Abstract
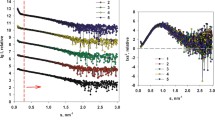
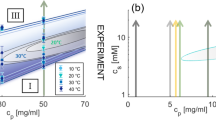
Adsorption of the viral matrix protein M1 on a substrate simulating the lipid membrane surface of the influenza virus was studied by surface plasmon resonance (SPR). It was found that a decrease of pH leads to an increase of the time to reach the saturated level of adsorption, despite the growth of its initial rate. Adsorption of M1 is irreversible in acidic and neutral media, but in the first case the saturated level of adsorption depends on the protein concentration. It was observed that despite the adsorption irreversibility, acidification of the solution down to pH 4 leads to a partial protein desorption from the adsorbed layer that was formed at pH 7. The findings suggest pH-induced changes in shape of the adsorbed M1 molecules. In an acidic medium, elongated protein molecules adsorb mostly laterally in dilute solutions and more orthogonally in concentrated ones. In a neutral medium, protein molecules take on a compact conformation in the adsorption layer, and its thickness does not depend on the concentration. Apparently, flexible C-terminal domain of the adsorbed protein plays a major role in the pH-induced conformational change.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ruigrok R.W.H. Structure of influenza A, B and C viruses. 1998. Textbook of Influenza. Eds. Nicholson K.G., Webster R.G., Hay A.J. Oxford: Blackwell Science, p. 29–42.
Sha B., Luo M. 1997. Structure of a bifunctional membrane-RNA binding protein, influenza virus matrix protein M1. Nat. Struct. Biol. 4 (3), 239–244.
Martin K., Helenius A. 1991. Nuclear transport of influenza virus ribo-nucleoproteins: The viral matrix protein (M1) promotes export and inhibits import. Cell. 67, 117–130.
Ruigrok R.W.H., Calder L.J., Wharton S.A. 1989. Electron microscopy of the influenza virus submembranal structure. Virology. 173, 311–316.
Rossman J.S., Lamb R.A. 2011. Influenza virus assembly and budding. Virology. 411 (2), 229–223.
Gómez Puertas P., Albo C., Perez Pastrana E., Vivo A., Agustín Portela A. 2000. Influenza virus matrix protein is the major driving force in virus budding. J. Virol. 74 (24), 11538–11547.
Ali A., Avalos R., Ponimaskin E., Nayak D.P. 2000. Influenza virus assembly: The effect of influenza viral glycoproteins on the membrane-association of M1 protein. J. Virol. 74, 8709–8719.
Zhang K., Wang Z., Liu X., Yin C., Basit Z., Xia B., Liu W. 2012. Dissection of influenza A virus M1 protein: pH-Dependent oligomerization of N-terminal domain and dimerization of C-terminal domain. PLoS One. 7 (5), e37786. doi:10.1371.
Ruigrok R.W.H., Barge A., Durrer P., Brunner J., Ma K., Whittaker G.R. 2000. Membrane interaction of influenza virus M1 protein. Virology. 267 (2), 289–298.
Compans R.W., Content J., Duesberg P.H. 1972. Structure of the ribonucleoprotein in influenza virus. J. Virol. 10, 795–800.
Pons M.W., Schulze I.T., Hirst G.K., Hauser R. 1969. Isolation and characterization of the ribonucleoprotein oh influenza virus. Virology. 39, 250–259.
Bui M., Whittaker G., Helenius A. 1996. Effect of M1 protein and low pH on nuclear transport of influenza virus ribonucleoproteins. J. Virol. 70, 8391–8401.
Ichimura T., Hatae T., Ishida T. 1997. Direct measurement of endosomal pH in living cells of the rat yolk sac epithelium by laser confocal microscopy. Eur. J. Cell Biol. 74 (1), 41–48.
Arzt S., Baudin F., Barge A., Timmins P., Burmeister W.P., Ruigrok R.W. 2001. Combined results from solution studies on intact influenza virus M1 protein and from a new crystal form of its N-terminal domain show that M1 is an elongated monomer. Virology. 279 (2), 439–446.
Harris A., Forouhar F., Qiu S., Sha B., Luo M. 2001. The crystal structure of the influenza matrix protein M1 at neutral pH: M1-M1 protein interfaces can rotate in the oligomeric structures of M1. Virology. 289 (1), 34–44.
Shishkov A., Bogacheva E., Fedorova N., Ksenofontov A., Badun G., Radyukhin V., Lukashina E., Serebryakova M., Dolgov A., Chulichkov A., Dobrov E., Baratova L. 2011. Spatial structure peculiarities of influenza A virus matrix M1 protein in an acidic solute. FEBS J. 278, 4905–4909.
Shtykova E.V., Baratova L.A., Fedorova N.V., Radyukhin V.A., Ksenofontov A.L., Volkov V.V., Shishkov A.V., Dolgov A.A., Shilova L.A., Batishchev O.V., Jeries C.M., Svergun D.I. 2013. Structural analysis of Influenza A virus matrix protein M1 and its self-assemblies at low pH. PLoS One. 8, e82431. doi: 10.1371/journal.pone.0082431.
Fontana J., Steven A.C. 2013. At low pH, influenza virus matrix protein M1 undergoes a conformational change prior to dissociating from the membrane. J. Virol. 87 (10), 5621–5628.
http://www.uniprot.org/uniprot/P03485
El Karadaghi S., Zakomirdin J., Shimane C., Bucher D., Tverdislov V., Kharitonenkov I. 1984. Interaction of influenza virus proteins with planar bilayer lipid membranes. I. Characterization of their adsorption and incorporation into lipid bilayers. Biochim. Biophys. Acta. 778 (2), 269–275.
Harris A., Cardone G., Winkler D., Heymann J., Brecher M., White J., Steven A. 2006. Influenza virus pleiomorphy characterized by cryoelectron tomography. Proc. Natl. Acad. Sci. USA. 103 (50), 19123–19127.
Knyaziev D.G., Radyukhin V.A., Sokolov V.S. 2008. The study of intermolecular interactions of influenza virus proteins M1 on the surface of the model lipid membrane by the method of intramembranous field compensation. Biomembranes. 25 (6), 488–498.
Helenius A. 1992. Unpacking the incoming influenza virus minireview. Cell. 69, 577–578.
Brevnov V.V., Fedorova N.V., Indenbom A.V. 2013. Adsorption of viral matrix protein M1 in acidic medium. Biomembranes. 30 (3), 214–220.
Zhirnov O.P. 1992. Isolation of matrix M1 from influenza viruses by acid-dependent extraction with nonionic detergent. Virology. 186, 324–330.
Latour R.A. 2008. Biomaterials: Protein–surface interactions. In: The encyclopedia of biomaterials and bioengineering. Wnek G.E., Bowlin G.L., Eds. Informa Healthcare. 1, 270–284.
Latour R.A. 2011. Molecular simulation methods to study protein adsorption behavior at the atomic level. In: Comprehensive biomaterials. Ducheyne P., Hutmacher D.W., Kirkpatrick C.J., Healy K., Eds. Oxford, UK: Elsevier. 3, 171–192.
Van der Vegte E.W., Hadziioannou G. 1997. Acid base properties and the chemical imaging of surface-bound functional groups studied with scanning force microscopy. J. Phys. Chem. B. 101 (46), 9563–9569.
http://www.scripps.edu/~cdputnam/protcalc.html, Protein Calculator v3.4.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.V. Brevnov, N.V. Fedorova, A.V. Indenbom, 2015, published in Biologicheskie Membrany, 2015, Vol. 32, No. 2, pp. 93–101.
Rights and permissions
About this article
Cite this article
Brevnov, V.V., Fedorova, N.V. & Indenbom, A.V. Effects of pH on the adsorption of the viral matrix protein M1. Biochem. Moscow Suppl. Ser. A 9, 84–91 (2015). https://doi.org/10.1134/S199074781502004X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S199074781502004X