Abstract
The results of a phenology study of 78 species of perennial plants over a 20-year period (1996–2015) in the Western Siberia forest steppe are discussed. Against the background of the detected meteorological trends of the warm season in Novosibirsk, the timing shift in phenological events of individual species and their ecological groups have been analyzed using linear trends. It is found that the start of vegetation in the vast majority of perennials was delayed (+3 to +7 days), and the terms of vegetation end changed differently (within –4 to +1 days), while the duration of vegetation decreased in all groups. The duration of the prefloral period significantly decreased (–6 to –9 days) due to the later vegetation start and an earlier start of flowering (–1 to –2 days). Fruiting trends are different in direction and magnitude. The rate of seasonal development of the species generally accelerated over the observation period. The trends describe 6–32% of phenological changes in ecological groups.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Plant phenology serves as one of the most reliable bioindicators of current climate change. Observed and predicted phenological shifts have significant implications for biodiversity (Peñuelas et al., 2009; Ray, 2015). The vast majority of research in situ in temperate ecosystems of the Northern Hemisphere shows an advance in the timing of spring phenophases and a delay in autumn ones, while the identified phenological trends generally correspond to patterns of temperature changes and the duration of the growing season (Walther et al., 2002; Parmesan and Yohe, 2003; Menzel et al., 2006; Cleland et al., 2007; Khanduri et al., 2008).
However, the amplitude of changes in phenology is not the same depending on the region, the studied species, the length of the time series, or its start and end dates, which are crucial for identifying trends (Sparks and Menzel, 2002; Bertin, 2008). Changes in spring phenology in North America were slower than in Europe, and in China they were faster. At the same time, the temporal trends of autumn phenomena are much weaker than spring ones, especially in Europe. On the contrary, the results of comparable studies in East Asia have shown that autumn phenology changes more quickly than spring phenology (Ibáñez et al., 2010; Piao et al., 2019).
The global mean surface air temperature data indicate a hiatus in global warming over the period 1998–2012 (Wang et al., 2019). With progressive trends in plant phenology, the pace slowed down or even reversed (Piao et al., 2019). According to Jeong et al. (2011), SOS advanced 5.2 days in 1982–1999, but only 0.2 days in 2000–2008 on average throughout the Northern Hemisphere, although the length of the growing season has increased. The EOS delay was 4.3 days and 2.2 days, respectively. Seasonal warming patterns apparently have spatial and temporal variations that control phenological events (Menzel et al., 2020).
It is shown that the patterns of the response of organisms to climate warming are not universal. Even within the same habitat, different species demonstrate contrasting long-term responses to directional climate change, as well as to its interannual fluctuations. Differences in the temporal responses of plant phenology to climate change are due to the unequal sensitivity of phenological phenomena and species (Miller-Rushing et al., 2008; Gordo and Sanz, 2010; Chmura et al., 2019).
Spring phenological traits such as leafing and flowering of temperate and boreal perennials are a function of the diurnal heat buildup following winter cooling. The need for plants to be exposed to low temperatures can delay development in the spring, when higher winter temperatures lengthen the period required for cooling (Bertin, 2008). In addition, an increase in winter temperatures may be accompanied by an increase in snowfalls and a delay in the time of snow melting, which has a significant effect on the timing of spring phenophases (Inouye et al., 2003). Autumn phenological traits, including bud formation, leaf fall, and the development of cold hardiness in perennials, are usually photoperiod-induced and are less dependent on temperature. However, extreme heat and drought in some years, combined with biotic disturbances, can lead to premature leaf senescence (Franks et al., 2013; Menzel et al., 2020).
The phenological sensitivity of species can vary greatly in different parts of the range due to adaptation to the local climate, but the reasons for these regional differences are currently unknown (Park et al., 2019). For example, a meta-analysis of the phenology of steppe plants showed unequal reactions of species against the background of general phenological trends that take place not only between populations throughout the species range, but also between individuals within a population (Huang et al., 2020). Ground-based observations provide information on the responses of individual species and intraspecific variability in a given microregion (Badeck, 2004; Khanduri et al., 2008).
In most cases, collections of living plants and herbarium specimens serve as the only source of long-term phenological data (Robbirt et al., 2011). The movement of plants in the experiment (introduction) makes it possible to provide a series of local observations for many species, thereby revealing the norms of phenological reactions in various ecological conditions (Wolkovich et al., 2014; Berend et al., 2019; MacKenzie et al., 2020). The collections make botanic gardens well-positioned to conduct observations and experiments related to plant conservation or global change (Donaldson, 2009).
Research on the impact of global warming on plant phenology has intensified in the Russian Federation in the last decade (Barinova and Kokhanovskaya, 2015; Bobretsov et al., 2017; Zorina et al., 2018). Observational data and model calculations show that the average temperature growth rate in Russia is one and a half times higher than on land in the Northern Hemisphere, amounting to +0.45°С per decade. The highest air temperature growth rate is recorded in spring (+0.62°С/10 years), summer, and autumn (+0.42°С/10 years). The increase in winter and autumn temperatures is unstable; cooling has been observed from the mid-1990s to 2010, except for the polar zone. There is also a trend towards an increase in annual precipitation, the most significant being in spring, and it is especially noticeable in the North Caucasus, the south of European Russia, the Urals, and Siberia (Sed’moe…, 2017).
In Siberia, there are very few studies of changes in plant phenology based on long-term ground-based observations. For example, Rosbach et al. (2021) analyzed phenological shifts in 67 species of woody and herbaceous plants in the territory of the Barguzinsky Nature Reserve for the period 1976–2018. The advancement of spring and summer events is shown, as is the delay of autumn ones. The growing season in Siberian boreal forests has lengthened by about 15 days. At the same time, the change in the phenology of individual species was ambiguous, both in direction and magnitude.
We (Fomin and Fomina, 2021) showed the consistency of phenological changes with trends in climatic parameters in plants of various phenorhythmotypes in the conditions of the forest steppe of Western Siberia. The earlier accumulation of sums of active temperatures accelerated the development of perennials, especially those that bloom in summer. In long-term vegetative autumn and winter green species, the growing season has significantly decreased, repeating the trend of snow cover. Thus, although data on shifts in plant phenology under the influence of climate change are accumulating, the responses of individual species remain unpredictable, and without long-term phenological data it is impossible to predict the effects of global warming on biodiversity.
The purpose of this study was to assess the change in the timing of phenological events in perennial plants from various ecological groups in connection with the climate change trend in Novosibirsk.
MATERIAL AND METHODS
This study was carried out in the Central Siberian Botanical Garden, Siberian Branch, Russian Academy of Sciences (Novosibirsk) (55°2′29.4″ N, 82°56′4.6″ E). The objects were 78 species of perennial plants from the collection of ornamental species of natural flora on which phenological observations were carried out during 1996‒2015. To characterize climatic indicators for the study period, we used data from the Ogurtsovo weather station (no. 29638).Footnote 1
The data of meteorological and phenological observations were processed by standard statistical methods using the MS Excel package and our own programs written in C++. During the study period, meteorological trends were calculated for air temperature indicators T, rainfall R, and hydrothermal coefficient (HTC) of the warm season. The method of constructing separate linear trends for each date from April 1 to October 31 was used, followed by the calculation of a simple moving average in a 30-day window in order to smooth out short-term fluctuations and identify major changes. Next, we calculated the phenological trends of phenophases and interphase periods, both for each species separately and for ecological groups.
The bootstrap method was used to build trends and evaluate their reliability—a computer statistical research method of studying probability distributions based on multiple generation and analysis of random samples based on the available experimental samples (Efron, 1979). Although it has a high computational cost, the advantage of bootstrap is that it is applicable to any distribution and different sample sizes, providing the same or better results than conventional statistical methods (Singh, 1981). Since most of the analyzed distributions are not normal, to present data, instead of arithmetic means with a standard deviation, we used their analogs applicable to any type of distribution—median values (Me) with confidence interval σ68.2, which includes 68.2% of the most likely events.
We analyzed the dates of phenological events over a 20-year period and their shifts due to linear trends over the same period of time. Correlations and determination coefficients were calculated using Pearson linear coefficient r. To interpret the correlation value, we used a scale in which the boundaries of the categories are shifted towards low values: |r| ≤ 0.1 is weak, 0.1 < |r| < 0.3 is average, and |r| ≥ 0.3 is high. This is justified for the analysis of phenological reactions of plants that exhibit high inertia in response to continuous, multiscale, and multidirectional changes in weather conditions, while the value of r does not necessarily mean small consequences (Koterov et al., 2019). The significance level is α = 0.05. Thus, a probability of error of pv ≤ 0.05 indicates the reliability of the trend.
The study point, Novosibirsk, is located in the southeastern part of the West Siberian Plain (Priobskoe plateau) in the forest-steppe zone, at the junction of the hemiboreal forest subzones and the northern forest steppe. The climate of the area is continental, moderately cold with insufficient moisture. Average temperatures in winter months are –15 to –18°C and, in the summer, 16–19°C. The frost-free period lasts 96–146 days. The average long-term precipitation is 440 mm, of which 320 mm falls during the warm season. The average annual sunshine duration is 2088 h. The climate warming trend in Novosibirsk over the past half-century has been manifested by an increase in the average annual air temperature by 0.27°C per decade, while the largest temperature increase is observed for transitional seasons, but the temperature background of the summer months remains almost constant. The moisture regime has changed: in the warm season, the amount of precipitation decreased by 11 mm, while the depth of snow cover increased by 4 cm per decade and the period of its stable occurrence lengthened by almost two decades (up to 160 days on average). One characteristic feature of the current climate change is the increase in the amplitude of interannual fluctuations in climatic indicators. Like in the Northern Hemisphere as a whole, with the general trend of climate warming in Novosibirsk, there are cyclical fluctuations in weather conditions towards cooling (Voronina and Gritsenko, 2011; Luchitskaya et al., 2014).
RESULTS
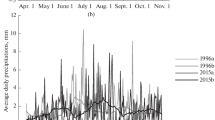
The analysis of meteorological data for the study period showed an increase in the average air temperature in the warm season in Novosibirsk by 0.16°C, while the total amount of precipitation from April to October did not change (–0.01 mm). Intraseasonal periods with significant changes in the main meteorological indicators have been identified (Fig. 1). April became noticeably warmer (+39.6°C), while May became colder and wetter (–63.1°C; +16.6 mm). In the first half of the summer, the likelihood of hot and dry weather increased (+32.7°C; –40.7 mm), while in the second half, the amount of precipitation increased significantly (+33.7 mm). The beginning of autumn became warmer and drier (+28.7°C; –14.4 mm). In October, humidity increased (+15.9 mm), but the temperature background did not change. The duration of the growing season with average daily air temperatures above 5°C significantly increased by 12 days; the period with active temperatures above 10°C increased by 8 days. At the same time, the frost-free period was reduced by 5 days due to the shift in the date of the last spring frost (due to cooling in May). The melting of the snow cover was delayed by 4 days at the same average long-term date of establishment. Values of HTC during the season significantly deviated from the long-term average of 1.0. Precipitation was distributed extremely unevenly, but, in general, the change in the moisture index was not significant.
The studied species are divided into four ecological groups (Fig. 2) in accordance with the work of T.I. Fomina (2012). To obtain more comparable samples, mesohygrophytes and hygrophytes are combined into one group. Phenological trends Δη are calculated by building a linear regression η (year) = α + β × year, where η is the parameter (the date of the onset of the phenophase or its duration); α and β are the parameters of linear regression, with a further calculation of the difference between the regression values at the boundaries of the interval of years Δη = η (2015) – η (1996). The resulting trends were compared with meteorological trends at the level of individual species and their groups.
Tables 1 and 2 show the results of a statistical analysis of the shift in the dates of phenological events (phenophases and interphase periods) by ecological groups of species. Because phenological characters tend to deviate in distribution from normal, and taking into account the large spread of phenodates, the median was used to represent the data (Me) and standard deviation σ with confidence intervals of 68.2%. The processing of phenodata at the group level due to a significant increase in the volume of samples (up to 4000 records per group) makes it possible to increase the accuracy and reliability of the trends. The species of each group in percentage terms were divided into those significantly supporting the trend (N+) and significantly deviating in the opposite direction (N–), while difference 100 – N+ – N– represents the proportion of species whose phenology has not changed significantly.
It is important that the group effect is strong in the distribution of phenological traits. For example, by precisely defining the value N+, we cannot just as accurately designate the species composition—there is always a part of the species that balances on the verge of reliability; thus, the trend is supported by one or the other participants, depending on the weather conditions of a particular season. Coefficients of determination above zero values (R2 ≥ 0.1) show that there is a significant share in the interannual variability in the timing of phenological events (≥10%) due to the trend.
The diagrams in Fig. 3 display the expected (i.e., most likely) timing of phenological events for species by group, obtained from trends for the beginning (1996) and end (2015) of the study period. The phenospectra give an idea of the direction of phenological shifts in individual species and make it possible to approximately estimate the amplitude of these shifts. Figure 4 shows phenological trends for a number of Siberian species. Median phenodate values (Me) are indicated by circles; the box sizes correspond to the boundaries of the confidence interval σ68.2%, and the length of the whiskers reflects the range of “outliers” (all possible values).
DISCUSSION
The phenology of perennials over the period under consideration has undergone noticeable changes that occurred against the background of intraseasonal (from April to October) trends in air temperature and precipitation. At the same time, the variations in phenological events reflected the ambiguous responses of species to changes in climatic parameters. Previously, we (Fomin and Fomina, 2021) revealed positive correlations between the timing of the start of vegetation and the start of flowering with the date of the snow cover melting, as well as the dates of the transition of average daily air temperatures in spring through 5°С and 10°С upwards. Meanwhile, the shifts in the dates of the onset of phenophases and the timing of interphase periods differ in the direction and amplitude of variability.
The dates of the start of vegetation (spring regrowth) in all ecological groups significantly shifted to later, within 3–7 days, which is consistent with the snowmelt trend (+4 days). The percentage of species growing earlier, i.e., not supporting the trend, was the lowest in xerophytes (Table 1). This may be due to the trends of the cold period of the year. An increase in winter temperatures (+0.23°C/10 years) together with an increase in snow depth (+4 cm/10 years) (Luchitskaya et al., 2014) worsen the conditions for overwintering of xerophytes (the risk of damping out under the snow increases), which affects at the beginning of their seasonal development.
The flowering phase came in all groups with a slight lead of 1–2 days (Table 2). To a greater extent, the duration of the prefloral period changed towards reduction of 6–9 days, with the maximum trend value for xerophytes. The duration of flowering significantly increased in all groups, with the exception of xerophytes (–5 days). With later regrowth dates, on average, these species accelerated to the maximum in phenological development, as is evidenced by a significant decrease in the duration of both the prefloral period and the flowering period. The main contribution to the earlier start of perennial flowering was made by elevated air temperatures in April (r = 0.34), while May temperatures did not have an effect (r = 0.09). Fruiting trends in the groups are significant, with the exception of mesophytes, and are multidirectional. The period of seed ripening was reduced in hygrophilous species and mesoxerophytes: 7 and 10 days, respectively. In xerophytes, on the contrary, there has been a positive trend (+10 days).
The dates of the end of the growing season in xerophytes and, especially, hygrophilous species have shifted to earlier dates, so the growing season in these two groups has decreased significantly: for 10 and 14 days, respectively. In the groups of mesoxerophytes and mesophytes, the vegetation period decreased, mainly due to the later dates of spring regrowth. In general, the dependence of autumn phenophases on external conditions is more complicated than that of spring–summer ones (Menzel et al., 2006; Ge et al., 2015). An increase in summer temperatures accelerates the end of the growing season, while an increase in precipitation delays it (Huang et al., 2020). On the contrary, an increase in autumn temperatures slows down the degradation of chlorophyll in perennials, prolonging the vegetation period (Shi et al., 2014), and delays the onset of frosts. In addition, if spring processes in plants are triggered by temperatures, then the end of vegetation in herbaceous species may be due to endogenous factors that do not depend on external conditions (Karpisonova, 1985; Zhmylev et al., 2001).
The studied perennials are dominated by species that support the trends of a later start of the growing season and its earlier end. At the same time, the percentage of species that maintain vegetation trends in spring is much higher than in autumn. Determination coefficients R2 of the discussed signs are low and medium, and they vary within 0.07–0.21. The duration of the growing season is determined primarily by the biomorph of the species, while ecological features play a secondary role. The coefficients of determination of reproductive phenology, with the exception of the beginning of flowering, are higher, especially the timing of the appearance of mature seeds (R2 = 0.18–0.32). Thus, our data indicate a greater phenological sensitivity of the phases of vegetation and the beginning of flowering of perennials.
An analysis of the phenospectra illustrated the extremely high interspecies variability of phenodates over the study period (Fig. 3). The positive trend of the dates of the beginning of the growing season was 1–14 days, while in 17 species from different ecological groups the trend is negative, within 1–7 days. Multidirectional trends in the dates of the beginning of flowering reached 22 days, and the dates of mature seeds reached 25 days. Trends in the timing of the end of the growing season had a maximum value of up to 27 days in both directions. For a small number of species, no shifts were found for individual phenophases. Therefore, the dates of the beginning of the growing season and mature seeds did not change in three species, and the dates of the beginning of flowering and the end of the growing season did not change in seven species. However, not a single perennial has been found whose phenology has not undergone noticeable shifts, although the reliability of these changes has not been proven for the most part.
The phenology of Siberian species also showed multidirectional and unequal shifts. The diagrams (Fig. 4) show a number of species with significant changes in phenological characters (pv ≤ 0.05) or high coefficients of determination (R2 ≥ 0.2), indicating that the interannual variability of phenodates is not high enough to mask the presence of a trend. In Aizopsis aizoon (L.) Grulich, for example, the beginning of the growing season was late (+6 days), and all subsequent phenophases were ahead 6–8 days at low trend confidence values. Lilium pumilum Delile bloomed in 2015 at the same time as in 1996, but the phase of mature seeds was significantly shifted (+12 days); the growing season changed (–4 days for its start and +6 days for its end). Anemonoides altaica (C.A. Mey.) Holub shows positive trends of all phenodates with a shift of 2–6 days and high coefficients of determination of phenodate, except for the date of the beginning of flowering. The opposite change in phenological traits was found in Filipendula ulmaria (L.) Maxim. All trends are negative, with a significant maximum value for the date of mature seeds (–11 days).
In Rheum rhabarbarum L., phenodate shifts are also negative, except for a much later end of the growing season (+7 days). At the same time, trend determination coefficients are high for all phenophases, except for regrowth (–5 days). Thalictrum flavum L. revealed an earlier start and end of vegetation (vegetation duration decreased 4 days), while the phases of flowering and mature seeds shifted to later periods (+7 and +16 days, respectively). The change in the start of flowering of this species is significantly due to the trend. In Gentiana macrophylla Pall., shifts in the dates of the beginning of vegetation (–4 days) and the beginning of flowering (–5 days) are unreliable, but the delay in the timing of seed maturation and the end of vegetation is strongly determined by the trend. The phenology of Hemerocallis minor Mill. has changed: the vegetation start date shows a strong positive trend (+10 days) and flowering start dates and mature seeds show strong negative trends (–14 and –10 days, respectively) at a significance level, while there was no shift in the timing of the end of the growing season. In the presented species from different ecological groups, the dates of mature seeds showed strong trends (0.26 ≤ R2 ≤ 0.65) and, for all species, the determination of trends decreased in the direction of the end of the growing season → the beginning of flowering → the beginning of the growing season.
The results of the study show the complex nature of changes in plant phenology under conditions of climate warming. One of the factors of phenological variability is that the phases of seasonal development in different species fall on different time intervals (Bertin, 2008). Also, phenological sensitivity depends on the biomorph; in herbaceous plants in northern and boreal latitudes, it is higher than in tree species (Root et al., 2003). The ecological characteristics of species are of great importance (Zorina et al., 2018). It is believed that, under conditions of rapid climate change, the phenotypic plasticity of structural and functional traits, including phenological ones, plays a major role in plant adaptation. Ultimately, the level of synchronization of phenology with the climatic conditions of the habitat determines the reproductive success, growth, and survival of individuals (Cleland et al., 2007; Franks et al., 2013; Gratani, 2014).
At the same time, phenological reactions are triggered in response to regional climate changes that are more significant for plants than global averages (Walter et al., 2002). Therefore, long-term trends in phenological events can serve as an indicator of recent climate changes in the region. Most of the period of our study coincided with a pause in warming that has been recorded in different parts of the Northern Hemisphere since the late 1990s (Piao et al., 2019; Wang et al., 2019). Cooling events were observed in Western Siberia as part of cyclic climate fluctuations (Voronina and Gritsenko, 2011). Thus, May became the coolest month of the warm season (Luchitskaya et al., 2014). This variation of climate warming was manifested by a slowdown in the rate of increase in air temperatures and, as a result, a decrease or absence of a shift in phenophases, making it difficult to identify general trends in phenological changes.
CONCLUSIONS
The results confirm the change in the phenology of perennials under conditions of climate warming and the presence of displacements that are heterogeneous in amplitude and direction. Plant responses to interannual fluctuations and long-term trends in climatic parameters are species-specific and manifest themselves as different trends in shifting the timing of phenological events. It is shown that phenological sensitivity is to a certain extent associated with the ecological characteristics of the species. Some of the calculated trends are consistent with the literature data: first and foremost, the trend of the advanced beginning of flowering. However, the dominant trends of a delayed start of the growing season, an advanced end of the growing season, and a reduction in the duration of the growing season of perennials (despite the fact that the season has lengthened) can be partly explained by the trend of cooling of the region’s climate during the study period.
One characteristic feature of recent climate change is significant interannual and intraseasonal fluctuations in meteorological indicators, which provide a very high variability of phenological shifts and, thus, complicate the construction of phenological trends. All studied species have undergone changes in phenology to a greater or lesser extent over the course of 20 years, while most of the identified shifts are not statistically significant and are primarily due to the high interannual variability of climatic parameters.
Notes
https://rp5.ru/ Weather_archive_in_Ogurtsovo.
REFERENCES
Badeck, F.-W., Bondeau, A., Böttcher, K., Doktor, D., Lucht, W., Schaber, J., and Sitch, S., Responses of spring phenology to climate change, New Phytol., 2004, vol. 162, pp. 295–309. https://doi.org/10.1111/j.1469-8137.2004.01059.x
Barinova, G.M. and Kokhanovskaya, M.I., Manifestations of climate variability in the dynamics of seasonal development of plants in the South-Eastern Baltic region, Vestn. Balt. Fed. Univ. im. I. Kanta, 2015, vol. 1, pp. 8–18.
Berend, K., Haynes, K., and McDonough MacKenzie, C., Common garden experiments as a dynamic tool for ecological studies of alpine plants and communities in no.ortheastern America: Featured review, Rhodora, 2019, vol. 121, no.o. 987, pp. 174–212. https://doi.org/10.3119/18-16
Bertin, R.I., Plant phenology and distribution in relation to recent climate change, J. Torrey Bot. Soc., 2008, vol. 135, no. 1, pp. 126–146. https://doi.org/10.3159/07-RP-035R.1
Bobretsov, A.V., Tertitsa, T.K., and Teplova, V.P., The impact of climate change on the phenology of plants and animals of the south-eastern part of the Komi Republic (The Pechora-Ilych biosphere reserve), Probl. Ekol. Monit. Model. Ekosist., 2017, vol. 28, no. 4, pp. 74–93. https://doi.org/10.21513/0207-2564-2017-4-74-93
Chmura, H.E., Kharouba, H.M., Ashander, J., Ehlman, S.M., Rivest, E.B., and Yang, L.H., The mechanisms of phenology: The patterns and processes of phenological shifts, Ecol. Monogr., 2019, vol. 89, no. 1, p. e01337. https://doi.org/10.1002/ecm.1337
Cleland, E.E., Chuine, I., Menzel, A., Mooney, H.A., and Schwartz, M.D., Shifting plant phenology in response to global change, Trends Ecol. Evol., 2007, vol. 22, no. 7, pp. 357–365. https://doi.org/10.1016/j.tree.2007.04.003
Donaldson, J.S., Botanic gardens science for conservation and global change, Trends Plant Sci., 2009, vol. 14, no. 11, p. 608. https://doi.org/10.1016/j.tplants.2009.08.008
Efron, B., Bootstrap methods: Another look at the jackknife, Ann. Stat., 1979, vol. 7, no. 1, pp. 1–26. https://doi.org/10.1214/aos/1176344552
Fomin, E.S. and Fomina, T.I., Changes in the phenology of perennial plants in Western Siberia against the background of global climate warming, Contemp. Probl. Ecol., 2021, no. 5, pp. 543–556. https://doi.org/10.15372/SEJ20210504
Fomina, T.I., Biologicheskie osobennosti dekorativnykh rastenii prirodnoi flory v Zapadnoi Sibiri (Biological features of ornamental plants of no.atural flora in Western Siberia), Novosibirsk: GEO, 2012.
Franks, S.J., Weber, J.J., and Aitken, S.N., Evolutionary and plastic responses to climate change in terrestrial plant populations, Evol. Appl., 2013, vol. 7, no. 1, pp. 123–139. https://doi.org/10.1111/eva.12112
Ge, Q., Wang, H., Rutishauser, T., and Dai, J., Phenological response to climate change in China: A meta-analysis, Global Change Biol., 2015, vol. 21, no. 1, pp. 265–274. https://doi.org/10.1111/gcb.12648
Gordo, O. and Sanz, J., Impact of climate change on plant phenology in Mediterranean ecosystems, Global Change Biol., 2010, vol. 16, pp. 1082–1106. https://doi.org/10.1111/j.1365-2486.2009.02084.x
Gratani, L., Plant phenotypic plasticity in response to environmental factors, Adv. Bot., vol. 2014, p. 208747. https://doi.org/10.1155/2014/208747
Huang, W., Dai, J., Wang, W., Li, J., Feng, C., and Du, J., Phenological changes in herbaceous plants in China’s grasslands and their responses to climate change: A meta-analysis, Int. J. Biometeorol., 2020, vol. 64, pp. 1865–1876. https://doi.org/10.1007/s00484-020-01974-1
Ibáñez, I., Primack, R.B., Ellwood, E., Miller-Rushing, A.J., Higuchi, H., Lee, S.D., Kobori, H., and Silander, J.A., Forecasting phenology under global warming, Philos. Trans. R. Soc., B, 2010, vol. 365, no. 1555, pp. 3247–3260. https://doi.org/10.1098/rstb.2010.0120
Inouye, D.W., Saavedra, F., and Lee-Yang, W., Environmental influences on the phenology and abundance of flowering by Androsace septentrionalis (Primulaceae), Am. J. Bot., 2003, vol. 90, no. 6, pp. 905–910.
Jeong, S.J., Ho, C.H., Gim, H.J., and Brown, M.E., Phenology shifts at start vs. end of growing season in temperate vegetation over the northern Hemisphere for the period 1982–2008, Global Change Biol., 2011, vol. 17, pp. 2385–2399. https://doi.org/10.1111/j.1365-2486.2011.02397.x
Karpisonova, R.A., Travyanistye rasteniya shirokolistvennykh lesov SSSR: ekologo-floristicheskaya i introduktsionnaya kharakteristika (Herbaceous Plants of Broad-Leaved Forests of the USSR: Ecological-Floristic and Introduction Characteristics), Moscow: Nauka, 1985.
Khanduri, V.P., Sharma, C.M., and Singh, S.P., The effects of climate change on plant phenology, Environmentalist, 2008, vol. 28, pp. 143–147. https://doi.org/10.1007/s10669-007-9153-1
Koterov, A.N., Ushenkova, L.N., Zubenkova, E.S., Kalinina, M.V., Biryukov, A.P., Lastochkina, E.M., Molodtsova, D.V., and Vainson, A.A., Strength of Association. Report 2. Graduations of correlation size, Med. Radiol. Radiats. Bezop., 2019, vol. 64, no. 6, pp. 12–24. https://doi.org/10.12737/1024-6177-2019-64-6-12-24
Luchitskaya, I.O., Belaya, N.I., and Arbuzov, S.A., Klimat no.ovosibirska i ego izmeneniya (Climate Change in Novosibirsk), Yagudin, R.A., Ed., Novosibirsk: Sib. Otd. Ross. Akad. Nauk, 2014.
McDonough MacKenzie, C., Gallinat, A.S., and Zipf, L., Low-cost observations and experiments return a high value in plant phenology research, Appl. Plant Sci., 2020, vol. 8, no. 4, p. e11338. https://doi.org/10.1002/aps3.11338
Menzel, A., Sparks, T.H., Estrella, N., Koch, E., Aasa, A., Ahas, R., Kübler, K.A., Bissolli, P., Braslavská, O., Briede, A., Chmielewski, F.M., Crepinsek, Z., Curnel, Y., Dahl, A., Defila, C., Donnelly, A., Filella, Y., Jatczak, K., Måge, F., Mestre, A., Nordli, Ø., Peñuelas, J., Pirinen, P., Remišová, V., Scheifinger, H., Striz, M., Susnik, A., Vanvliet, A.J.H., Wielgolaski, F.E., Zach, S., and Zust, A., European phenological response to climate change matches the warming pattern, Global Change Biol., 2006, vol. 12, pp. 1969–1976. https://doi.org/10.1111/j.1365-2486.2006.01193.x
Menzel, A., Yuan, Y., Matiu, M., Scheifinger, H., Gehrig, R., and Estrella, N., Climate change fingerprints in recent European plant phenology, Global Change Biol., 2020, vol. 26, pp. 2599–2612. https://doi.org/10.1111/gcb.15000
Miller-Rushing, A.J., Inouye, D.W., and Primack, R.B., How well do first flowering dates measure plant responses to climate change? The effects of population size and sampling, J. Ecol., 2008, vol. 96, pp. 1289–1296. https://doi.org/10.1111/j.1365-2745.2008.01436.x
Park, I., Jones, A., and Mazer, S.J., PhenoForecaster: A software package for the prediction of flowering phenology, Appl. Plant Sci., 2019, vol. 7, no. 3, p. e1230. https://doi.org/10.1002/aps3.1230
Parmesan, C. and Yohe, G., A globally coherent fingerprint of climate change impacts across natural systems, Nature, 2003, vol. 421, pp. 37–42. https://doi.org/10.1038/nature01286
Peñuelas, J., Rutishauser, T., and Filella, I., Phenology feedbacks on climate change, Science, 2009, vol. 324, p. 887. https://doi.org/10.1126/science.1173004
Piao, S., Liu, Q., Chen, A., Janssens, I.A., Fu, Y., Dai, J., Liu, L., Lian, X., Shen, M., and Zhu, X., Plant phenology and global climate change: Current progresses and challenges, Global Change Biol., 2019, vol. 25, pp. 1922–1940. https://doi.org/10.1111/gcb.14619
Rai, P.K., A concise review on multifaceted impacts of climate change on plant phenology, Environ. Skeptics Critics, 2015, vol. 4, no. 4, pp. 106–115.
Robbirt, K.M., Davy, A.J., Hutchings, M. J., and Roberts, D.L., Validation of biological collections as a source of phenological data for use in climate change studies: A case study with the orchid Ophrys sphegodes, J. Ecol., 2011, vol. 99, pp. 235–241. https://doi.org/10.1111/j.1365-2745.2010.01727.x
Root, T.L., Price, J.T., Hall, K.R., Schneider, S.H., Rosenzweigk, C., and Pounds, J.A., Fingerprints of global warming on wild animals and plants, Nature, 2003, vol. 421, pp. 57–60. https://doi.org/10.1038/nature01333
Rosbakh, S., Hartig, F., Sandanov, D.V., Bukharova, E.V., Miller, T.K., and Primack, R.B., Siberian plants shift their phenology in response to climate change, Global Change Biol., 2021, vol. 27, pp. 4435–4448. https://doi.org/10.1111/gcb.15744
Sed’moe no.atsional'noe soobshchenie Rossiiskoi Federatsii, predstavlennoe v sootvetstvii so stat’yami 4 i 12 Ramochnoi Konventsii Organizatsii Ob’’edinennykh no.atsii ob izmenenii klimata i stat’ei 7 Kiotskogo protokola (Seventh no.ational Communication of the Russian Federation, Submitted in Accordance with Articles 4 and 12 of the United no.ations Framework Convention on Climate Change and Article 7 of the Kyoto Protocol), Moscow, 2017.
Shi, C., Sun, G., Zhang, H., Xiao, B., Ze, B., Zhang, N., and Wu, N., Effects of warming on chlorophyll degradation and carbohydrate accumulation of alpine herbaceous species during plant senescence on the Tibetan Plateau, PLoS One, 2014, vol. 9, p. e107874. https://doi.org/10.1371/journal.pone.0107874
Singh, K., On the asymptotic accuracy of Efron’s bootstrap, Ann. Stat., 1981, vol. 9, no. 6, pp. 1187 – 1195. https://doi.org/10.1214/aos/1176345636
Sparks, T.H. and Menzel, A., Observed changes in seasons: An overview, Int. J. Climatol., 2002, vol. 22, pp. 1715–1725. https://doi.org/10.1002/joc.821
Voronina, L.V. and Gritsenko, A.G., Klimat i ekologiya no.ovosibirskoi oblasti (Climate and Ecology of the no.ovosibirsk Region), Novosibirsk: Sib. Gos. Geod. Akad., 2011.
Walther, G.-R., Post, E., Convey, P., Menzel, A., Parmesan, C., Beebee, T.J.C., Fromentin, J.-M., Hoegh-Guldberg, O., and Bairlein, F., Ecological responses to recent climate change, Nature, 2002, vol. 416, pp. 389–395.
Wang, X., Xiao, J., Li, X., Cheng, G., Ma, M., Zhu, G., Arain, M.A., Black, T.A., Jassal, R.S., No trends in spring and autumn phenology during the global warming hiatus, Nat. Commun., 2019, vol. 10, p. 2389. https://doi.org/10.1038/s41467-019-10235-8
Wolkovich, E.M., Cook, B.I., and Davies, T.J., Progress towards an interdisciplinary science of plant phenology: Building predictions across space, time and species diversity, New Phytol., 2014, vol. 201, pp. 1156–1162. https://doi.org/10.1111/nph.12599
Zhmylev, P.Yu., Zhmyleva, A.P., Karpukhina, E.A., and Titovets, A.V., Possible reasons for changes in the seasonal development of plants due to climate warming, Vestn. Ross. Univ. Druzhby Nar., 2001, no. 9, pp. 98–103.
Zorina, A.A., Shuiskaya, E.A., Kurakina, I.V., Ogurtsov, S.S., and Stepanov, S.N., The biota reaction on local climatic changes in the Central Forest no.ature reserve, Vestn. Tversk. Gos. Univ., Ser.: Biol. Ekol., 2018, no. 1, pp. 58–77.
ACKNOWLEDGMENTS
In preparing the article, materials from the Bioresource Scientific Collection of the Central Siberian Botanical Garden, Siberian Branch, Russian Academy of Sciences, unique scientific installations “Collections of Living Plants in Open and Protected Ground,” no. USU 44053, were used.
Funding
This study was carried out as part of State Task project no. АААА-А21-121011290025-2 “Evaluation of the Morphogenetic Potential of Plant Populations in North Asia by Experimental Methods” and Budget Project no. FWNR-2022-0020 “Systems Biology and Bioinformatics: Reconstruction, Analysis and Modeling of the Structural and Functional Organization and Evolution of Human, Animal, Plant, and Microorganism Gene Networks.”
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest. The authors declare that they have no conflicts of interest.
Statement of the welfare of animals. The article does not contain any studies involving animals in experiments performed by any of the authors.
Additional information
Publisher’s Note.
Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fomin, E.S., Fomina, T.I. Phenological Reactions of Perennial Plants to Climate Change in Western Siberia. Contemp. Probl. Ecol. 16, 698–707 (2023). https://doi.org/10.1134/S1995425523060070
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1995425523060070