Abstract
Based on electron-microscope studies of the phytoplankton of two left-bank tributaries of the Cheboksary reservoir, 30 species of silica-scaled chrysophytes from the Chrysophyceae class, including 3 species new for the Russian flora and 1 species potentially new for science, have been found. Data on the abundance and biomass of phytoplankton with the role of chrysophyte algae are presented. The dominance of silica-scaled chrysophytes in spring and early summer algocenoses of the Kerzhenets River is noted.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
INTRODUCTION
Chrysophyte algae is a widespread group of freshwater phytoflagellates belonging to the Stramenopiles or Heterokontophyta groups, which plays an important role in the functioning of freshwater ecosystems, especially in reservoirs of medium and low productivity (Kristiansen, 1995; Gusev et al. 2020). The structure of their thalli is characterized by a wide variety of structural types, including species without hard cell integuments, with formations in the form of cellulosic lorica, as well as species with specialized silica formations (scales) covering cells (Voloshko, 2017b).
Early studies on phytoplankton of the Volga River (Pavlinova, 1930; Esyreva, 1935, 1945; Korshikov, 1939) provided lists of algal species, including Mallomonas spp., Synura uvella Ehrenberg emend. Korschikov, S. echinulata Korschikov, and S. petersenii Korshikov.
The flood control of the Volga River and the creation of a cascade of reservoirs stimulated the study of phytoplankton and the composition of chrysophyte algae. In the latest reports on the algal flora of the water bodies of the Volga River, information on 43 taxa of silica-scaled chrysophyte algae from the genus Mallomonas (Gusev et al., 2019), 18 species of the genus Synura, 4 species of Spiniferomonas, 4 species of Paraphysomonas, and 5 species of Chrysosphaerella is provided (Balonov, 1976; Korneva, 2015).
The diversity of phytoplankton of the Volga River is formed by, among other things, the impact of the lateral inflow system—small rivers that largely determine the hydrological and hydrochemical regimes and have a noticeable effect on the structure of the biota (Okhapkin, 1997; Vodeneeva, 2006). In the few available data on the composition of chrysophyte algae of some Volga tributaries (the Kerzhenets River), Mallomonas acaroides var. striatula Asmund, M. caudata Ivanov emend. Krieger, M. eoa Takahaschi in Asmund et Takahaschi, M. tonsurata Teiling emend. Krieger, Synura uvella Ehrenberg emend. Korschikov, S. petersenii Korschikov, S. petersenii var. glabra (Korschikov) Huber-Pestalozzi, and S. spinosa Korschikov (Balonov and Kuzmin, 1975; Balonov, 1976) were mentioned in the composition of phytoplankton. Information about silica-scaled chrysophytes of the Vetluga River is very limited. According to the results of a study of the phytoplankton of this river carried out in the 1970s, Mallomonas akrokomos Ruttner in Pascher, Mallomonas caudata, Mallomonas sp., and Synura verrucosa Pascher (Esyreva and Yulova, 1968; Okhapkin, 1997) were revealed; however, species identification was carried out only using light microscopy and requires confirmation by electron microscopy.
Despite the large amount of literature data on the composition and structure of phytoplankton in reservoirs and watercourses in the basin of the Volga River, there are few special taxonomic studies on the identification of silica-scaled chrysophytes using electron microscopy and molecular genetic methods (Korneva, 2006; Gusev et al., 2017; Gusev et al., 2019). Electron microscopy and the ultrastructure of scales have long been used in the study of the taxonomy of this group of organisms. Several genera have been described and studied exclusively using EM (SEM and TEM methods). The use of new approaches has shown the significance of small differences in the ultrastructure of scales and the possibility of their use in assessing evolutionary relationships (Škaloud et al., 2013a; Gusev et al., 2016).
The aim of this study was to analyze the species composition of silica-scaled chrysophytes using modern approaches (electron microscopy) for determining this group.
MATERIALS AND METHODS
In terms of morphological parameters (watershed and length), the studied watercourses are medium (Kerzhenets River) and large (Vetluga River) rivers. They are typically plain streams, with a pronounced spring flood and a predominance of the snow type of inflow. Flowing through flat terrain with a predominance of forests, lakes, and marshes, they have very curved and shallow channels, small longitudinal slopes, low flow rates, and well-developed valleys with gentle slopes passing into the surrounding area. The watershed area of the two studied rivers is flat, which significantly complicates the clear delineation of the boundaries of the catchment areas of these watercourses (Panfilov, 1974).
The river flow of the left-bank tributaries of the Middle Volga is formed in the subzone of the southern taiga with a forested and swampy watershed. This has a significant effect on the formation of the chemical composition of surface waters and is reflected in the nature and values of mineralization, the content of biogenic elements, and organic substances of humic origin (humic and fulvic acids) and it determines the increased water color index and pH value and reduces the content of HCO3 ions in comparison with the Volga River (Table 1).
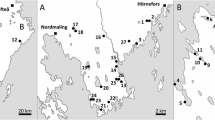
To investigate the composition of silica-scaled chrysophytes, phytoplankton samples collected during the navigation season were used. Samples were taken during the growing season (May to October) 2017 at a station near the village of Rustai (56°49′79.12″ N, 44°79′42.40″ E) from the Kerzhenets River; samples from the Vetluga River were obtained at the station near the village of Chernysikha (56°85′73.18″ N, 45°43′74.22″ E) in autumn (October 2019) (Fig. 1). We also analyzed archival materials of the authors on the seasonal dynamics of phytoplankton in 2014 and 2016.
During the study period, the temperature, pH, and electrical conductivity of the water were measured (Table 1).
Quantitative samples were collected in the surface layer of the flow (0.5 L) and fixed with an iodine-formalin fixative. Then the samples were concentrated by filtration through Vladipor membrane filters with an MFAS-M membrane with a disk diameter of 35 mm and a pore size of about 2 μm and the volume was adjusted to 5 mL.
To investigate the scales of golden algae, the samples were washed from an iodine-formalin fixative, incubated for 4–5 min in sulfuric acid with potassium dichromate, and washed with distilled water. Droplets of suspension were placed on formvar film-coated grids (EMS FF200-Cu-50, Electron Microscopy Sciences). The samples were examined under a JEM-1011 transmission electron microscope at the Center of Collective Use for Electron Microscopy at the Papanin Institute for Biology of Inland Waters, Russian Academy of Sciences.
RESULTS
In the phytoplankton of the studied watercourses, 30 species of the class Chrysophyceae—orders Mallomonadaceae, Synuraceae, Lepidochromonadaceae, and Paraphysomonadaceae—were found. Out of them, 19 species belong to the genus Mallomonas, 9 to Synura, 1 to Chrysosphaerella and 1 to Lepidochromonas. In the Kerzhenets River, all the revealed taxa of the genera Mallomonas, Synura, Chrysosphaerella, and Lepidochromonas were detected; only two species of the genus Synura were recorded in the Vetluga River.
A list of the species is provided below with a brief description of the morphology of scales and their distribution. The species are illustrated with original photographs of the scales obtained using a transmission microscope.
Mallomonas akrokomos Ruttner (Fig. 2a). Discovered in the Kerzhenets River for the first time. The species is widespread. It was recorded in the water bodies of northern Russia (Voloshko, 2017b) and in the Upper Volga (Balonov, 1976).
Scales of taxa of the genus Mallomonas: (a) M. akrokomos, (b–d) M. annulata, (e) M. alata, (f) M. allorgei, (g) M. calceolus, (h) M. сaudata, (i) M. сostata, (j) M. сrassisquama var. crassisquama, (k) M. heterospina, (l–n) M. jubata, (o) M. mangofera, (p) M. multinuca, (q, r) M. munda, and (s, t) M. papillosa var. papillosa.
M. annulata (Bradley) Harris 1967 (Figs. 2b–2d). Discovered in the Kerzhenets River for the first time. The species is widespread in Europe, Central and South America, Asia, and Australia (Kristiansen, Preisig, 2007). In the territory of Russia, it was noted in the Volga River (Balonov, 1976) and in the water bodies of northern Russia (Voloshko, 2017b).
M. alata Asmund, Cronberg et Dürrschmidt (Fig. 2e). Discovered in the Kerzhenets River for the first time. The species is widespread (Kristiansen, Preisig, 2007). Found in European countries and Russia (Voloshko, 2017b).
M. allorgei (Deflande) Conrad (Fig. 2f). Discovered in the Kerzhenets River for the first time. The species is widespread and was revealed in the water bodies of Europe, North, Central and South America, Africa, and Australia (Kristiansen, Preisig, 2007). In Russia it was recorded in northern water bodies (Voloshko, 2017b).
M. calceolus Bradley (Fig. 2g). Discovered in the Kerzhenets River for the first time. The species is widespread (Kristiansen, Preisig, 2007). In Russia it was recorded in Karelia (Balonov, 1979), Nevskaya Bay, Vologda oblast, and water bodies of the Bolshezemelskaya tundra (Voloshko, 2017b; Siver et al., 2005).
M. сaudata Ivanov emend. Krieger (Fig. 2h). Previously revealed in the plankton of the Kerzhenets River and Volga reservoirs (Balonov, Kuzmin, 1975). One of the most widespread species, cosmopolitan (Kristiansen, 2005) and oligosaprobiont (Watson, Satchwill, 2003). In addition to the reservoirs of the Volga cascade, it was found in large tributaries of the Volga (Shosha, Soz, Oka, Kama) (Balonov and Kuzmin, 1975).
M. сostata Dürrschmidt (Fig. 2i). Discovered in the Kerzhenets River for the first time. The species is widespread; it was found in water bodies of Europe, North America, Asia, Australia, and in water bodies of northern Russia (Bolshezemelskaya and Vorkutinskaya tundras) (Siver et al., 2005; Voloshko, 2017b). Acidophilus, in water bodies with low salinity.
M. crassisquama (Asmund) Fott var. crassisquama (Fig. 2j). Discovered in the Kerzhenets River for the first time. The species is widespread, especially in temperate latitudes (Kristiansen, Preisig, 2007). Recorded in water bodies of northern Russia (Voloshko, 2017b).
M. heterospina Lund (Fig. 2k). Discovered in the Kerzhenets River for the first time. The species is widespread (Kristiansen and Preisig, 2007). It was recorded in the plankton of the Rybinsk Reservoir (Balonov and Kuzmin, 1975) and in water bodies of northern Russia (Voloshko, 2017b).
M. jubata Němcová, Kreidlová, Pusztai & Neustupa (Figs. 2l–2n). A recently described species isolated from the M. pumilio group (Němcová et al., 2013). First find in the territory of Russia. Recorded in the Kerzhenets River. The scales of the species are characterized by a number of morphological features. Apical scales forming the collar are asymmetric, elongated, with an expressed dome (Fig. 2l). Body scales are rhombic without a dome (Fig. 2m, 2n). The reticulum of the shield consists of irregular cells and contains one to three pores. The size of the scales in the population from the Kerzhenets River ranged from 3.9 to 4.7 µm in length and 2.6 to 3.1 µm in width. Distributed in European countries (France and Greece) in small ponds, basins, and sphagnum bogs. Occurs at pH 4.6–7.2 (Němcová et al., 2013).
M. mangofera Harris et Bradley (Fig. 2o). Discovered in the Kerzhenets River for the first time. The species was described from the water bodies of the United Kingdom (Harris and Bradley, 1960); it was found in other regions of Europe (Němcová et al., 2012; Škaloud et al., 2013b). The species is considered cosmopolitan (Kristiansen and Preisig, 2007); however, recent studies have shown that the detected morphotype is characteristic in the European territory, while morphotypes from other regions (South America, tropical Asia, and Africa) are other species (Gusev et al., 2019). In Russia, it was previously recorded only in the northwest of the European part (Safronova, 2014).
M. multiunca Asmund (Fig. 2p). Discovered in the Kerzhenets River for the first time. The species is widespread. Recorded in water bodies of northern Russia (Voloshko, 2017b) and reservoirs of the Volga cascade (Balonov and Kuzmin, 1975) and in Europe, North and Central America, and Asia (Kristiansen and Preisig, 2007).
M. munda (Asmund, Cronberg & Dürrschmidt) Němcová (Figs. 2q, 2r). Discovered in the Kerzhenets River for the first time. Initially, scales of this type were described as M. pumilio var. munda Asmund, Cronberg & Dürrschmidt (Dürrschmidt, 1982). Geometric morphometry allowed us to increase the rank of the variety to species, distinguishing it from the M. pumilio group (Němcová et al., 2013). Bipolar species (Kristiansen and Preisig, 2007). In Russia it was recorded in water bodies of the Bolshezemelskaya tundra (Siver et al., 2005), in the karst lakes of Central Russia (Gusev, 2013), and in small water bodies in Leningrad oblast. (Safronova, 2014).
M. papillosa Harris et Bradley var. papillosa (Figs. 2s, 2t) was discovered in the Kerzhenets River for the first time. (Kristiansen and Preisig, 2007). The species was found in the Volga River (Balonov, 1976) and the water bodies of northern Russia (Voloshko, 2017b).
M. punctifera Korschikov var. punctifera (Figs. 3a, 3b). Discovered in the Kerzhenets River for the first time. The species is widespread, common in northern and temperate latitudes (Kristiansen and Preisig, 2007). It was recorded in the plankton of the Volga reservoirs (Balonov and Kuzmin 1975) and in the water bodies of northern Russia (Voloshko, 2017b).
Scales of taxa of the genus Mallomonas, Synura, Chrysosphaerella, and Lepidochromonas: (a, b) Mаllomonas punctifera var. punctifera, (c, d) M. retifera, (e) M. striata var. striata, (f) Mallomonas sp., (g) M. tonsurata, (h–j) Synura echinulata, (k, l) S. multidentata, (m) S. punctulosa, (n) S. spinosa f. spinosa, (o) S. spinosa f. longispina, (p, q) Synura uvella, (r, s) Chrysosphaerella brevispina, and (t) Lepidochromonas poteriophora.
M. retifera Dürrschmidt (Figs. 3c, 3d). Recorded for the first time in the territory of Russia in the Kerzhenets River. The scales are three-sided, slightly asymmetric, 4.7–4.8 µm long and 2.1–2.9 µm wide. They have a dome with scattered papillae. There is a V-shaped rib with expressed hood. The anterior submarginal rib is thick. The posterior edge is wide, smooth; the anterior margin is narrow with rows of papillae. It occurs in Europe (Pichrtova et al., 2007; Němcová et al., 2012), South America (Dürrschmidt, 1982), and East Asia (Kristiansen and Preisig, 2007). It is a rare species.
M. striata Asmund var. striata (Fig. 3e). Discovered in the Kerzhenets River. A widespread species, noted in the plankton of the Volga reservoirs and in the large Volga tributaries (Shosha, Soz, Oka, Kerzhenets, and Kama) (Balonov and Kuzmin, 1975).
M. sp. (Fig. 3f). Discovered in the Kerzhenets River. The scale that was found does not match the description of the previously known species. Belongs to the Striatae Asmund & Kristiansen section. Scales are elliptical, 4.6 µm long and 2.8 µm wide. The dome of the scale is almost rounded, with longitudinal ribs. The shield has 18 arcuate curved transverse ridges. It has a widely rounded V-rib with a well-defined hood. Several large pores are located in the corner of the V-shaped rib. The anterior edge with ribs. It is a new species for science, requiring a more detailed study for the description.
M. tonsurata Teiling emend. Krieger (Fig. 3g). Discovered in the Kerzhenets River. Previously the species was noted in the plankton of the Kerzhenets River (Balonov and Kuzmin, 1975). Cosmopolite, revealed in the Volga reservoirs and the delta of the Volga River, and recorded in the north of European Russia (Balonov and Kuzmin, 1975; Voloshko, 2017b).
Chrysosphaerella brevispina Korschikov (Figs. 3r, 3s). Registered in the Kerzhenets River; widespread (cosmopolitan), common in freshwater plankton: rivers, lakes, reservoirs, and sphagnum bogs (Snit’ko et al., 2016; Voloshko, 2017b).
Lepidochromonas poteriophora (Moestrup & Kristiansen) Kapustin & Guiry (syn. Paraphysomonas poteriophora Moestrup & Kristiansen, Clathromonas poteriophora Scoble & Cavalier-Smith) (Fig. 3t). First discovered in the territory of Russia in the Kerzhenets River. It is characterized by reticulate scales, rounded at the base, forming a protrusion (outgrowth), expanding at the end (Thomsen et al., 1981). It was found in only five European habitats in Denmark (Thomsen et al., 1981), Germany, Great Britain (Finlay and Clarke, 1999), and the Czech Republic (Škaloud et al., 2013b). A revision of the genus Paraphysomonas carried out in 2014 using molecular genetic methods (Scoble and Cavalier-Smith, 2014) showed the importance of a number of characters (the shape of the basal plate, the length and shape of the spine, crown scales, etc.) for the identification of the species. As a result, a new genus was identified, Clathromonas Scoble & Cavalier-Smith, whose representatives were characterized by scales with a reticular structure. However, this genus is invalid (Art. 52.1 of the International Code of Nomenclature for Algae, Fungi, and Plants), since it includes the phylum of the genus Lepidochromonas Kristiansen—L. diademifera (E. Takah.) Kristiansen. The authors of the study (Kapustin and Guiry, 2019) restored the forgotten genus Lepidochromonas and transferred the species of the genus Clathromonas into it.
Synura echinulata Korschikov (Figs. 3h–3j). Discovered in the Kerzhenets River for the first time. The species is widespread. Previously it was detected in the Upper Volga basin (Balonov and Kuzmin, 1974), in the water bodies of northern Russia (Voloshko, 2017b), and in small rivers of anthropogenically transformed landscapes (Gusev et al., 2017). It is typical for fresh water bodies of different types with pH from neutral to slightly acidic.
S. multidentata (Balonov et Kuzmin) Péterfi et Momeu (Figs. 3k, 3l). Discovered in the Kerzhenets River for the first time. The species is confined to the water bodies of temperate latitudes. Found in Europe and North America. In the territory of Russia it was recorded in the Upper Volga (as a variety of S. echinulata f. mulidentata Balonov et Kuzmin), in the water bodies of the Vologda region (Balonov, 1980), in the Chukotka Peninsula, and in the basin of the Kolyma River (Kuzmin, 1985).
S. punctulosa Balonov (Fig. 3m). Discovered in the Kerzhenets River for the first time. The species is rare, being recorded in temperate latitudes. It was detected in Europe and Asia. In Russia it was registered in the basins of the Upper Volga (Balonov, 1976) and the Lower Yenisei (Bessudova, 2016).
S. spinosa Korshikov f. spinosa (Fig. 3n). The species was previously found in the Kerzhenets River (Balonov and Kuzmin, 1974). It was registered for the first time in the Vetluga River. Cosmopolitan. It was noted in most of the studies devoted to the silica-scaled chrysophytes (Balonov, 1976; Balonov, 1979; Balonov and Kuzmin, 1986; Siver et al., 2005; Voloshko, 2010; Safronova and Voloshko, 2013; Voloshko, 2017b; Gusev et al., 2017).
S. spinosa f. longispina Petersen et Hansen (Fig. 3o). Discovered in the Kerzhenets River for the first time. The species is widespread, but it was infrequently detected. In Russia, it was shown to exist in the basin of the Volga River (Balonov and Kuzmin, 1974; Balonov, 1976), the water bodies of northern European Russia (Voloshko, 2017b), and the Polar Urals (Voloshko, 2010). There were findings in the small rivers of the urban area (Gusev et al., 2017).
S. uvella Ehrenberg emend. Korschikov (Figs. 3p, 3q). It was previously noted in the plankton of the Kerzhenets River (Balonov and Kuzmin, 1974). Cosmopolitan. Found in the water bodies of the Volga basin (Balonov, 1976), in northern European Russia (Voloshko and Gavrilova, 2001; Safronova and Voloshko, 2013), in the polar Urals (Voloshko, 2010), in the karst lakes of the Vladimir oblast (Gusev, 2013), and in a small river in Nizhny Novgorod (Gusev et al., 2017).
Identifying representatives of the genus Synura from the Peterseniae section is particularly difficult, since the determination is carried out based on the size of the scales and their structural elements (Table 2, Fig. 3).
S. conopea Kynčlova et Škaloud (Figs. 4a, 4b; Table 2). Discovered in the Kerzhenets River for the first time. Earlier the species was recorded in small urban watercourses in the city of Nizhny Novgorod (Gusev et al., 2017). Found in the water bodies of Europe and South America (Škaloud et al., 2012).
S. glabra Korshikov em. Škaloud et Kynčlova in Škaloud (Figs. 4c–4e, Table 2). The species was revealed in the Kerzhenets River. Previously was noted as S. petersenii var. glabra (Balonov and Kuzmin, 1974). A widespread species (Balonov and Kuzmin, 1974; Balonov, 1976; Kuzmin, Kuzmina 1987; Voloshko, 2010; Voloshko, 2017b; Gusev et al., 2017). The species was found in different types of water bodies.
S. petersenii Korshikov (Figs. 4f–4j; Table 2). Found in all studied watercourses (in Kerzhenets and Vetluga rivers). Previously recorded in the plankton of the Kerzhenets River (Balonov and Kuzmin, 1974). The species is widespread (Škaloud et al., 2012; Boo et al., 2010). Identification based on the structure of the scales does not allow making an unambiguous conclusion about the taxonomic affiliation, since confusion with other previously described species of the S. petersenii complex is possible. Molecular genetic analysis based on strains from the collection of algae of the Papanin Institute for Biology of Inland Waters, Russian Academy of Sciences (Gusev et al., 2016), confirmed the presence of this species in European Russia.
DISCUSSION
Despite more than a century of studying the phytoplankton of water bodies and watercourses in the Nizhny Novgorod region, specialized studies of chrysophyte algae using electron microscopy only started in the 1970s. (Balonov, 1976) and continued further based on the example of small rivers in the city of Nizhny Novgorod (Gusev et al., 2017; Gusev et al., 2019). The lists of algal flora provided in studies of A.G. Okhapkin and E.L. Vodeneeva contain information on the composition of chrysophyte algae on the left-bank Volga tributaries (Okhapkin, 1997; Vodeneeva, 2006); however, these findings were not confirmed by electron microscopic studies and required clarification.
Chrysophyte algae are an important component of algocenoses in the total abundance and biomass of phytoplankton in the studied watercourses. As a rule, the seasonal dynamics of phytoplankton in the studied watercourses is characterized by three increases in biomass: in spring, summer, and autumn. Diatom and chrysophyte algae provide the spring and autumn development of phytoplankton; summer development is provided by green algae, diatoms, often with the involvement of chrysophyte algae and blue-green algae.
The average long-term abundance and biomass of phytoplankton in the Kerzhenets River for the growing seasons of 2014–2016 did not differ significantly; they ranged from 1.95 ± 0.78 to 4.79 ± 0.29 million cells/L and from 1.39 ± 0.24 to 2.07 ± 0.68 g/m3 and corresponded to value of these indicators of mesotrophic waters. The amount of phytoplankton in the Vetluga River varied from 4.34 ± 0.76 to 6.6 ± 1.3 million cells/L and biomass varied from 0.74 ± 0.11 to 1.51 ± 0.42 g/m3. The development of chrysophyte algae in the studied rivers was not the same throughout different periods of the hydrological cycle; the maximum values of indicators were recorded in spring. In the Vetluga River, the abundance of chrysophyte algae reached 33% of the total abundance (0.33 million cells/L) and 28% of the total biomass of phytoplankton (0.11 g/m3). Among chrysophyte algae, representatives of the genera Dinobryon (D. divergens O.E. Imhof, D. sociale (Ehrenberg) Ehrenberg), Chrysococcus (C. biporus Skuja) prevailed; the proportion of silica-scaled chrysophytes in the formation of the abundance and biomass of algocenoses was insignificant (<5%). The contribution of silica-scaled chrysophytes to the formation of plankton in the Kerzhenets River was more essential. In 2017, the dominance of species from the genus Mallomonas spp. and Synura spp. was noted in spring and early summer algocenoses; the abundance and biomass of these species reached 0.15–0.19 million cells/L and 0.12–0.16 g/m3, respectively.
Despite the low rates of development of silica-scaled chrysophytes in the studied watercourses, their composition turned out to be relatively rich (30 species from 4 genera). Among them, Mallomonas jubata, Mallomonas retifera, and Lepidochromonas poteriophora were revealed for the first time in Russia. Most of the species in the studied rivers were found for the first time. An analysis of the geographical distribution showed that most of the identified taxa are cosmopolitan and widespread species. A bipolar range is characteristic for a number of species (Mallomonas jubata, M. munda, and M. retifera); some taxa were recorded in northern and temperate latitudes (M. allorgei, M. multinuca, M. punctifera, and Synura multidentata).
The high taxonomic diversity of this group is in good agreement with the results of other studies in the region (Gusev et al., 2017, 2019). This diversity was probably influenced by the unique location of the studied watercourses in the zone of the boreal-nemoral ecotone, the boreal part of which is characterized by the predominance of forests, lakes, and bogs. Despite the high species richness, silica-scaled chrysophytes occupy a leading position only in the spring algocenoses of the Kerzhenets River.
Most of the species were found in the Kerzhenets River, characterized by oligo-acidic waters with an increased value of water color index. In a number of studies (Ilmavirta, 1988; Korneva, 2006; Voloshko, 2017a) it was noted that the main factor forming the composition of the flora of chrysophyte algae in lake ecosystems is the pH of the water, and their representation in algocenoses is determined by a set of indicators: pH, trophy level, water color index, and total nitrogen concentration. The presence of rivers with swampy watersheds has a significant effect on the composition of the flora of chrysophyte algae. Studies carried out earlier based on the example of the rivers of Nizhny Novgorod, flowing from bogs, showed a high diversity of silica-scaled chrysophytes (Gusev et al., 2017; Gusev et al., 2019).
CONCLUSIONS
The species composition of silica-scaled chrysophytes of two left-bank tributaries of the Volga River is rich and includes 30 species and intraspecific taxa from the genera Mallomonas, Synura, Chrysosphaerella, and Lepidochromonas from four families with a predominance of species of the genus Mallomonas. The species richness of the studied group of chrysophytes is definitely associated with the set and variety of environmental factors formed in watercourses and maintaining high species richness (variation of pH values characteristic of oligo-acidic-neutral waters, high water color indices and trophy of waters, and biotopic heterogeneity of river systems). Most species have a wide geographical distribution, three of them were characterized by a bipolar range, three species (Mallomonas jubata, Mallomonas retifera, and Lepidochromonas poteriophora) were discovered in Russia for the first time, and one species is potentially new to science. The indicators of the quantitative development of the studied algae group, in contrast to their species richness, turned out to be insignificant; however, the maximum abundance and biomass observed in the spring algocenoses of the Kerzhenets River reached 48 and 43% of the total indicators, respectively.
Change history
12 March 2022
An Erratum to this paper has been published: https://doi.org/10.1134/S199508292207001X
REFERENCES
Balonov, I.M., Vidy sem. Synuraceae Lemm. (Chrysophyta) Volgi i ee basseina (Species of the Family Synuraceae Lemm. (Chrysophyta) of the Volga River and Its Basin), Biol. Vnutr. Vod: Inf. Byull., Leningrad: Nauka, 1976, no. 29, p. 16.
Balonov, I.M., Chrysophytes of the family Synuraceae Lemm. of water bodies of Karelia, Tr. Inst. Biol. Vnutr. Vod, Akad. Nauk SSSR, 1979, no. 42 (45), p. 3.
Balonov, I.M., Zolotistye vodorosli vodoemov Vologodskoi oblasti (Chrysophytes of Water Bodies of the Vologda Oblast), Biol. Vnutr. Vod: Inf. Byull., Leningrad: Nauka, 1980, no. 45, p. 31.
Balonov, I.M. and Kuz’min, G.V., Electron microscopic study of species of the genus Mallomonas Petry (Chrysophyta) from the Volga cascade reservoirs. I. Series Planae Harris et Bradley, Bot. Zh., 1975, vol. 60, no. 9, p. 1142.
Balonov, I.M. and Kuz’mina, A.E., Zolotistye vodorosli. Gidrokhimicheskie i gidrobiologicheskie issledovaniya Khantaiskogo vodokhranilishcha (Chrysophytes. Hydrochemical and Hydrobiological Studies of the Khantai Reservoir), Tr. Limnol. Inst., Sib. Otd., Akad. Nauk SSSR, Novosibirsk: Nauka, 1986, p. 59.
Bessudova, A.Yu., Cheshuichatye zolotistye vodorosli basseina Nizhnego Eniseya i zalivov Karskogo morya s elementami autekologii (Silica-Scaled Chrysophytes of the Lower Yenisei Basin and the Kara Sea Bays with Elements of Autecology), Irkutsk: Inst. Geogr. im. V. B. Sochavy, Sib. Otd., Ross. Akad. Nauk, 2016.
Boo, S.M., Kim, H.S., Shin, W., et al., Complex phylogeographic patterns in the freshwater alga Synura provide new insights into ubiquity vs endemism in microbial eukaryotes, Mol. Ecol., vol. 19, no. 19, p. 4328. https://doi.org/10.1111/j.1365-294X.2010.04813.x
Dürrschmidt, M., Studies on the Chrysophyceae from South Chilean inland waters by means of scanning and transmission electron microscopy II, Arch. Hidrobiol. Algol. Stud., 1982, vol. 31, suppl. 63, p. 121.
Esyreva, V.I., Phytoplankton of the Volga River according to observations in 1931 on the section in front of Gorky, Uch. Zap. Gor’k. Univ., 1935, no. 4, p. 83.
Esyreva, V.I., Algal flora of the Volga River from Rybinsk to Gorky, Tr. Bot. Sada, Mosk. Gos. Univ., 1945, book 5, no. 82, p. 10.
Esyreva, V.I. and Yulova, G.A., Some data on the algae of the Vetluga River according to observations in 1966, Uch. Zap. Gor’k. Univ., 1968, no. 90, p. 118.
Finlay, B.J. and Clarke, K.J., Apparent global ubiquity of species in the protist genus Paraphysomonas, Protist, 1999, vol. 150, p. 419.
Gusev, E.S., Silica-scaled chrysophytes from karst lakes in Central Russia, Nova Hedwigia. Beiheft, 2013, vol. 142, p. 17.
Gusev, E.S., On the flora of scaly golden algae of Lake Frolikha (northern Transbaikalia), Tr. Inst. Biol. Vnutr. Vod, Ross. Akad. Nauk, 2016, no. 76 (79), p. 25.
Gusev, E.S., Kapustin, D.A., and Martynenko, N.A., Morphological and molecular genetic study of species of the genus Synura Ehrenb. (Chrysophyceae) from the collection of the Papanin Institute for Biology of Inland Waters, Russian Academy of Sciences, in Molekulyarnaya genetika gidrobiontov (Molecular Genetics of Aquatic Organisms), Tr. Inst. Biol. Vnutr. Vod, Ross. Akad. Nauk, Borok, 2016, no. 73 (76), p. 5.
Gusev, E.S., Perminova, O.S., Startseva, N.A., and Okhapkin, A.G., Genus Synura (Synurales, Synurophyceae) in small urban rivers of Nizhny Novgorod, Nov. Sist. Nizshikh Rast., 2017, no. 51, p. 57. https://doi.org/10.31111/nsnr/2017.51.57
Gusev, E.S., Perminova, O.S., Guseva, E.E., and Startseva, N.A., The genus Mallomonas in small urban rivers in Nizhniy Novgorod (Russia), Nova Hedwigia Beiheft, 2019, vol. 148, p. 77. https://doi.org/10.1127/nova-suppl/2019/091
Gusev, E.S., Gusakov, V.A., Guseva, E.E., et al., Flora of silica-scaled chrysophytes (Chrysophyceae: Synurales, Paraphysomonadales) of the Mekong Delta, Inland Water Biol., 2020, vol. 13, no. 3, p. 349. https://doi.org/10.1134/S1995082920020224
Harris, K. and Bradley, D.E., A taxonomic study of Mallomonas, J. Gen. Microbiol., 1960, vol. 22, p. 750.
Ilmavirta, V., Phytoflagellates and their ecology in Finnish broun-water lakes. Flagellates in freshwater ecosystems, Microbiol. Sci. Dev. Hydrobiol., 1988, vol. 45, p. 255.
Kapustin, D.A. and Guiry, M.D., Reinstatement of Lepidochromonas kristiansen (Lepidochromonadaceae fam. nov., Chrysophyceae), Phytotaxa, 2019, vol. 413, no. 1, p. 049. https://doi.org/10.11646/phytotaxa.413.1.5
Korneva, L.G., Fitoplankton vodokhranilishch basseina Volgi (Phytoplankton of Reservoirs in the Volga River Basin), Kostroma: Kostr. Pechatn. Dom, 2015.
Korneva, L.G., Taxonomic composition and ecology of golden algae (Chrysophyta) in low-mineralized forest lakes of the Vologda oblast, Biol. Vnutr. Vod, 2006, no. 2, p. 3.
Korshikov, A.A., Materials for the knowledge of algae in the Gorky oblast. Phytoplankton of the Oka River in August 1932, Uch. Zap. Gor’k. Univ., 1939, no. 9, p. 101.
Kristiansen, J., History of chrysophyte research, in Chrysophyte Algae. Distribution, Phylogeny and Development, New York: Cambridge Univ. Press, 1995, p. 1.
Kristiansen, J., Golden Algae. A Biology of Chrysophytes, Koenigstein: Gantner, 2005.
Kristiansen, J. and Preisig, H.R., Chrysophyte and haptophyte algae. Part 2: Synurophyceae, in Süsswasserflora von Mitteleuropa, Berlin: Springer-Verlag, 2007, vols. 1/2.
Kuz’min, G.V., Vidovoi sostav fitoplanktona vodoemov zony zatopleniya Kolymskoi GES (Species Composition of Phytoplankton in Water Bodies in the Flood Zone of the Kolyma HPP), Magadan: Inst. Biol. Probl. Severa, Dal’nevost. Nauchn. Tsentra, Akad. Nauk SSSR, 1985.
Kuz’min, G.V. and Kuz’mina, V.A., Testate representatives of golden algae from the Magadan oblast, Nov. Sist. Nizshikh Rast., 1987, vol. 23, p. 40.
Němcová, Y., Kreidlová, J., Kosová, A., and Neustupa, J., Lakes and pools of Aquitaine region (France)—a biodiversity hotspot of Synurales in Europe, Nova Hedwigia Beiheft, 2012, vol. 95, nos. 1–2, p. 1.
Němcová, Y., Kreidlová, J., Pusztai, M., and Neustupa, J., Mallomonas pumilio group (Chrysophyceae/Stramenopiles)—a revision based on the scale/scale-case morphology and analysis of scale shape, Nova Hedwigia Beiheft, 2013, vol. 142, p. 27.
Okhapkin, A.G., The structure and succession of phytoplankton in the regulation of river flow (as exemplified by the Volga River and its tributaries), Doctoral (Biol.) Dissertation, St. Petersburg, 1997.
Panfilov, V.D., Waters, in Priroda Gor’kovskoi oblasti (The Nature of the Gorky Oblast), Gorky: Volgo-Vyatskoe Knizhn. Izd., 1974, p. 146.
Pavlinova, R.M., Biological survey of the Volga River in the area from the Gorodets town to the Sobchinskii backwater in 1926–1927, Tr. Inst. Sooruzh. Tsentr. Kom. Vodookhran., 1930, no. 11, p. 3.
Pichrtova, M., Rezacova-Skaloudova, M., and Skaloud, P., Silica-scaled chrysophytes of the Czech-Moravian Highlands, Fottea, 2007, vol. 7, no. 1, p. 43.
Safronova, T.V., Seasonal changes in the composition of golden algae (Chrysophyceae, Synurophyceae) in the ponds of the Botanical Garden of the Botanical Institute of the Russian Academy of Sciences (St. Petersburg), Bot. Zh., 2014, vol. 99, no. 4, p. 443.
Safronova, T.V. and Voloshko, L.N., Silica-scaled chrysophytes in the waterbodies of protected areas of the North-West of Russia, Nova Hedwigia Beiheft, 2013, vol. 142, p. 97.
Scoble, J.M. and Cavalier-Smith, T., Scale evolution in Paraphysomonadida (Chrysophyceae): sequence phylogeny and revised taxonomy of Paraphysomonas, new genus Clathromonas, and 25 new species, Protistologica, 2014, vol. 50, p. 551. https://doi.org/10.1016/j.ejop.2014.08.001
Siver, P.A., Voloshko, L.N., Gavrilova, O.V., and Getsen, M.V., The scaled chrysophyte flora of the Bolshezemelskaya tundra (Russia), Nova Hedwigia Beiheft, 2005, vol. 128, p. 125.
Škaloud, P., Kynčlová, A., Benada, O., et al., Toward a revision of the genus Synura, section Petersenianae (Synurophyceae, Heterokontophyta): morphological characterization of six pseudo-cryptic species, Phycologia, 2012, vol. 51, p. 303.
Škaloud, P., Kynčlová, J., and Škaloudova, M., Developments in the taxonomy of silica-scaled—from morphological and ultrastructural to molecular approaches, Nord. J. Bot., 2013a, vol. 31, p. 385.
Škaloud, P., Škaloudova, M., Pichrtová, M., et al., www.chrysophytes.eu—a database on distribution and ecology of silica-scaled chrysophytes in Europe, Nova Hedwigia Beiheft., 2013b, vol. 142, p. 141.
Snit’ko, L.V., Snit’ko, V.P., Blinov, I.A., and Voloshko, L.N., Golden algae (Chrysophyceae, Synurophyceae) in the water bodies of the Eastern foothills of the Southern and Middle Urals, Bot. Zh., 2016, vol. 101, no. 12, p. 1361.
Thomsen, H.A., Zimmermann, B., Moestrup, O., and Kristiansen, J., Some new freshwater species of Paraphysomonas (Chrysophyceae), Nord. J. Bot., 1981, vol. 1, p. 559. https://doi.org/10.1111/j.1756-1051.1981.tb00724.x
Vodeneeva, E.L., Composition and structure of phytoplankton of humous-acidic reservoirs (as exemplified by water bodies of the Kerzhenskii Reserve), Cand. Sci. (Biol.) Dissertation, Nizhny Novgorod, 2006.
Voloshko, L.N., The chrysophycean algae from glacial lakes of Polar Ural (Russia), Nova Hedwigia Beiheft, 2010, vol. 136, p. 191.
Voloshko, L.N., Chrysophytes of the North of Russia: ecological conditions of habitation, Astrakh. Vestn. Ekol. Obraz., 2017a, no. 4 (42), p. 45.
Voloshko, L.N., Zolotistye vodorosli vodoemov Severa Rossii (Chrysophytes of Water Bodies of the North of Russia), St. Petersburg: Renome, 2017b.
Voloshko, L.N. and Gavrilova, O.V., A checklist of silica-scaled chrysophytes in Russia with an emphasis on the flora of Lake Ladoga, Nova Hedwigia Beiheft, 2001, vol. 122, p. 147.
Watson, S.B., Satchwill, T., Dixon, E., and McCauley, E., Under-ice blooms and source-water odour in a nutrient-poor reservoir: biological, ecological and applied perspectives, Freshwater Biol., 2001, vol. 46, p. 1553.
Watson, S.B. and Satchwill, T., Chrysophyte odour production: the impact of resources at the call and population levels, Phycologia, 2003, vol. 42, p. 393.
ACKNOWLEDGMENTS
We are grateful to the staff at the Center of Collective Use for Electron Microscopy at the Papanin Institute for Biology of Inland Waters, Russian Academy of Sciences
Funding
This study was supported by the Russian Foundation for Basic Research, project no. 20-34-90144.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests. The authors declare that they have no conflict of interest.
Statement on the welfare of animals. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Translated by V. Mittova
Abbrevistions: EM—electron microscopy.
The original online version of this article was revised: The title of the article should read as follows: Taxonomic Composition and Morphology of Silica-Scaled Chrysophytes of Some Left-Bank Volga Tributaries
Rights and permissions
About this article
Cite this article
Kulizin, P.V., Gusev, E.S., Vodeneeva, E.L. et al. Taxonomic Composition and Morphology of Silica-Scaled Chrysophytes of Some Left-Bank Volga Tributaries. Inland Water Biol 14, 357–367 (2021). https://doi.org/10.1134/S1995082921030093
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1995082921030093