Abstract
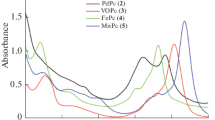
The effect of the nature of the metal in metal phthalocyanites on the electrokinetic potential and рН of the isoelectric point (рН(I)) in suspensions of these substances in NaCl solution (0.1 mol/L) is elucidated. The рН(I) values of unmodified МРс increases in a sequence MnPc < CoPc < СuPc ≈ ZnPc on account of the decreasing effective charge of the coordinated metal ion that corresponds to the Irving–Williams series. The impact of the functional group upon heterogeneous modification of МРс nanoparticles is studied, as well. It is established that the modifying group (the oxygenic properties) exerts a stronger influence on (pH(I)) than the metal nature.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Pashkovskaya, A.A., Maizlish, V.E., Kotova, E.A., et al, Biochim. Biophys. Acta, 2008, vol. 1778, no. 2, p. 541.
Strakhovskaya, M.G., Antonenko, Y.N., Pashkovskaya, A.A., Kotova E.A., Kireev, V., et al, Biochemistry (Moscow), 2009, vol. 74, no. 12, p. 1305.
Strakhovskaya, M.G., et al, Klin. Prakt., 2013, no. 1, p. 25.
Khafizov, N.R. and Saifullin, R.S., in Prikladnaya elektrokhimiya (Applied Electrochemistry), Kazan, 1988, p. 120.
Golubchikov, O.A., Larionov, A.V., Maizlish, V.E., and Balmasov, A.V, Izv. Vyssh. Uchebn. Zaved., Khim. Khim. Tekhnol., 2014, vol. 57, no. 12, p. 60.
Golubchikov, O.A., Larionov, A.V., Balmasov, A.V., and Semeikin, A.S, Makrogeterotsikly, 2014, vol. 7, p. 225.
Vinokurov, E.G., Arsenkin, A.M., Grigorovich, K.V., and Bondar’, V.V, Prot. Met. Phys. Chem. Surf., 2006, vol. 42, no. 2, p. 204.
Dong, J., Corti, D.S., and Franses, E.I, Langmuir, 2010, vol. 26, p. 6995.
Grishina, A.D., Gorbunova, Yu.G., Zolotarevsky, V.I., et al., J. Porphyrins Phthalocyanines, 2009, vol. 13, no. 1, p. 92.
Jenkins, P., Basu, S., Keir, R.I., et al., J. Colloid Interface Sci., 1999, vol. 211, p. 252.
Linstead, R.P, J. Chem. Soc., 1934, p. 1016.
Bao, Z., Lovinger, A.J., and Brown, J, J. Am. Chem. Soc., 1998, vol. 120, no. 1, p. 207.
Hesse, K. and Schlettwein, D, J. Electroanal. Chem., 1999, vol. 476, p. 148.
Zuev, K.V., Smrchek, V.A., Fedoseeva, M.S., et al, Khim. Prom-st. Segodnya, 2015, no. 8, p. 24.
http://www.niic.nsc.ru/images/document/000747/dissertation_ semushkina_g.i..pdf. Accessed January 23, 2015.
Tarasevich, M.R., Radyushkina, K.A., and Bogdanovskaya, V.A., in Elektrokhimiya porfirinov (Electrochemistry of Porphyrins), Moscow: Nauka, 1991, p. 37.
Irving, H. and Williams, R.J.P, Nature, 1948, vol. 162, p. 746.
Miličević, A., Branica, G., and Raos, N, Molecules, 2011, vol. 16, p. 1103.
Riddick, J.A., Bunger, W.B., and Sakano, Th.K., Techniques of Chemistry, New York: John Wiley & Sons, 1986, vol.2.
Dean, J.A., Handbook of Organic Chemistry, New York: McGraw-Hill, 1987, p. 8.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © E.G. Vinokurov, A.G. Muradova, K.V. Zuev, F.N. Zhigunov, V.P. Perevalov, 2017, published in Fizikokhimiya Poverkhnosti i Zashchita Materialov, 2017, Vol. 53, No. 5, pp. 521–524.
Rights and permissions
About this article
Cite this article
Vinokurov, E.G., Muradova, A.G., Zuev, K.V. et al. Electrokinetic properties of metal phthalocyanite nanoparticles. Prot Met Phys Chem Surf 53, 847–849 (2017). https://doi.org/10.1134/S2070205117050264
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2070205117050264