Abstract
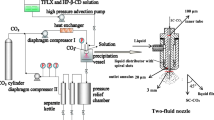
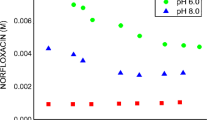
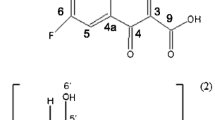
A new method for the synthesis of guest–host inclusion complex of moxifloxacin (MF) with 2-hydroxypropyl-β-cyclodextrin (HPCD) by supercritical fluid technology in the rapid expansion of supercritical solutions (RESS) mode was suggested for developing new drug formulations with improved bioaccessibility. The MF–HPCD complex was synthesized by this method as particles of 2–4 μm, which is the optimum size for creating the oral and inhalation MF drug forms. According to the scanning electron microscopy data, the morphology of MF–HPCD particles (irregular polyhedra) obtained by RESS processing differs from that of the starting components and from that of unbound MF and HPCD obtained by lyophilization from aqueous solution. According to X-ray diffraction data, the crystallinity of MF decreased from 95% to 20–30% after the formation of the HPCD complex. The IR spectroscopic and equilibrium dialysis data showed that RESS provides higher efficiency of drug inclusion in the complex with HPCD compared with that of conventional methods such as lyophilization or mixing of solid components.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
N. R. Cozarelli, Science (Washington, DC, U. S.) 207, 953 (1980).
F. Schmitz, B. Hofmann, B. Hansen, S. Scheuring, and M. J. Lückefahr, Antimicrob. Chemother. 41, 481 (1998).
A. Bryskier and J. Lowther, Expert Opin. Invest. Drugs 11, 233 (2002).
S. H. Gillespie, Eur. Respir. Rev. 25, 19 (2016).
M. Fouad and J. C. Gallagher, Ann. Pharmacother. 45, 1439 (2011).
E. N. Padeiskaya and V. P. Yakovlev, Fluoroquinolones (Bioinform, Moscow, 1995) [in Russian].
I. M. Le-Deygen, A. A. Skuredina, and E. V. Kudryashova, Russ. J. Bioorg. Chem. 43, 487 (2017).
N. Bandi, W. Wei, C. B. Roberts, L. P. Kotra, and U. B. Kompella, Eur. J. Pharm. Sci. 23, 159 (2004).
E. Fenyvesi, Cyclodext. News 27, 1 (2013).
I. M. Deygen, A. M. Egorov, and E. V. Kudryashova, Vestn. Mosk. Univ., Ser. 2: Khim. 56, 387 (2015).
A. A. Skuredina, I. M. Le-Deygen, I. V. Uporov, and E. V. Kudryashova, Colloid J. 79, 668 (2017).
Y. Guo, S. R. Byrn, and G. Zografi, Pharm. Res. 17, 930 (2000).
V. N. Bagratashvili, A. M. Egorov, L. I. Krotova, A. V. Mironov, V. Ya. Panchenko, O. O. Parenago, V. K. Popov, I. A. Revelsky, P. S. Timashev, and S. I. Tsypina, Russ. J. Phys. Chem. B 6, 804 (2012).
V. N. Bagratashvili, S. E. Bogorodskii, A. M. Egorov, L. I. Krotova, V. K. Popov, and V. I. Sevast’yanov, Russ. J. Phys. Chem. B 10, 1123 (2016).
M. J. Whitaker, J. Hao, O. R. Davies, G. Serhatkulu, S. Stolnik-Trenkic, S. M. Howdle, and K. M. Shakesheff, J. Control. Release 101, 85 (2005).
M. Sauceau, E. Rodier, and J. Fages, J. Supercrit. Fluids 47, 326 (2008).
A. M. Vorobei, O. I. Pokrovskiy, K. B. Ustinovich, L. I. Krotova, O. O. Parenago, and V. V. Lunin, Russ. J. Phys. Chem. B 10, 1072 (2016).
K. Moribe, Y. Tozuka, and K. Yamamoto, Adv. Drug Deliv. Rev. 60, 328 (2008).
E. V. Kudryashova, I. M. Deygen, K. V. Sukhoverkov, L. Yu. Filatova, N. L. Klyachko, A. M. Vorobei, O. I. Pokrovskiy, K. B. Ustinovich, O. O. Parenago, E. N. Antonov, A. G. Dunaev, L. I. Krotova, V. K. Popov, and A. M. Egorov, Russ. J. Phys. Chem. B 10, 1201 (2016).
S. R. Rudrangi, W. Kaialy, M. U. Ghori, V. Trivedi, M. J. Snowden, and B. D. Alexander, Eur. J. Pharm. Biopharm. 104, 164 (2016).
A. H. Al-Marzouqi, B. Jobe, A. Dowaidar, F. Maestrelli, and P. Mura, J. Pharm. Biomed. Anal. 43, 566 (2007).
S. R. S. Rudrangi, V. Trivedi, J. C. Mitchell, S. R. Wicks, and B. D. Alexander, Int. J. Pharm. 494, 408 (2015).
A. H. Al-Marzouqi, I. Shehatta, B. Jobe, and A. Dowaidar, J. Pharm. Sci. 95, 292 (2006).
T. Toropainen, S. Velaga, T. Heikkila, L. Matilainen, P. Jarho, J. Carlos, et al., J. Pharm. Sci. 95, 2235 (2006).
M. Charoenchaitrakool, F. Dehghani, and N. R. Foster, in Proceedings of the 5th International Symposium on Supercritical Fluids, Atlanta, USA, 2000.
S. Sahoo, C. K. Chakraborti, S. C. Mishra, U. N. Nanda, and S. Naik, J. Pharm. Res.4, 1129 (2011).
V. L. Dorofeev, Khim.-Farm. Zh., No. 38, 45 (2004).
K. P. Sambasevam, S. Mohamad, N. M. Sarih, and N. A. Ismail, Int. J. Mol. Sci. 14, 3671 (2013).
N. F. A. Dsugi and A. A. Elbashir, Spectrochim. Acta A: Mol. Biomol. Spectrosc. 137, 804 (2015).
S. D. Bhise, Int. J. Res. Pharm. Biomed. Sci. 2, 596 (2011).
M. Charoenchaitrakool, F. Dehghani, N. R. Foster, and H. K. Chan, Ind. Eng. Chem. Res. 39, 4794 (2000).
M. Charoenchaitrakool, F. Dehghani, and N. R. Foster, Int. J. Pharm. 239, 103 (2002).
T. A. Martin, N. Bandi, R. Schulz, C. B. Roberts, and U. B. Kompella, AAPS Pharm. Sci. Tech. 3, 3 (2002).
P. Coimbra, D. Fernandes, M. H. Gil, and H. C. J. de Sousa, Chem. Eng. Data 53, 1990 (2008).
F. M. Gumerov, A. A. Sagdeev, and D. G. Amirkhanov, Substance Solubility in Supercritical Fluid Systems (Lap Lambert Academic, Saarbrücken, 2016) [in Russian].
A. H. Al-Marzouqi, A. Solieman, I. Shehadi, and A. J. Adem, Incl. Phenom. Macrocycl. Chem. 60, 85 (2008).
S. Zu, L. Yang, J. Huang, C. Ma, W. Wang, C. Zhao, and Y. Zu, Int. J. Mol. Sci.13, 8869 (2012).
I. M. Le-Deygen, A. A. Skuredina, I. V. Uporov, and E. V. Kudryashova, Anal. Bioanal. Chem. 409, 6451 (2017).
L. Gradon and T. R. Sosnowski, Adv. Powder Technol. 25, 43 (2014).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © K.V. Sukhoverkov, I.M. Le-Deygen, A.M. Egorov, E.V. Kudryashova, 2017, published in Sverkhkriticheskie Flyuidy. Teoriya i Praktika, 2017, Vol. 12, No. 4, pp. 66–83.
Rights and permissions
About this article
Cite this article
Sukhoverkov, K.V., Le-Deygen, I.M., Egorov, A.M. et al. Physicochemical Properties of the Inclusion Complex of Moxifloxacin with Hydroxypropyl-β-Cyclodextrin Synthesized by RESS. Russ. J. Phys. Chem. B 12, 1193–1204 (2018). https://doi.org/10.1134/S1990793118070126
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990793118070126