Abstract
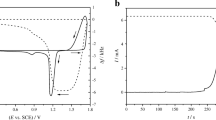
The effect of the concentration of sodium hydroxide with and without potassium oleate on electrochemical behavior of lead has been investigated using cyclic voltammetry. It has been shown that the number of anode and cathode peaks on the curve depends on the potential scan rate and the concentration of alkali. Anode peaks are related to the formation of lead compounds (PbO and PbO2), the presence of which has been confirmed by X-ray phase analysis. It has been determined that the dissolution of lead in sodiumhydroxide solution with and without potassium oleate proceeds under diffusion-kinetic control. Potassium oleate facilitates lead passivation in the solutions with lower concentration of alkali decreasing the potential and current of active–passive transition and does not affect or restricts it in more concentrated solutions.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Nguen, D.K. and Sukhotin, A.V., Zh. Prikl. Khim., 1990, vol. 63, no. 8, pp. 1821–1824.
Nguen, D.K. and Sukhotin, A.V., Zashch. Met., 1988, vol. 24, no. 5, pp. 822–826.
Nguen, D.K. and Sukhotin, A.V., Zh. Prikl. Khim., 1990, vol. 63, no. 8, pp. 1824–1828.
Berezhnaya, A.G., Ogarev, P.I., and Ekilik, V.V., Korroz.: Mater., Zashch., 2012, no. 2, pp. 16–21.
Ekilik, V.V., Tikhomirova, K.S., and Berezhnaya, A.G., Kondens. Sredy Mezhfaznye Granitsy, 2012, vol. 14, no. 1, pp. 104–113.
Berezhnaya, A.G. and Kaz’mina, M.A., Korroz.: Mater., Zashch., 2016, no. 2, pp. 32–40.
Galus, Z., Fundamentals of Electrochemical Analysis, Chichester: Ellis Horwood, 1976.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.G. Berezhnaya, M.A. Kaz’mina, V.A. Volochaev, 2016, published in Korroziya: Materialy, Zashchita, 2016, No. 8, pp. 11–16.
Rights and permissions
About this article
Cite this article
Berezhnaya, A.G., Kaz’mina, M.A. & Volochaev, V.A. Evaluation of the Effect of the Concentration of Sodium Hydroxide with and without Potassium Oleate on the Behavior of Lead by Cyclic Voltammetry. Prot Met Phys Chem Surf 53, 1193–1198 (2017). https://doi.org/10.1134/S207020511707005X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S207020511707005X