Abstract
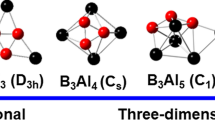
The structures and properties of binary boron-aluminum B5Aln0/-/+ (n = 1–4) clusters have been systematically explored using the density functional theory method at the B3LYP/6–311+G(d) level and the coupled cluster method at the CCSD(T)/6–311+G(2df)//B3LYP/6–311+G(d) level. Lowest-energy structures, stabilities, growth behaviors and chemical bonding of these clusters were analyzed. Our results show that when the number of doped Al atoms is one or two, the Al atoms are located at the periphery, and the host B5 cluster preferentially forms a W-shape core, which is only slightly affected by the Al atoms. When there are three or four Al atoms, the B5Aln0/-/+ (n = 3, 4) clusters have their lowest energy structures preferentially in capped bipyramid configurations. Neutral B5Aln (n = 1, 3) clusters are somewhat more stable than their neighboring n clusters, while anionic and cationic B5Aln-/+ (n = 2, 4) clusters tend to be somewhat more stable. We also simulated the infrared (IR) spectrum and photoelectron spectroscopy (PES) of these clusters for future experimental comparison. Adaptive natural density partitioning (AdNDP) analysis shows that a variety of delocalized multicenter bonds appear in these clusters, which may enhance the stability of the clusters.
Graphical abstract

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
B. Qi, C. Wu, X. Li, D. Wang, L. Sun, B. Chen, W. Liu, H. Zhang, X. Zhou, ChemCatChem 10, 2285 (2018).
D. Tu, S. Cai, C. Fernandez, H. Ma, X. Wang, H. Wang, C. Ma, H. Yan, C. Lu, Z. An, Angew. Chem. Int. Ed. 58, 9129 (2019).
W.L. Li, X. Chen, T. Jian, T.T. Chen, J. Li, L.S. Wang, Nat. Rev. Chem. 1, 0071 (2017).
L.S. Wang, Int. Rev. Phys. Chem. 35, 69 (2016).
H.-J. Zhai, B. Kiran, J. Li, L.S. Wang, Nat. Mater. 2, 827 (2003).
A.N. Alexandrova, A.I. Boldyrev, H.-J. Zhai, L.S. Wang, Coord. Chem. Rev. 250, 2811 (2006).
J. Warneke, G.-L. Hou, E. Aprà, C. Jenne, Z. Yang, Z. Qin, K. Kowalski, X.-B. Wang, S.S. Xantheas, J. Am. Chem. Soc. 139, 14749 (2017).
J. Warneke, S.Z. Konieczka, G.-L. Hou, E. Aprà, C. Kerpen, F. Keppner, T.C. Schäfer, M. Deckert, Z. Yang, E.J. Bylaska, G.E. Johnson, J. Laskin, S.S. Xantheas, X.-B. Wang, M. Finze, Phys. Chem. Chem. Phys. 21, 5903 (2019).
M. Mayer, V. van Lessen, M. Rohdenburg, G.L. Hou, Z. Yang, R.M. Exner, E. Apra, V.A. Azov, S. Grabowsky, S.S. Xantheas, K.R. Asmis, X.B. Wang, C. Jenne, J. Warneke, Proc. Natl. Acad. Sci. USA 116, 8167 (2019).
M. Rohdenburg, Z. Yang, P. Su, E. Bernhardt, Q. Yuan, E. Apra, S. Grabowsky, J. Laskin, C. Jenne, X.-B. Wang, J. Warneke, Phys. Chem. Chem. Phys. 22, 17713 (2020).
E. Apra, J. Warneke, S.S. Xantheas, X.B. Wang, J. Chem. Phys. 150, 164306 (2019).
N.M. Tam, L.V. Duong, H.T. Pham, M.T. Nguyen, M.P. Pham-Ho, Phys. Chem. Chem. Phys. 21, 8365 (2019).
A. Kumar, N. Vyas, A.K. Ojha, Int. J. Hydrogen Energy 45, 12961 (2020).
D. Kang, W. Sun, H. Shi, C. Lu, X. Kuang, B. Chen, X. Xia, G. Maroulis, Sci. Rep. 9, 14367 (2019).
C. Romanescu, A.P. Sergeeva, W.-L. Li, A.I. Boldyrev, L.-S. Wang, J. Am. Chem. Soc. 133, 8646 (2011).
W.-L. Li, C. Romanescu, T.R. Galeev, L.-S. Wang, A.I. Boldyrev, J. Phys. Chem. A 115, 10391 (2011).
T.R. Galeev, C. Romanescu, W.-L. Li, L.-S. Wang, A.I. Boldyrev, J. Chem. Phys. 135, 104301 (2011).
B. Song, Y. Zhou, H.-M. Yang, J.-H. Liao, L.-M. Yang, X.-B. Yang, E. Ganz, J. Am. Chem. Soc. 141, 3630 (2019).
L. Wen, G. Li, L.-M. Yang, H. Pan, E. Ganz, Mater. Today Commun. 24, 100914 (2020).
A.P. Sergeeva, B.B. Averkiev, H.J. Zhai, A.I. Boldyrev, L.-S. Wang, J. Chem. Phys. 134, 224304 (2011).
A.D. Becke, J. Chem. Phys. 98, 5648 (1993).
C. Lee, W.T. Yang, R.G. Parr, Phys. Rev. B 37, 785 (1988).
J.S. Binkley, J.A. Pople, W.J. Hehre, J. Am. Chem. Soc. 102, 939 (1980).
M.S. Gordon, J.S. Binkley, J.A. Pople, W.J. Pietro, W.J. Hehre, J. Am. Chem. Soc. 104, 2797 (1982).
G.D. Purvis, R.J. Bartlett, J. Chem. Phys. 76, 1910 (1982).
D.Y. Zubarev, A.I. Boldyrev, Phys. Chem. Chem. Phys. 10, 5207 (2008).
D.Y. Zubarev, A.I. Boldyrev, J. Org. Chem. 73, 9251 (2008).
D.Y. Zubarev, A.I. Boldyrev, J. Phys. Chem. A 113, 866 (2009).
D.Y. Zubarev, N. Robertson, D. Domin, J. McClean, J. Wang, W.A. Lester Jr, R. Whitesides, X. You, M.J. Frenklach, Phys. Chem. C 114, 5429 (2010).
M. Frisch, G. Trucks, H. Schlegel, G. Scuseria, M. Robb, J. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. Petersson, Gaussian 09, Revision A. 02 (Gaussian Inc., Wallingford, CT, 2009).
T. Lu, F. Chen, J. Comput. Chem. 33, 580 (2012).
Q.S. Li, H.W. Jin, J. Phys. Chem. A 106, 7042 (2002).
X.-J. Feng, Y.-H. Luo, J. Phys. Chem. A 111, 2420 (2007).
M. Böyükata, Z.B. Güvenç, J. Alloys Compd. 509, 42144234 (2011).
P. Pyykkö, J. Phys. Chem. A 119, 2326 (2015).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supplementary material in the form of one pdf file available from the Journal web page at https://doi.org/10.1140/epjd/e2020-10409-8.
Electronic supplementary material
Suplementary material
Rights and permissions
About this article
Cite this article
Wen, L., Li, G., Yang, LM. et al. Structural evolution in boron-based clusters B5Aln0/-/+ (n = 1–4): Al atoms transition from the periphery of the planar W-shaped B5 ring to the vertex of the bipyramid. Eur. Phys. J. D 74, 223 (2020). https://doi.org/10.1140/epjd/e2020-10409-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1140/epjd/e2020-10409-8