Abstract—
The results of investigation of electrogenic transport by the Na+,K+-ATPase, the enzyme providing the active transport of Na+ and K+ ions through cell membrane, are reviewed. The main contribution to electric current generated through the functioning of the Na+,K+-ATPase is assigned to the movements of ions in access channels—the channel-like structures connecting the ion binding sites with the solutions. The electrogenic transport was studied in a model system consisting of a bilayer lipid membrane with adsorbed membrane fragments containing the Na+,K+-ATPase. The impedance method applied to this study allowed the investigation of access channels in the Na+,K+-ATPase. The review notes a significant contribution of Yu.A. Chizmadzhev to the development of the theoretical model of transport processes in the Na+,K+-ATPase.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
INTRODUCTION
Studies of ion transport through the membrane began to develop intensively after the method of forming a bilayer lipid membrane, a simple model of a biological membrane suitable for measuring electric currents, was described in 1963 [1]. This membrane is a good insulator, but once proteins or simpler compounds are imbedded in the membrane, it can conduct an electric current, the measurement of which makes it possible to study the mechanisms of ionic permeability induced by these compounds. At the Institute of Electrochemistry, such studies were started at the initiative of A.N. Frumkin and considerably intensified owing to Yu.A. Chizmadzhev after the Laboratory of Bioelectrochemistry had been established. The studies were conducted at both theoretical and experimental level. Originally, their objects were simple compounds and peptides making the membrane permeable to alkaline metal or hydrogen ions. The theoretical models of such transport were described in the monograph [2]. Subsequently, more complex types of passive exchange transport of potassium and hydrogen ions induced by nigericin-like compounds [3–5], active proton transport performed by bacteriorhodopsin [6], and Na+,K+-ATPase performing active transport of potassium and sodium ions were the objects of research. Although the Na+,K+-ATPase is a rather complex molecular machine and its enzymatic cycle includes transitions between a large number of states, certain stages in these states are electrogenic, i.e., accompanied by electric charge transfer inside the membrane, and they can be studied by relatively simple methods. It has been shown that active ion transport is performed due to successive opening and closing of special protein structures, resembling an ion channels closed at one end. These structures are referred to as “access channels”. The access channels allow the transfer of potassium or sodium ions to the binding sites in the protein. The functioning cycle per se is referred to as “Post–Albers cycle” [7] (Fig. 1). The most significant feature of the Post–Albers cycle is the presence of two major protein conformations, E1 and E2, with open access of ions to the binding sites either from the cytoplasm or from the extracellular medium. The conformation transition from E1 to E2 can occur due to ATP hydrolysis, with the phosphate group remaining bound to the protein (phosphorylation state) and released only during the reverse transition from E2 to E1. Originally, the Post–Albers cycle was established in biochemical studies as a formal scheme, but later it was supported by protein structures in various states of this cycle determined by X-ray scattering techniques [8–13]. Similar mechanisms are typical of quite a large number of proteins performing active transport of different ions [14–17].
The simplified scheme of sodium ion transport by the Na+,K+-ATPase on the basis of the Post–Albers enzymatic cycle modified according to [18]. Left and right sides of the diagram represent the reactions on the cytoplasmic and extracellular sides of the membrane, respectively. E1 and E2 designate two major protein conformations; arrows indicate transitions between the states. Hypothetical states of the Na+,K+-ATPase, where protons can be involved to perform ion transport, are shown in a rectangular frame.
ELECTROGENIC TRANSPORT BY THE Nа+,K+-ATPase
Considerable progress in the studies of electrogenic transport performed by the Na+,K+-ATPase has been achieved by the works of several groups from Germany and the United States published in the 1980s. The ion transfer through the access channels between the solution and the binding site in protein is electrogenic and passive, i.e., the distribution of ions between the binding site and the solution can be affected by an electric field applied to the membrane [19–22]. A fast change in potential of the membrane with the Na+,K+-ATPase leads to the appearance of a transient electrical current due to redistribution of ions between the binding site and the solution which disappears after establishment of a new equilibrium. The study of this process, first discovered in electrophysiological studies in isolated cardiac myocytes [23], provided information about the properties of the access channel and the rate of ion transport in it, which allows the estimation of the channel depth. The latter can be determined by measuring the “dielectric coefficient”, which characterizes the “degree of electrogenicity” of ion transfer in the channel. This parameter defines the relative depth of the region in the membrane where charge transfer occurs as its contribution to the measured electric current. However, this is not really channel depth: the contribution to the current recorded in the external circuit depends not only on the value of charge shift but also on polarizability of the medium where this shift takes place [19, 24]. The dielectric coefficient determines the fraction of the potential applied to the membrane that affects charge transfer in the protein, or how much less charge is carried by the external circuit under short-circuit conditions compared to the charge carried inside the protein [25]. Electrogenic transport was studied in isolated cardiac myocytes, oocytes, squid giant axons, as well as in isolated membrane fragments (by the patch-clamp technique) [26–28]. Due to the fact that the electric current generated by the Na+,K+-ATPase is low compared to the typical currents recorded in ion channels, the standard patch-clamp technique proved to be inapplicable, and a “giant patch-clamp” technique was developed for recording such currents, which made it possible to greatly expand the area of an isolated membrane fragment [29, 30].
RESEARCH IN BILAYER MEMBRANES
The study of the Na+,K+-ATPase in bilayer lipid membranes (BLM), began with the works of two research teams from Germany [31, 32]. They failed to incorporate the Na+,K+-ATPase into a lipid bilayer, and membrane fragments containing this protein were adsorbed on the BLM surface; as a result, a sandwich-like structure consisting of two membranes was formed (Fig. 2).
(a) Schematic representation of BLM with the adsorbed membrane fragment containing the Na+,K+-ATPase [25]. The solid line shows the electric potential profile appearing as a result of charging of the contacting membranes during ion transfer by the Na+,K+-ATPase under short circuit conditions. (b) An equivalent electrical scheme: Cp, total capacitance of Na+,K+-ATPase-containing membrane fragments adsorbed on BLM; CF, capacitance of the BLM region contacting the fragments; Ip(t), current generated by all active Na+,K+-ATPases; I(t), current recorded in the experiment.
One of the sides of the membrane with the Na+,K+-ATPase in this case faces the cleft between the latter and the BLM. Such structure does not allow controlling the potential on the membrane with the Na+,K+-ATPase and recording the direct electric current (dc) passing through the latter; only an alternating current (ac) can be recorded there under short circuit conditions. It is due to the fact that charge shift in the protein leads to a change in the charge of electrical capacitance of the Na+,K+-ATPase-containing membrane fragment and, for its compensation, in the external circuit there is a charge shift in the reverse direction, as is recorded in the experiment. The transient currents in the Na+,K+-ATPase were initiated using the so-called “Caged” compounds [33], where the substrate necessary for the Na+,K+-ATPase functioning was quickly released from the inactive complex under the exposure to UV light flash. The most popular compound was Caged-ATP that made it possible to “switch on” the Na+,K+-ATPase owing to rapid release of ATP molecules. Later on, other compounds were used in the study of the Na+,K+-ATPase: Caged-P for the rapid release of phosphate [34], as well as Caged-H+ for quick changes in pH.
This model system was used at the Laboratory of Bioelectrochemistry to study the Na+,K+-ATPase. The studies started at the initiative of Yu.A. Chizmadzhev. Great assistance was provided by Professor H.J. Apell from the University of Konstanz (Germany). At the beginning of the research it was already known that electric currents appear in the Na+,K+-ATPase in the absence of potassium ions, when the protein cannot perform a full cycle of functioning, and the main contribution to the electric current under these conditions is made by the transfer of sodium ions in the access channel open on the extracellular side of the protein. Such transfer results in the appearance of transient currents, which were observed in the experiments with cells in response to the abrupt change in membrane potential. The controlled application of potential to the membrane fragments adsorbed on BLM is impossible, and transient currents were observed in response to the rapid release of ATP from Caged-ATP. To study the effect of electric field on these transient currents, an original approach was developed based on the application of a sinusoidal alternating voltage to the membrane, resulting in the appearance of the alternating component of the current. In the experiment, this component of the current could be measured as a change in capacitance and conductance of the membrane with adsorbed fragments containing Na+,K+-ATPase [35] in response to the rapid release of ATP from Caged-ATP. This approach made it possible to study the sodium current in the Na+,K+-ATPase within a wide range of sodium ion concentrations. In addition to the transfer of sodium ions in the extracellular channel, which was rather well-studied, this approach made it possible to detect the transfer of sodium ions in the intracellular channel. Such process results in appearance of an electric current of a lower value compared to the current assigned to the movement in the extracellular channel because the depth of the intracellular channel is considerably less than the depth of the extracellular one. The movement of sodium ions in this channel occurs in the absence of ATP; but after the binding and hydrolysis of ATP, this channel closes and the extracellular channel opens. This, ion transport in the intracellular channel could not be detected in the experiments with cells, where electric currents were recorded in the presence of ATP. The experiments carried out by this method made it possible to investigate in detail the movement of sodium ions both in the extracellular and intracellular channels, to determine the rate of movement and the relative depth of the channels (dielectric coefficient). For this purpose, with direct participation of Yu.A. Chizmadzhev, a theoretical model of the movement of sodium ions in the Na+,K+-ATPase channels was developed [36] (Fig. 3).
Theoretical model of nonstationary transport of sodium ions by the Na+,K+-ATPase [36]. Left, the set of states of the Na+,K+-ATPase during ATP hydrolysis. c, cytoplasmic side of the system; e, extracellular side. E1 and E2 designate the protein in two different conformations. On the right, the dynamic potential barriers introduced into consideration by Lauger and Apell [19, 24]. The sodium ion involved in the transport is marked with a black circle.
THEORETICAL MODEL OF SODIUM ION MOVEMENT IN THE CHANNELS OF Na+,K+-ATPase
The transport of sodium ions is considered as a transition between several protein states with the access channels for ions open at the cytoplasmic (designated as E1) or extracellular (E2) sides of the membrane. From the physical point of view, all states of the protein can be presented as a sequential change of potential barriers for sodium ions in the membrane. There is also a state of occlusion when the ion is locked (is in a potential pit between two high potential barriers). In all other states, only one of these high barriers remains, closing the channels from either extracellular (states c0 and c1) or cytoplasmic side (states 2 and 3). Only the movements of sodium ions in the cytoplasmic and extracellular access channels are considered to be electrogenic (the transitions between states c0 and c1, as well as between states 2 and 3). These transitions are Na+ diffusion or migration in the access channels when there are no conformational rearrangements in the protein. The movement of ions in the channels occurs due to the application of electric potential to the membrane and results in appearance of an electric current in the external circuit. If the electric field and the concentration of Na+ ions in the channels are low, the ion transport in these channels can be considered as the hops over low (compared to those considered above) potential barriers. Mathematically, the transitions between the states were represented by differential equations, the solution of which made it possible to determine the changes in capacitance and conductance of the membrane depending on the concentration of sodium ions and the frequency of applied alternating voltage. This explained the experimental results obtained in a model system consisting of Na+,K+-ATPase-containing membrane fragments adsorbed on BLM.
IMPEDANCE MEASUREMENTS ON BLM
In the experiment, the alternating voltage was applied to the membrane and the kinetics of current during the photoinduced release of ATP from Caged-ATP were recorded [37]. The time course of short-circuit current, its integral, as well as changes in the capacitance and conductance of BLM with the adsorbed fragments containing Na+,K+-ATPase in response to ATP release from Caged-ATP due to UV light flash are shown Fig. 4. The records are presented at two concentrations of sodium ions: 150 mM (left) and 3 mM (right). At a high concentration of sodium ions, the ATP release causes an increase in the membrane capacitance and conductance, while at a low concentration, the capacitance decreases. The decrease in the membrane capacitance at low concentration of sodium ions, in accordance with the model considered above, is due to recording the difference signal in the experiment: the alternating current measured after the introduction of ATP is subtracted from the current measured in the absence of ATP. Therefore, the negative increment of capacitance and conductance can be caused by the electrogenic ion transport, which is suppressed during ATP hydrolysis and subsequent phosphorylation of the Na+,K+-ATPase. Such transport is an electrogenic movement of ions in the cytoplasmic channel of the Na+,K+-ATPase.
Signals recorded at two concentrations of sodium ions: 150 mM (left) and 3 mM (right). From top to bottom: the current recorded in the experiment, the integral of this current, and the calculated changes in capacitance and conductance over time, used to determine changes in membrane admittance. Arrows indicate the moments of UV light flashes. Aqueous solutions contained: 150 mM NaCl, 10 mM MgCl2, 30 mM imidazole, 1 mM EDTA, pH 6.5, as well as 100 µM Caged-ATP and the suspension of fragments with Na+,K+-ATPase (0.05 mg protein per 1 mL solution).
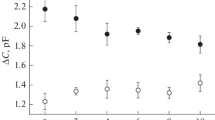
The dependences of the increments of the membrane capacitance and conductance on the frequency of alternating voltage were measured at different sodium ion concentrations in the range from 3 mM to 1 M. According to the model under consideration, the frequency dependences of the increments of capacitance ΔC and conductance ΔG were described by the linear combination of the Lorenz functions with different characteristic frequencies. In the frequency domain, these functions are equivalent to the exponents in the time domain, which were used to describe transient currents after a sudden change in potential in the experiments on cells. Each of these functions represents the transition of protein between the two states, while the respective characteristic frequency represents the rate of such transition. Characteristic frequency ω0 represents the rate of conformational transition from E1 to E2. This is the slowest stage. Characteristic frequencies ω1 and ω2 represent the rate of movement of sodium ions in the extracellular and cytoplasmic access channels, respectively. The amplitudes of these functions depend on sodium ion concentrations, reaching the maximum value at a concentration close to the dissociation constant of ion binding site at this stage. The amplitude of C2 with the negative sign represents the contribution to the alternating current of the movement of sodium ions in the cytoplasmic channel, which closes after the transition of the Na+,K+-ATPase into state E2 as a result of ATP hydrolysis:
As a result of approximation of the frequency dependencies of capacitance and conductance by these functions, the values of their parameters at different concentrations of sodium ions in solution were obtained. The dependences of the C0, C1, C2 amplitudes and the characteristic frequencies ω0, ω1, ω2, of three Lorenzians on the concentration of sodium ions are shown in Fig. 5.
The dependence of amplitudes (above) and characteristic frequencies (below) of the Lorenz functions approximating the frequency dependences of changes in capacitance and conductance, caused by stepwise release of ATP during the functioning of the Na+,K+-ATPase, on the concentration of sodium ions in the solution. Dots represent the average results of at least three experiments. Solid lines are theoretical curves [37].
The assignment of the characteristic frequency ω0 to the rate of conformational transition E1–E2 of the Na+,K+-ATPase was confirmed by studying of the effects of high salt concentrations on the kinetics of protein functioning. It was shown that the salt at high concentrations slows this conformational transition [38]. The experiments on BLM performed by the techniques used have shown that the increase in the concentration of anions Cl–, Br–, or I– leads to a considerable decrease in the value of ω0 [39]. The values of the rates ω0 and ω1 proved to be several times lower than the time constants of exponents approximating the transient currents measured by the voltage clamp method at a squid giant axon [26]. Such difference can be caused by several factors. One of them may be significant temperature dependence of the Na+,K+-ATPase: measurements on cells were usually performed at about 37°C, whereas in the present work the measurements were performed at lower temperatures (20–25°C).
The Na+ dissociation constant in the binding site, 1/Ke, turned out to be 0.74 M on the extracellular side and 1.5 mM on the intracellular side. These values are close to the published data obtained with fluorescent probes, where they were 500 mM and 1–5 mM, respectively (in the latter case, the value depended on the concentration of magnesium ions) [40, 41]. The constants of the rate of binding and release of sodium ions in the cytoplasmic channel have been determined for the first time: the former (k12, the rate constant of the transition from state c1 to state c2) is about 2.4 s–1, the latter (k21, the reverse transition) is about 500 s–1. The rate constant of the transition between states 2 and 3 (the release of Na+ from the extracellular access channel) is about 1500 s–1. The dielectric coefficient of sodium movement (characterizing the relative depth of the channel connecting the solution with the binding site) on the cytoplasmic side (0.2) proved to be much less than that on the extracellular side (0.89). Thus, the binding site of the third sodium ion is localized in the membrane asymmetrically and is closer to the cytoplasmic side of the membrane. Although the estimates obtained are rather coarse, they are in good agreement with the estimates of dielectric coefficients obtained by independent methods, where the values of dielectric coefficients were 0.25 for the cytoplasmic channel and 0.7 for the extracellular channel [18, 42, 43].
INVOLVEMENT OF PROTONS IN THE FUNCTIONING OF THE Na+,K+-ATPase
Subsequent works were devoted to a more detailed study of the binding and transport of sodium and potassium ions on the cytoplasmic side of the protein. It has been shown that ion binding occurs in exchange for protons. The proton transport and the effect of pH on the transport of other ions by the Na+,K+-ATPase were investigated by several research teams [44–47]. It has been shown that pH not only affects the rate of the Na+,K+-ATPase functioning, but protons are involved in the transport cycle and, under particular conditions, can be transferred by the protein instead of sodium and potassium ions. This can be expected because of homological and structural similarity between the Na+,K+-ATPase and other P-type ATPases capable to transport protons across the membrane, especially the K+,H+-ATPase. The latter is most similar in structure to Na+,K+-ATPase, and under certain conditions they can “substitute” for each other [48, 49]. The involvement of protons in the Na+,K+-ATPase functioning is also indicated by X-ray structural data showing that ion binding site in the protein involves negatively charged dissociated amino acid residues, which can be in a protonated state at low ion concentrations [12]. Our studies carried out on BLM with the application of alternating voltage have shown that ATP-triggering electric currents depend on pH and can be observed in the absence of Na+ ions, suggesting the possibility of proton transport by the Na+,K+-ATPase [50]. Another compound, Caged-H+, was used instead of Caged-ATP to initiate electric signals for the more detailed study of the involvement of protons in the Na+,K+-ATPase functioning. This makes it possible to study the displacement currents in the cytoplasmic channel of the Na+,K+-ATPase caused by a rapid pH jump. As a result of these studies, we have succeeded in determining the dissociation constants of sodium and potassium ions in the binding site on the cytoplasmic side of the protein [51–53].
CONCLUSIONS
The impedance method used to study active ion transport by the Na+,K+-ATPase allowed us to obtain significant information on the mechanism of this transport, in particular, to estimate kinetic and equilibrium parameters of ion binding, as well as the relative depths of channels connecting ion binding sites with solutions on both sides of the protein.
REFERENCES
Mueller P., Rudin D.O., Tien H.T., Wescott W.C. 1963. Methods for the formation of single bimolecular lipid membranes in aqueous solution. J. Phys. Chem. 67, 534–535.
Markin V.S., Chizmadzhev Yu.A. 1974. Indutsirovannyi ionny transport (Induced ion transport). Moscow: Nauka.
Markin V.S., Sokolov V.S. 1986. Membrane potential during coupled electrogenic transport. Thermodynamic consideration. Biol. Membrany (Rus.). 3, 638–649.
Sokolov V.S., Markin V.S. 1984. Electrogenic transport of potassium and hydrogen ions across the membrane performed by antibiotics nigericin and grizorixin. Biol. Membrany (Rus.). 1 (10), 1071–1086.
Markin V.S., Sokolov V.S. 1990. A new concept of electrochemical membrane equilibrium. Coupled transport and membrane potential. Bioelectrochem. Bioenerg. 23, 1–16.
Portnov V.I., Mirsky V.M., Markin V.S. 1990. Bacteriorhodopsin: Current-voltage characteristics. Bioelectrochem. Bioenerg. 23, 45–63.
Post R.L., Hegyvary C., Kame S. 1972. Activation by adenosine triphosphate in the phosphorylation kinetics of sodium and potassium ion transport adenosine triphosphatase. J. Biol. Chem. 247, 6530–6540.
Morth J.P., Pedersen B.P., Toustrup-Jensen M.S., Sorensen T.L., Petersen J., Andersen J.P., Vilsen B., Nissen P. 2007. Crystal structure of the sodium-potassium pump. Nature. 450 (7172), 1043–1049.
Ogawa H., Shinoda T., Cornelius F., Toyoshima C. 2009. Crystal structure of the sodium-potassium pump (Na+,K+-ATPase) with bound potassium and ouabain. Proc. Natl. Acad. Sci. USA. 106 (33), 13742–13747.
Shinoda T., Ogawa H., Cornelius F., Toyoshima C. 2009. Crystal structure of the sodium-potassium pump at 2.4 A resolution. Nature. 459 (7245), 446–450.
Kanai R., Ogawa H., Vilsen B., Cornelius F., Toyoshima C. 2013. Crystal structure of a Na+-bound Na+,K+-ATPase preceding the E1P state. Nature. 502 (7470), 201–206.
Toyoshima C., Kanai R., Cornelius F. 2011. First crystal structures of Na+,K+-ATPase: New light on the oldest ion pump. Structure. 19 (12), 1732–1738.
Nyblom M., Poulsen H., Gourdon P., Reinhard L., Andersson M., Lindahl E., Fedosova N., Nissen P. 2013. Crystal structure of Na+,K+-ATPase in the Na+-bound state. Science. 342 (6154), 123–127.
Bublitz M., Morth J.P., Nissen P. 2011. P-type ATPases at a glance. J. Cell Sci. 124 (Pt 15), 2515–2519.
Axelsen K.B., Palmgren M.G. 1998. Evolution of substrate specificities in the P-type ATPase superfamily. J. Mol. Evol. 46 (1), 84–101.
Palmgren M.G., Axelsen K.B. 1998. Evolution of P-type ATPases. Biochim. Biophys. Acta. 1365 (1–2), 37–45.
Pedersen C.N., Axelsen K.B., Harper J.F., Palmgren M.G. 2012. Evolution of plant p-type ATPases. Front. Plant Sci. 3, 31.
Wuddel I., Apell H.J. 1995. Electrogenicity of the sodium transport pathway in the Na,K-ATPase probed by charge-pulse experiments. Biophys. J. 69 (3), 909–921.
Lauger P. 1991. Electrogenic ion pumps. Sunderland, Massachusets, USA: Sinauer Associates, Inc.
De Weer P., Gadsby D.C., Rakowski R.F. 1988. Voltage dependence of the Na-K pump. Ann. Rev. Physiol. 50, 225–241.
Gadsby D.C., Rakowski R.F., De Weer P. 1993. Extracellular access to the Na,K-pump: Pathway similar to ion channel. Science. 260, 100–103.
Apell H.-J., Borlinghaus R., Lauger P. 1989. Electrogenic properties of the Na/K pump-voltage dependence and kinetics of charge translocation. Curr. Top. Membr. Transp. 34, 229–252.
Nakao M., Gadsby D.C. 1986. Voltage dependence of Na translocation by the Na/K pump. Nature. 323 (6089), 628–630.
Apell H.J., Borlinghaus R., Lauger P. 1987. Fast charge translocations associated with partial reactions of the Na,K-pump: II. Microscopic analysis of transient currents. J. Membrane Biol. 97 (3), 179–191.
Pavlov K.V., Sokolov V.S. 2000. Electrogenic ion transport by Na+,K+-ATPase. Membr. Cell Biol. 13 (6), 745–788.
Holmgren M., Wagg J., Bezanilla F., Rakowski R.F., De Weer P., Gadsby D.C. 2000. Three distinct and sequential steps in the release of sodium ions. Nature. 403, 898–901.
Rakowski R.F., Paxson C.L. 1988. Voltage dependence of Na/K pump current in Xenopus oocytes. J. Membrane Biol. 106, 173–182.
Holmgren M., Rakowski R.F. 2006. Charge translocation by the Na+/K+ pump under Na+/Na+ exchange conditions: Intracellular Na+ dependence. Biophys. J. 90 (5), 1607–1616.
Hilgemann D.W. 1994. Channel-like function of the Na,K pump probed at microsecond resolution in giant membrane patches. Science. 263, 1429–1432.
Lu C.-C., Kabakov A., Markin V.S., Mager S., Frazier S., Frazier G.A., Hilgemann D.W. 1995. Membrane transport mechanisms probed by capacitance measurements with megahertz voltage clamp. Proc. Natl. Acad. Sci. USA. 1995, 11220–11224.
Fendler K., Grell E., Haubs M., Bamberg E. 1985. Pump currents generated by the Na+,K+-ATPase from kidney on black lipid membranes. EMBO J. 4, 3079–3085.
Borlinghaus R., Apell H.J., Lauger P. 1987. Fast charge translocations associated with partial reactions of the Na,K-pump: I. Current and voltage transients after photochemical release of ATP. J. Membrane Biol. 97 (3), 161–178.
McCray J.A., Trentham D.R. 1989. Properties and uses of photoreactive caged compounds. Annu. Rev. Biophys. Biophys. Chem. 18, 239–270.
Apell H.J., Roudna M., Corrie J.E., Trentham D.R. 1996. Kinetics of the phosphorylation of Na,K-ATPase by inorganic phosphate detected by a fluorescence method. Biochemistry. 35 (33), 10922–10930.
Sokolov V.S., Pavlov K.V., Dzhandzhugazyan K.N., Bamberg E. 1992. Capacitance and conductivity changes during Na+,K+-ATPase action in model membranes. Biol. Membranes. 6 (9), 1263–1272.
Shcherbakov A.A., Chizmadzhev Yu.A., Lenz A.A., Sokolov V.S. 2005. Impedance spectroscopy of sodium ion transport in Na+,K+-ATPase. Biol. Membrany (Rus.). 22 (6), 511–523.
Sokolov V.S., Shcherbakov A.A., Lenz A.A., Chizmadzhev Yu.A., Apell H.J. 2008. Electrogenic transport of sodium ions in cytoplasmic and extracellular ion access channels of Na+,K+-ATPase probed by admittance measurement technique. Biochem. (Moscow) Suppl. Series A: Membr. Cell Biol. 2 (2), 161–180.
Post R.L., Suzuki K. 1991. The sodium pump: Structure, mechanism and regulation. Ed. De Weer P., Kaplan J.H. New York: Rockfeller University Press, p. 202–209.
Sokolov V.S., Ayuyan A.G., Apell H.J. 2001. Assignment of charge movements to electrogenic reaction steps of Na,K-ATPase by analysis of salt effects on the kinetics of charge movements. Eur. Biophys. J. 30 (7), 515–527.
Schneeberger A., Apell H.J. 2001. Ion selectivity of the cytoplasmic binding sites of the Na,K-ATPase: II. Competition of various cations. J. Membrane Biol. 179 (3), 263–273.
Schneeberger A., Apell H.J. 1999. Ion selectivity of the cytoplasmic binding sites of the Na,K-ATPase: I. Sodium binding is associated with a conformational rearrangement. J. Membrane Biol. 168 (3), 221–228.
Heyse S., Wuddel I., Apell H.J., Sturmer W. 1994. Partial reactions of the Na,K-ATPase: Determination of rate constants. J. Gen. Physiol. 104 (2), 197–240.
Domaszewicz W., Apell H. 1999. Binding of the third Na+ ion to the cytoplasmic side of the Na,K-ATPase is electrogenic. FEBS Lett. 458 (2), 241–246.
Apell H.J., Diller A. 2002. Do H+ ions obscure electrogenic Na+ and K+ binding in the E1 state of the Na,K-ATPase? FEBS Lett. 532 (1–2), 198–202.
Apell H.J., Benz G., Sauerbrunn D. 2011. Proton diet for the sodium pump. Biochemistry. 50 (3), 409–418.
Vasilyev A., Khater K., Rakowski R.F. 2004. Effect of extracellular pH on pre-steady-state and steady-state current mediated by the Na+/K+ pump. J. Membrane Biol. 198 (2), 65–76.
Vedovato N., Gadsby D.C. 2014. Route, mechanism, and implications of proton import during Na+/K+ exchange by native Na+/K+-ATPase pumps. J. Gen. Physiol. 143 (4), 449–464.
Polvani C., Blostein R. 1988. Protons as substitutes for sodium and potassium in the sodium pump reaction. J. Biol. Chem. 263 (32), 16 757–16 763.
Polvani C., Sachs G., Blostein R. 1989. Sodium ions as substitutes for protons in the gastric H,K-ATPase. J. Biol. Chem. 264 (30), 17 854–17 859.
Grishanin K.O., Tashkin V.Yu., Lenz A.A., Apell H.-J., Sokolov V.S. 2010. On potential involvement of protons in the functioning of Na+,K+-ATPase. Biol. Membrany (Rus.). 27 (6), 512–518.
Tashkin V.Yu., Gavril’chik A.N., Ilovaysky A.I., Apell H.-J., Sokolov V.S. 2015. Electrogenic binding of ions at the cytoplasmic side of Na+,K+-ATPase. Biochem. (Moscow) Suppl. Series A: Membr. Cell Biol. 9 (2), 92–99.
Tashkin V.Yu., Shcherbakov A.A., Apell H.-J., Sokolov V.S. 2013. The competition transport of sodium ions and protons at the cytoplasmic side of Na,K-ATPase. Biochem. (Moscow) Suppl. Series A: Membr. Cell Biol. 7 (2), 113–121.
Vishnyakova V.E., Tashkin V.Yu., Terentjev A.O., Apell H.-J., Sokolov V.S. 2018. Binding of potassium ions inside the access channel at the cytoplasmic side of Na+,K+-ATPase. Biochem. (Moscow) Suppl. Series A: Membr. Cell Biol. 12 (4), 344–351.
Funding
The work was supported by the Ministry of Science and Higher Education of the Russian Federation (registration number of the project, 122011300058-3).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest.
This article does not contain any studies involving animals or human participants performed by any of the authors.
Additional information
Translated by E. Makeeva
Rights and permissions
About this article
Cite this article
Sokolov, V.S. Investigations of Electrogenic Ion Transport by Na+,K+-ATPase in Bilayer Lipid Membranes by Impedance Method. Biochem. Moscow Suppl. Ser. A 16, 282–290 (2022). https://doi.org/10.1134/S1990747822050117
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990747822050117