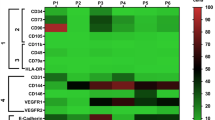
Abstract
Embryo implantation, which is an absolute requirement for reproduction, starts with blastocyst apposition to the uterine endometrium, followed by attachment to the endometrial surface epithelium. Recent clinical studies reported an increase in implantation and pregnancy rates among women receiving intrauterine human chorionic gonadotropin (hCG) prior to embryo transfer suggesting that, at least in some cases, female infertility is a result of inadequate secretion of hCG. In this study, we characterized the effect of hCG on trophoblast–epithelial interaction by further developing our recently described in vitro model of implantation. Here, we confirmed hCG increased attachment of trophoblast to epithelial cells, using a single-cell trophoblast–epithelial coculture system in addition to a blastocyst-like spheroid–epithelial coculture system. Furthermore, we discovered that the source and concentration was pivotal; the first preparation of hCG affected 2 molecules related to implantation, MUC16 and osteopontin, while the second preparation required additional cytokines to mimic the effects. Using this system, we can develop a comprehensive knowledge of the cellular and gene targets of hCG and other factors involved in embryo apposition and implantation and potentially increase the number of therapeutic targets for subfertile patients.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Psychoyos A. Uterine receptivity for nidation. Ann N Y Acad Sci. 1986;476:36–42.
Norwitz ER, Schust DJ, Fisher SJ. Implantation and the survival of early pregnancy. N Engl J Med. 2001;345(19):1400–1408.
Mansour R, Tawab N, Kamal O, et al. Intrauterine injection of human chorionic gonadotropin before embryo transfer significantly improves the implantation and pregnancy rates in in vitro fertilization/intracytoplasmic sperm injection: a prospective randomized study. Fertil Steril. 2011;96(6):1370–1374.e1371.
Keay SD, Vatish M, Karteris E, Hillhouse EW, Randeva HS. The role of hCG in reproductive medicine. BJOG. 2004;111(11):1218–1228.
Reisinger K, Baal N, McKinnon T, Munstedt K, Zygmunt M. The gonadotropins: tissue-specific angiogenic factors? Mol Cell Endocrinol. 2007;269(1–2):65–80.
Licht P, Fluhr H, Neuwinger J, Wallwiener D, Wildt L. Is human chorionic gonadotropin directly involved in the regulation of human implantation? Mol Cell Endocrinol. 2007;269(1–2):85–92.
Kane N, Kelly R, Saunders PT, Critchley HO. Proliferation of uterine natural killer cells is induced by human chorionic gonadotropin and mediated via the mannose receptor. Endocrinology. 2009;150(6):2882–2888.
Kosaka K, Fujiwara H, Tatsumi K, et al. Human chorionic gonadotropin (HCG) activates monocytes to produce interleukin-8 via a different pathway from luteinizing hormone/HCG receptor system. J Clin Endocrinol Metabol. 2002;87(11):5199–5208.
Schumacher A, Brachwitz N, Sohr S, et al. Human chorionic gonadotropin attracts regulatory T cells into the fetal-maternal interface during early human pregnancy. J Immunol. 2009;182(9):5488–5497.
Wan H, Versnel MA, Cheung WY, et al. Chorionic gonadotropin can enhance innate immunity by stimulating macrophage function. J Leukoc Biol. 2007;82(4):926–933.
Wan H, Versnel MA, Leijten LM, et al. Chorionic gonadotropin induces dendritic cells to express a tolerogenic phenotype. J Leukoc Biol. 2008;83(4):894–901.
Kornyei JL, Lei ZM, Rao CV. Human myometrial smooth muscle cells are novel targets of direct regulation by human chorionic gonadotropin. Biol Reprod. 1993;49(6):1149–1157.
Sherwin JR, Sharkey AM, Cameo P, et al. Identification of novel genes regulated by chorionic gonadotropin in baboon endometrium during the window of implantation. Endocrinology. 2007;148(2):618–626.
Perrier d’Hauterive S, Charlet-Renard C, Berndt S, et al. Human chorionic gonadotropin and growth factors at the embryonicendometrial interface control leukemia inhibitory factor (LIF) and interleukin 6 (IL-6) secretion by human endometrial epithelium. Hum Reprod. 2004;19(11):2633–2643.
Perrier d’Hauterive S, Charlet-Renard C, Dubois M, et al. Human endometrial leukemia inhibitory factor and interleukin-6: control of secretion by transforming growth factor-beta-related members. Neuroimmunomodulation. 2005;12(3):157–163.
Sugihara K, Kabir-Salmani M, Byrne J, et al. Induction of trophinin in human endometrial surface epithelia by CGbeta and IL-1beta. FEBS Lett. 2008;582(2):197–202.
Meseguer M, Aplin JD, Caballero-Campo P, et al. Human endometrial mucin MUC1 is up-regulated by progesterone and downregulated in vitro by the human blastocyst. Biol Reprod. 2001; 64(2): 590–601.
Gipson IK, Blalock T, Tisdale A, et al. MUC16 is lost from the uterodome (pinopode) surface of the receptive human endometrium: in vitro evidence that MUC16 is a barrier to Trophoblast adherence. Biol Reprod. 2008;78(1):134–142.
Johnson GA, Burghardt RC, Bazer FW, Spencer TE. Osteopontin: roles in implantation and placentation. Biol Reprod. 2003;69(5):1458–1471.
Holmberg JC, Haddad S, Wunsche V, et al. An in vitro model for the study of human implantation. Am J Reprod Immunol. 2012;67(2):169–178.
Straszewski-Chavez SL, Abrahams VM, Alvero AB, et al. The isolation and characterization of a novel telomerase immortalized first trimester trophoblast cell line, Swan 71. Placenta. 2009;30(11):939–948.
Schumacher A, Heinze K, Witte J, et al. Human chorionic gonadotropin as a central regulator of pregnancy immune tolerance. J Immunol. 2013;190(6):2650–2658.
Jensen F, Wallukat G, Herse F, et al. CD19+CD5+ cells as indicators of preeclampsia. Hypertension. 2012;59(4):861–868.
Birken S, Kovalevskaya G, O’Connor J. Immunochemical measurement of early pregnancy isoforms of HCG: potential applications to fertility research, prenatal diagnosis, and cancer. Arch Med Res. 2001;32(6):635–643.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Racicot, K.E., Wünsche, V., Auerbach, B. et al. Human Chorionic Gonadotropin Enhances Trophoblast–Epithelial Interaction in an In Vitro Model of Human Implantation. Reprod. Sci. 21, 1274–1280 (2014). https://doi.org/10.1177/1933719114522553
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719114522553