Abstract
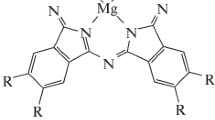
Optical spectroscopy in the UV and visible ranges, dynamic light scattering, and 1H NMR spectroscopy are used to study monomerization of magnesium octa[(4′-benzo-15-crown-5)oxy]phthalocyaninate (Mgcr8Pc) in aqueous solutions of anionic surfactants: sodium dodecyl benzenesulfonate, sodium bis-(2-ethylhexyl)sulfosuccinate, and sodium deoxycholate (SDBS, AOT, and SDC, accordingly). Structural differences in surfactant molecules determine organization of Mgcr8Pc molecules in microheterogeneous media. It is found that Mgcr8Pc is in a monomeric form in aqueous SDBS solutions at concentrations in the range of CMC1 to CMC2 (critical micelle concentration) and it is in a dimeric and/or aggregate form at concentrations below CMC1. In AOT/water solutions (direct micelles) or hexane/AOT (0.1 M)/water (inverted micelles), Mgcr8Pc is solubilized forming dimeric and/or aggregate forms. In micellar solutions of SDC, the Mgcr8Pc monomer is predominantly formed in the presence of NaCl. The results obtained using the NMR spectroscopy technique agree with the binding of Mgcr8Pc by SDBS micelles similar to solubilization of phthalocyanine by micellar solutions of sodium dodecyl sulfate.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Zhang, X.-F., Xi, Q., and Zhao, J., J. Mater. Chem., 2010, vol. 20, p. 6726.
Lang, K., Kubát, P., Mosinger, J., and Wagnerová, D.M., J. Photochem. Photobiol. A, 1998, vol. 119, p. 47.
Tsivadze, A.Yu., Russ. Chem. Bull., 2004, vol. 73, no. 1, p. 5.
Logacheva, N.M., Tsivadze, A.Y., Baulin, V.E., Basova, T.V., and Sheludyakova, L.A., Russ. Chem. Bull., 2008, vol. 57, no. 7, p. 1467.
Ovsyannikova, E.V., Goldshleger, N.F., Kurochkina, N.M., et al., Makrogeterotsikly, 2010, vol. 3, p. 125.
Gol’dshleger, N.F., Kalashnikova, I.P., Baulin, V.E., and Tsivadze, A.Yu., Prot. Met. Phys. Chem. Surf., 2011, vol. 47, no. 4, p. 471.
Goldshleger, N.F., Chernyak, A.V., Kalashnikova, I.P., Baulin, V.E., and Tsivadze, A.Yu., Russ. J. Gen. Chem., 2012, vol. 82, no. 5, p. 927.
Holmberg, K., Jönsson, B., Kronberg, B., and Lindman, B., Surfactants and Polymers in Aqueous Solution, Chichester: Wiley, 2002, 2nd ed.
Gordon, A. and Ford, R., Handbook of Practical Data, Technique and References, New York: Wiley, 1972.
Kasha, M., Rawls, H., and El-Bayoumi, M., Pure Appl. Chem., 1965, vol. 11, p. 371.
Wang, Y., Deng, K., Gui, L., et al., Colloid Interface Sci., 1999, vol. 213, p. 270.
Yu, D., Huang, F., and Xu, H., Anal. Methods, 2012, vol. 4, p. 47.
Domínguez, A., Fernández, A., González, N., et al., J. Chem. Educ., 1997, vol. 74, p. 1227.
Chatterjee, A., Moulik, S., Sanyal, S., et al., J. Phys. Chem. B, 2001, vol. 105, p. 12823.
Kumar, S., Sharma, D., Sharma, D., and Kabir-ud-Din, J. Surfactants Deterg., 2006, vol. 9, p. 77.
Palazzesi, F., Calvaresi, M., and Zerbetto, F., Soft Matter, 2011, vol. 7, p. 9148.
Lu, X.-Y., Jiang, Y., Cui, X-H., et al., Acta Phys.-Chim. Sin., 2009, vol. 25, p. 1357.
Hait, S.K., Majhi, P.R., Blume, A., and Moulik, S.P., J. Phys. Chem. B, 2003, vol. 107, p. 3650.
Cheng, D.C.H. and Gulari, E., Colloid Interface Sci., 1982, vol. 90, p. 410.
Rai, R., Baker, G., Behera, K., et al., Langmuir, 2010, vol. 26, p. 17821.
Kumar, S., Sharma, D., Ghosh, G., and Kabir-ud-Din, Langmuir, 2005, vol. 21, p. 9446.
Gehlen, M.H. and De Schryver, F.C., J. Phys. Chem., 1993, vol. 97, p. 11242.
Mata, J., Varade, D. Ghosh, G. and Bahadur, P., Colloids Surf., A, 2004, vol. 245, p. 69.
Sheu, E.Y., Chen, S-H, and Huang, J.S., J. Phys. Chem., 1987, vol. 91, p. 3306.
Ninomiya, R., Matsuoka, K., and Moroi, Y., Biochim. Biophys. Acta, 2003, vol. 1634, p. 116.
Pártay, L.B., Jedlovszky, P., and Sega, M., J. Phys. Chem. B, 2007, vol. 111, p. 9886.
D’Alagni, M., D’Archivio, A.A., Galantini, L., and Giglio, E., Langmuir, 1997, vol. 13, p. 5811.
Correia, R.F., Andrade, S.M., and Viseu, M.I., J. Photochem Photobiol., A, 2012, vol. 235, p. 21.
Göktürk, S., J. Photochem Photobiol., A, 2005, vol. 169, p. 115.
Arnaud-Neu, F., Delgado, R., and Chaves, S., Pure Appl. Chem., 2003, vol. 75, p. 71.
Tovstun, S.A. and Razumov, V.F., Russ. Chem. Rev., 2011, vol. 80, no. 10, p. 953.
Rosen, M.J., Surfactants and Interfacial Phenomena, New York: Wiley, 2004, 3rd ed.
Ghosh, S., Chem. Phys. Lett., 2010, vol. 500, p. 295.
Smith, R.J., Lotya, M., and Coleman, J.N., New J. Phys., 2010, vol. 12, p. 125008.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © N.F. Goldshleger, A.V. Chernyak, A.S. Lobach, I.P. Kalashnikova, V.E. Baulin, A.Yu. Tsivadze, 2015, published in Fizikokhimiya Poverkhnosti i Zashchita Materialov, 2015, Vol. 51, No. 2, pp. 160–169.
Rights and permissions
About this article
Cite this article
Goldshleger, N.F., Chernyak, A.V., Lobach, A.S. et al. Monomerization of crown-containing phthalocyanines in microheterogeneous organized systems. Prot Met Phys Chem Surf 51, 212–220 (2015). https://doi.org/10.1134/S2070205115020070
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2070205115020070