Abstract
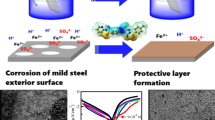
The corrosion behaviour of mild steel immersed in 0.5 M HCl in the absence and presence of 4-hydroxy-N'-[(1E, 2E)-3-phenylprop-2-en-1-ylidene] benzohydrazide (HPB) was evaluated using Tafel polarization and electrochemical impedance spectroscopy techniques. The effect of inhibitor concentration and temperature on the corrosion inhibition efficiency (% IE) of HPB was studied and discussed. It is observed that the inhibition efficiency increased with increase in inhibitor concentration and also with increase in temperature for a give inhibitor concentration. The polarization study revealed that HPB acts as a mixed type of inhibitor by inhibiting both anodic and cathodic reactions. The corrosion inhibition process follows Langmuir-adsorption isotherm and takes place predominantly through chemisorption.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Vishwanatham, S. and Haldar, N., Corros. Sci., 2008, vol. 50, p. 2999.
Bothi, R.P. and Sethuraman, M.G., Mater. Lett., 2008, vol. 62, p. 113.
Satapathy, A.K., Gunasekaran, G., Sahoo, S.C., et al., Corros. Sci., 2009, vol. 51, p. 2848.
Emregal, C.K. and Mustfa, H., Corros. Sci., 2006, vol. 48, p. 797.
Khaled, K.F., Babic-Samradzija, K., and Hackerman, N., Electrochim. Acta, 2005, vol. 50, p. 2515.
Roberge, P.R., Corrosion Inhibitors: Handbook of Corrosion Engineering, New York: McGraw-Hill, 1999.
Quartarone, G., Battilana, M., Bonaldo, L., et al., Corros. Sci., 2008, vol. 50, p. 3467.
Haksar, C.N., Malhotra, R.C., Pandya, G., et al., J. Sci. Technol. B, 1971, vol. 9, p. 34.
Kraatz, L.U., Wolfgang, B.K., Wolfram, B.G.A., et al. US Patent 5929118, 1999.
Quraishi, M.A., Sardar, R., and Jamal, D., Mater. Chem. Phys., 2001, vol. 71, p. 309.
Shanbhag, A.V., Venkatesha, T.V., Prabhu, R., et al., J. Appl. Electrochem., 2008, vol. 38, p. 279.
Renata, B.O., Elaine, M.S.F., and Rodrigo, P.P.S., Eur. J. of Med. Chem., 2008, vol. 43, p. 1984.
Flis, J. and Zakroczymski, T., J. Electrochem. Soc., 1996, vol. 143, p. 2458.
Rammelt, U., Kohler, S., and Reinhard, G., Electrochim. Acta, 2008, vol. 53, p. 6968.
Hsu, C.H. and Mansfeld, F., Corrosion, 2001, vol. 57, p. 747.
Larabi, L., Benali, O., Mekelleche, S., et al., Appl. Surf. Sci., 2006, vol. 253, p. 1371.
McCafferty, E. and Hackerman, N., J. Electrochem. Soc., 1972, vol. 119, p. 146.
Muralidharan, S., Phani, K.L., Pitchumani, S., et al., J. Electrochem. Soc., 1995, vol. 142, p. 1478.
Fontana, M.G., Corrosion Engineering, Singapore: McGraw-Hill, 1987, 3rd ed.
Rafique, M.Z.A., Saxena, N., Khan, S., et al., Indian J. Chem. Technol., 2007, vol. 14576.
Wang, L., Shinohara, T., Zhang, B., et al., J. Alloys Compd., 2010, vol. 496, p. 500.
Ferreira, E.S., Giancomelli, C., Giacomelli, F.C., et al., Mater. Chem. Phys., 2004, vol. 83, p. 129.
Shrier, L.L., Jarman, R.A., and Burstein, G.T. Corrosion, Oxford, UK: Butterworth-Heinemann, 2000, 3rd ed.
Yahalom, J., Corros. Sci., 1972, vol. 12, p. 867.
Abd Ei-Rehim, S.S., Ibrahim, M.A., and Khaled, K.F., J. Appl. Electrochem., 1999, vol. 29, p. 595.
Schmid, G.M. and Huang, H.J., Corros. Sci., 1980, vol. 20, p. 1041.
Li, W., He, Q., Zhang, S., et al., J. Appl. Electrochem., 2008, vol. 38, p. 289.
Ishwara Bhat, J. and Alva, V.D.P., Trans. Indian Inst. Met., 2011, vol. 64, p. 377.
Ashassi-Sorkhabi, H., Majidi, M.R., and Seyyed, K., Appl. Surf. Sci., 2004, vol. 225, p. 176.
Badr, G.E., Corros. Sci., 2009, vol. 51, p. 2529.
Paul, V. and Kar, B., ISRN Corros., 2012, vol. 2012.
Fekry, A.M. and Ameer, M.A., Int. J. Hydrogen Energy, 2010, vol. 35, p. 7641.
Singh, A.K. and Quraishi, M.A., Corros. Sci., 2011, vol. 53, p. 1288.
Oguzie, E.E.N., Joku, V.O., Enenebeaku, C.K., et al., Corros. Sci., 2008, vol. 50, p. 3480.
Durnie, W., Marco, R.D., Jefferson, A., et al., J. Electrochem. Soc., 1999, vol. 146, p. 17.
Martinez, S. and Stern, I., Appl. Surf. Sci., 2002, vol. 199, p. 83.
Bockris, J.O.M., Devanathan, M.A., and Mulle, K., Proc. R. Soc. A, 1963, vol. 274, p. 55.
Khaled, K.F. and Hackerman, N., Electrochem. Acta, 2003, vol. 48, p. 2715.
Popova, A., Sokolova, E., Raicheva, S., et al., Corros. Sci., 2003, vol. 45, p. 33.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Kumari P, P., Shetty, P. & Rao, S.A. Corrosion protection properties of 4-hydroxy-N'-[(1E, 2E)-3-phenylprop-2-en-1-ylidene] benzohydrazide on mild steel in hydrochloric acid medium. Prot Met Phys Chem Surf 51, 1034–1042 (2015). https://doi.org/10.1134/S2070205115060143
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2070205115060143